Abstract
Background and objective
Albuminuria is a critical early warning marker for the development of chronic kidney disease. The neutrophil-to-high-density lipoprotein cholesterol ratio (NHR) has been identified as a novel biomarker for the assessment of both inflammatory status and lipid metabolism. The present study was designed to explore the association between NHR and albuminuria.
Methods
We conducted a cross-sectional study using data from the National Health and Nutrition Examination Survey between 2017 and 2020. In total, 7772 eligible adult participants were included. Multivariate logistic regression models, threshold effect analysis, and sensitivity and subgroup analyses were performed to analyze the association between NHR and albuminuria.
Results
We found a notable positive association between the NHR and the likelihood of albuminuria. In fully adjusted models, each unit rise in the NHR was associated with a 1.13-fold higher likelihood of albuminuria (1.13 (1.09–1.18)). When classified according to quartiles, participants in the highest NHR quartile had a markedly elevated likelihood of albuminuria compared with those in the lowest quartile (2.01 (1.59–2.53)). The smooth curve analysis suggested a nonlinear relationship, with a threshold at 4.58. In subgroup analyses, the positive association between NHR and albuminuria differed significantly across race categories (interaction test p = 0.0166).
Conclusion
An increased level of NHR was significantly associated with an elevated odds ratio for the occurrence of albuminuria in our study. NHR may be an efficacious biomarker for the assessment of albuminuria risk, and future studies should further investigate its predictive value.
Keywords
Background
Albuminuria is a crucial early marker for kidney damage and a predictor of chronic kidney disease (CKD) progression. 1 Persistent albuminuria often indicates further deterioration of kidney function.2,3 An urinary albumin-to-creatinine ratio (ACR) >30 mg/g is a well-established threshold for diagnosing albuminuria, 4 and its accuracy and usability make it a valuable tool for early assessment of kidney function impairment. 5 Additionally, it is established that albuminuria is an unambiguous risk factor for cardiovascular events. 6 It also serves as an indicator of treatment efficacy for both CKD and cardiovascular diseases. 7 Therefore, early clinical screening and intervention for albuminuria are crucial.
Growing evidence indicates that in addition to systemic inflammation dysregulated lipid metabolism is a key contributor to kidney injury. Recent reviews have shown that ectopic accumulation of cholesterol, triglycerides, and lipid droplets in the kidney parenchyma can induce oxidative stress, activate inflammatory signaling pathways, and promote tubulointerstitial fibrosis, thereby accelerating CKD progression. 8 Furthermore, dysregulation of lipid metabolic pathways in CKD—including altered fatty acid uptake and oxidation, impaired cholesterol efflux, and changes in key regulators such as peroxisome proliferator-activated receptors (PPARs) and sterol regulatory element-binding proteins (SREBPs)—has been linked to inflammation-related renal damage and has emerged as a potential therapeutic target. 9
In this context, blood-cell– and lipid-based inflammatory markers have attracted growing attention. Neutrophil-based indices have been reported to be elevated in several inflammatory conditions, including thyroid disorders, inflammatory bowel disease, coronavirus disease 2019 (COVID-19), diabetes mellitus, gastrointestinal and cardiac diseases, and thyroiditis. Similarly, high-density lipoprotein cholesterol (HDL-C)-based markers have been associated with hypertension, hepatic steatosis, type 2 diabetes, thyroiditis, metabolic syndrome, prediabetes, diabetic retinopathy, diabetic kidney disease, and new-onset diabetes.10–16 Recent reviews further highlight that dysregulated lipid metabolism and chronic inflammation jointly contribute to kidney injury and CKD progression through pathways involving lipotoxicity, oxidative stress, and endothelial dysfunction.17–24 These data support an inflammation–lipid metabolism–kidney injury axis and provide a biological rationale for examining the association between NHR, as a composite marker integrating a neutrophil-based and an HDL-based component, and albuminuria.
The neutrophil-to-HDL cholesterol ratio (NHR) is a noteworthy biomarker that reflects the body’s inflammation and lipid metabolism status. Increasing evidence suggests an association between NHR and various diseases, including diabetes, 25 the severity of acute pancreatitis, 26 metabolic dysfunction-associated steatosis, 27 the prognosis of acute myocardial infarction and coronary artery stenosis,28,29 depression, 30 and periodontitis. 31
It has been previously demonstrated that inflammation is associated with lipid metabolism disorders and impaired kidney function.8,32 Dyslipidemia is evident even in the early stages of CKD. 33 Chronic inflammation disrupts normal lipid metabolism, promoting lipid abnormalities, which in turn activate inflammatory responses, forming a vicious cycle that accelerates kidney damage progression.25,34 However, the relationship between NHR, as a novel and readily accessible index of inflammatory and lipid metabolic processes, and albuminuria remains unclear.
Thus, using data from the National Health and Nutrition Examination Survey (NHANES), we aimed to investigate the relationship between NHR and albuminuria among adults in the United States.
Methods
Study population
The NHANES is a nation-wide representative survey that uses a multistage probability sampling method for the evaluation of the healthcare and nutritional profiles of non-institutionalized inhabitants of the United States. Approval for this study was granted by the National Center for Health Statistics (NCHS), and each participant provided written informed consent under the scrutiny of a rigorous ethical review process.
Our cross-sectional study used the NHANES data from 2017 to 2020. Initially, 15,560 participants were recruited. We excluded 6328 participants who were aged <20 years, 1322 with missing NHR data, and 138 with missing ACR data. Thus, the final sample comprised 7772 eligible participants (Figure 1).

Flow chart of participant selection. NHANES: National Health and Nutrition Examination Survey.
Exposure variable and outcome definition
This study employed NHR as its exposure variable, calculated by dividing the neutrophil count (103 cells/μL) by the HDL-C level (mmol/L). The participants were divided into quartiles according to their NHR values for analysis. Neutrophil counts were assessed using the Beckman Coulter DxH 800 system, while HDL-C concentrations were determined through enzymatic assays performed on the Roche Cobas 6000 and Modular P analyzers.
The outcome variable was albuminuria (ACR >30 mg/g). 35 Urinary albumin and creatinine levels were measured using solid-phase fluorescent immunoassays and modified Jaffe kinetic methods, respectively.
Covariates
Based on previous research, we considered multiple covariates that could confound the relationship between NHR and albuminuria. Covariates included a range of demographic, lifestyle, and clinical factors. Demographic characteristics included sex, age, race, and educational level; socioeconomic indicators included the poverty-to-income ratio and marital status; lifestyle factors included alcohol consumption, smoking status, and physical activity; and clinical markers included lipid profile (low-density lipoprotein cholesterol (LDL-C) and triglycerides), body mass index (BMI), renal function (blood urea nitrogen, uric acid, and glomerular filtration rate), glycemic control indices (glycated hemoglobin and fasting glucose), blood pressure (systolic and diastolic blood pressure), and clinical diagnoses of diabetes, hypertension, hyperlipidemia, and kidney stones. All NHANES data are publicly available at the Centers for Disease Control and Prevention (CDC) website (https://www.cdc.gov/nchs/nhanes/).
Statistical analyses
For analyzing the data obtained from NHANES, we accounted for its complex sampling design. Analysis of weighted variance was employed for continuous variables, whereas weighted chi-squared tests were used to compare categorical variables and identify differences between groups across NHR quartiles. Multivariable survey-weighted logistic regression models were fitted to examine the association between NHR and albuminuria. An unadjusted model (Model 1) was used as the baseline; Model 2 was adjusted for age, sex, and race; while Model 3 was adjusted for all covariates. Sensitivity analyses were conducted by dividing NHR into quartiles. To assess potential nonlinearity in the association between NHR and albuminuria, we first fitted a smooth curve using a generalized additive model, with NHR as a continuous predictor in survey-weighted logistic regression. When a nonlinear pattern was observed, we further applied a two-segment (piecewise) logistic regression model with a single breakpoint in the NHR. The breakpoint and its 95% confidence interval (CI) were estimated using maximum likelihood methods implemented in Empower software. Model fit for the linear versus two-segment models was compared using likelihood ratio tests, and statistical significance was reported as log likelihood ratio (LLR) p-values. Finally, subgroup analyses were performed using multivariate logistic regression stratified by sex, age, race, BMI, diabetes, and hypertension status to ascertain the stability of the association. The threshold for statistical significance was set at p < 0.05. All analyses were conducted using R software (version 4.1.3) and Empower software. Weighted analyses accounted for the complex sampling design of the NHANES, and weighted percentages were reported for categorical variables. Missing values for continuous covariates were imputed using the median, and missing values for categorical covariates were imputed using the most frequent category.
Results
Baseline characteristics
Table 1 presents the weighted distribution of participants’ clinical characteristics across NHR quartiles. The study included 7772 adult participants, with a mean age of 48.27 ± 17.22 years. The group comprised 48.34% men and 51.66% women, consistent with the weighted overall distribution in Table 1. Weighted prevalence by NHR quartiles is shown in Table 1. The NHR quartile ranges were 0.19–2.06, 2.06–2.98, 2.98–4.18, and 4.18–16.77. The overall prevalence of albuminuria was 10.21%, with 7.24%, 7.87%, 10.70%, and 14.67% in the first to fourth quartiles, respectively.
Baseline characteristics of the study population.

Mean ± SD for continuous variables: the p value was calculated using the weighted linear regression model; (%) for categorical variables: the p value was calculated using the weighted chi-square test. NHR: neutrophil to high-density lipoprotein cholesterol ratio; LDL: low-density lipoprotein; eGFR: estimated glomerular filtration rate.
Association between the NHR and albuminuria
As demonstrated in Table 2, elevated NHRs were linked to an elevated probability of albuminuria. The unadjusted model (1.18 (1.14, 1.22)), demographic-adjusted model (1.25 (1.20, 1.30)), and fully adjusted model (1.13 (1.09, 1.18)) all demonstrated a positive association between NHR and albuminuria. Sensitivity analysis of NHR quartiles confirmed this positive association, with statistically significant results. Compared with the lowest NHR quartile, the fourth quartile demonstrated an approximately two-fold increased likelihood of albuminuria occurrence (2.01 (1.59, 2.53); p for trend <0.0001). The smooth-fitting curve revealed a nonlinear relationship between NHR and albuminuria (Figure 2). The two-segment linear regression model identified a threshold at 4.58, with a significant LLR p-value of 0.024 (Table 3).
Associations between NHR and albuminuria.
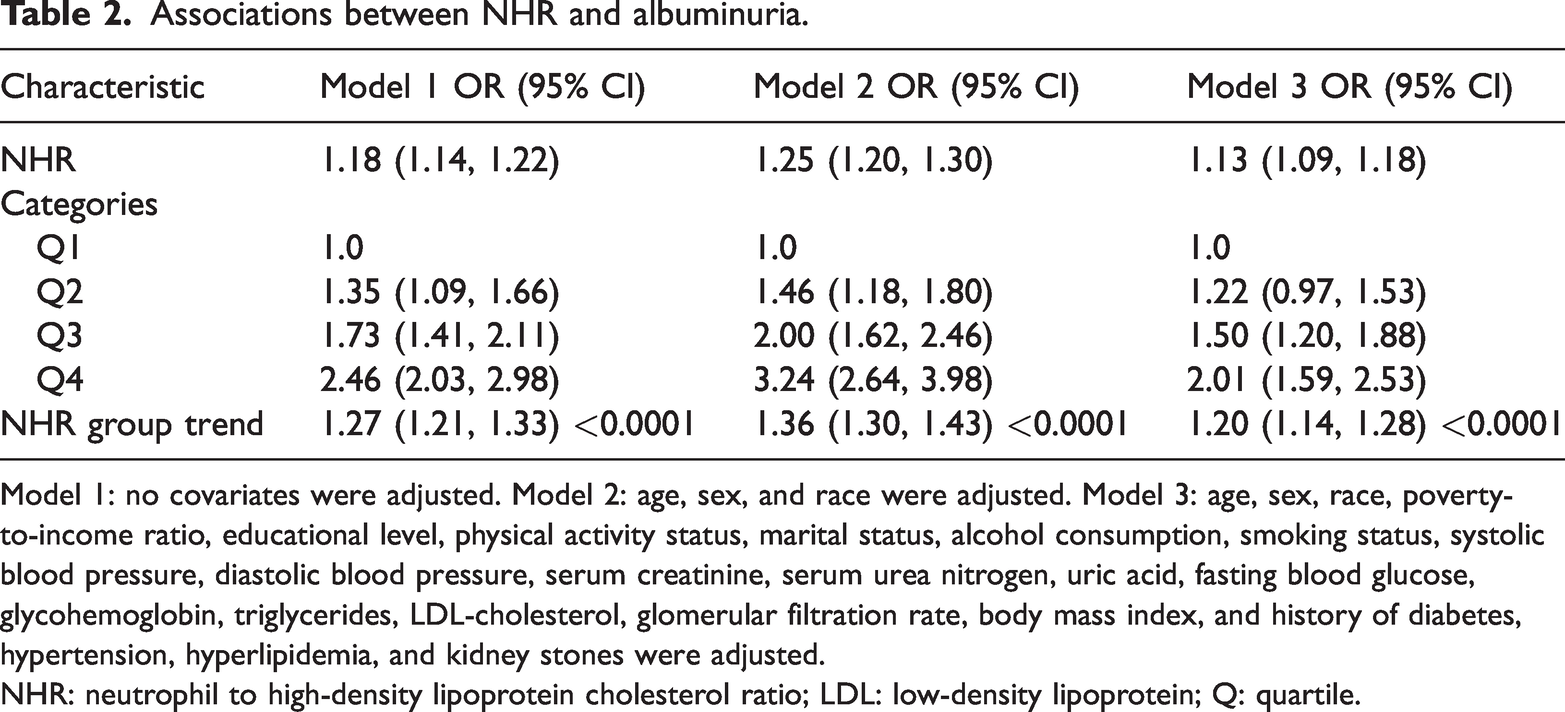
Model 1: no covariates were adjusted. Model 2: age, sex, and race were adjusted. Model 3: age, sex, race, poverty-to-income ratio, educational level, physical activity status, marital status, alcohol consumption, smoking status, systolic blood pressure, diastolic blood pressure, serum creatinine, serum urea nitrogen, uric acid, fasting blood glucose, glycohemoglobin, triglycerides, LDL-cholesterol, glomerular filtration rate, body mass index, and history of diabetes, hypertension, hyperlipidemia, and kidney stones were adjusted.
NHR: neutrophil to high-density lipoprotein cholesterol ratio; LDL: low-density lipoprotein; Q: quartile.

The nonlinear association between the NHR and albuminuria. The solid red line represents the smooth curve fit between variables. Blue bands represent the 95% of confidence interval from the fit.
Threshold effect analysis of NHR on albuminuria using a two-piecewise linear regression model.

The model was adjusted for sex, age, race, poverty-to-income ratio, educational level, physical activity status, marital status, alcohol consumption, smoking status, systolic blood pressure, diastolic blood pressure, serum creatinine, serum urea nitrogen, uric acid, fasting blood glucose, glycohemoglobin, triglycerides, LDL-Cholesterol, glomerular filtration rate, body mass index, and history of diabetes, hypertension, hyperlipidemia, and kidney stones.
NHR: neutrophil to high-density lipoprotein cholesterol ratio; LDL: low-density lipoprotein.
Subgroup analyses
Subgroup analyses were performed to confirm the robustness of the positive association between NHR and albuminuria in the fully adjusted model. The results revealed differences in the association among racial subgroups, with interaction p-values <0.05 indicating statistical significance. In race-stratified analyses, the association estimates were larger in the Mexican American group (OR: 1.21 (95% CI: 1.09–1.36)) and other race group (OR: 1.28 (95% CI: 1.16–1.40)) (interaction test p = 0.0166). In other subgroups, the association between NHR and albuminuria remained consistent, and no statistically significant interaction p-values were observed, suggesting that the relationship between NHR and albuminuria was stable across sex, age, BMI, diabetes status, and hypertension status groups (Table 4).
Subgroup analysis of the association between NHR and albuminuria.

The model was adjusted for sex, age, race, poverty-to-income ratio, educational level, physical activity status, marital status, alcohol consumption, smoking status, systolic blood pressure, diastolic blood pressure, serum creatinine, serum urea nitrogen, uric acid, fasting blood glucose, glycohemoglobin, triglycerides, LDL-cholesterol, glomerular filtration rate, body mass index, and history of diabetes, hypertension, hyperlipidemia, and kidney stones.
NHR: neutrophil to high-density lipoprotein cholesterol ratio; LDL: low-density lipoprotein.
Discussion
This cross-sectional study involving 7772 US adults aimed to evaluate the association between NHR and albuminuria. We found that heightened NHR levels were associated with an elevated probability of albuminuria. The smooth curve analysis suggested a nonlinear relationship between NHR and albuminuria, with a threshold at 4.58. Subgroup analyses revealed that the association was consistent across most subgroups; however, it was more pronounced in the Mexican American and other race groups. The findings imply that NHR is a potential predictive marker for albuminuria. Therefore, early screening for albuminuria in individuals with elevated NHR levels and clinical intervention may help mitigate abnormal albumin excretion.
NHR is a newly developed comprehensive indicator for assessing inflammation and lipid metabolism. Its applications are already well established in several disciplines, particularly in cardiovascular research.36–38 Albuminuria not only serves as an early marker of kidney damage but may also indicate systemic endothelial dysfunction, suggesting the presence of underlying vascular lesions. 39 Previous studies have shown that inflammation and dyslipidemia are related to kidney damage. A cohort study involving 4962 participants followed up for up to 15 years revealed that higher levels of inflammatory markers were linked to an increased risk of CKD. 40 Another cross-sectional study involving 36,463 participants found that elevated systemic immune-inflammatory index levels were linked to an increased probability of albuminuria. 41 Dyslipidemia, especially imbalances in the HDL-C-to-triglyceride ratio, increases the risk of atherosclerosis, leading to microvascular damage in the kidneys. 34 A longitudinal cohort study from China indicated that residual cholesterol mediated by inflammatory status was positively associated with CKD, highlighting the importance of inflammation and dyslipidemia in kidney function impairment.
NHR, as a novel marker of inflammation and lipid metabolism, has shown a significant positive association with albuminuria in our study. Although the exact mechanisms underlying this association remain unclear, we can draw insights from previous research. Neutrophils, as nonspecific immune cells, play a role in conditions involving chronic metabolic inflammation, such as cardiovascular disease, diabetes, and insulin resistance.42–44 Activated neutrophils release neutrophil elastase, which directly damages kidney cells and releases proinflammatory cytokines that exacerbate inflammation.45–47 This process disrupts the integrity of the glomerular basement membrane and tubular epithelial cells, leading to impaired glomerular filtration and albumin leakage, resulting in albuminuria. In an experimental study involving diet-induced obese mice, neutrophils, through the release of neutrophil elastase and proinflammatory cytokines, directly contributed to insulin resistance, particularly by degrading insulin receptor substrate-1 and inhibiting insulin signaling pathways. 48 This not only affects systemic metabolism but also negatively impacts glucose metabolism in the kidneys, enhancing the progression of kidney damage. HDL-C plays a role in reverse cholesterol transport and exerts anti-inflammatory and endothelial-protective effects.49–51 When HDL-C levels decrease, cholesterol transport is impaired, leading to increased cholesterol deposition in kidney micro vessels, which promotes microvascular sclerosis and narrowing. HDL-C also plays a role in inhibiting inflammatory cell activation and reducing the production of proinflammatory cytokines, such as interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α).51,52 Chronic inflammation is a key factor in glomerular sclerosis and fibrosis. When HDL-C levels are low, its anti-inflammatory and antioxidant abilities are weakened, and the number of inflammatory factors increases, promoting inflammatory responses that damage the glomerular basement membrane and increase the likelihood of albuminuria. Additionally, HDL-C exerts varying degrees of anti-atherosclerotic effects,53,54 promoting nitric oxide synthesis to maintain normal vascular relaxation and improve endothelial dysfunction, thereby maintaining normal kidney blood flow regulation. 54 HDL-C also activates the phosphatidylinositol 3-kinase/Akt pathway and upregulates the anti-apoptotic protein Bcl-2, protecting endothelial cells from apoptosis. 55 HDL-C plays a critical role in protecting vascular endothelial cells. When HDL-C levels decrease, these protective mechanisms are weakened, making the kidneys more susceptible to chronic inflammation, oxidative stress, and microvascular damage, eventually leading to kidney damage and CKD progression. The interaction between inflammation and dyslipidemia in CKD creates a vicious cycle, accelerating the progression of kidney damage.34,56,57 Therefore, we believe that there is an association between NHR and albuminuria.
We observed that in race-stratified analyses, the association between NHR and albuminuria was more pronounced in the Mexican American and other race groups, and the interaction test for race was statistically significant (interaction test p = 0.0166). This finding suggests that race modifies the relationship between NHR and albuminuria, and the observed heterogeneity may not be attributable to chance alone. Our findings are broadly consistent with those of previous studies showing that composite inflammatory ratios, such as the neutrophil-to-lymphocyte and platelet-to-lymphocyte ratios, are associated with adverse renal outcomes, including CKD progression and albuminuria, and that the burden of both inflammation and kidney disease differs across racial and ethnic groups. This could be attributed to differences in genetic susceptibility, environmental factors, and lifestyle factors across races, which may influence inflammation and lipid metabolism. In future applications of NHR as a predictive marker for albuminuria, racial differences may need to be considered. The clinical use of NHR may require race-specific adjustments to improve its accuracy.
Several nonmutually exclusive mechanisms may underlie this effect modification. First, racial and ethnic differences in lipid metabolism and inflammatory pathways, potentially influenced by genetic variation in apolipoprotein and cytokine genes, could alter the balance between pro- and anti-inflammatory processes captured by the NHR. Second, environmental and clinical factors that vary by race or ethnicity—such as dietary patterns (e.g. high intake of fructose or sugar-sweetened beverages), obesity, diabetes, and conditions such as obstructive sleep apnea—may amplify the impact of systemic inflammation and dyslipidemia on glomerular and endothelial injury, thereby strengthening the NHR–albuminuria pathway in specific populations. These explanations remain hypothesis-generating and require confirmation in future studies designed to disentangle genetic, environmental, and sociocultural contributors to race-related disparities in kidney disease.
The strengths of our study lie in its large, nationally representative sample, which boosts the generalizability of our findings. However, certain limitations should be considered. As a cross-sectional study, it does not allow us to establish a causal link between NHR and albuminuria. In addition, both NHR and albuminuria were assessed at a single time point in the NHANES, which may not reflect long-term biological variability and could lead to misclassification of exposure or outcome. Although we adjusted for a broad set of covariates, residual confounding cannot be excluded. In particular, detailed information on factors that may influence both NHR and albuminuria, including diet, use of medications such as statins and antihypertensive agents, and inflammatory comorbidities, was limited. Moreover, we could not assess oxidative stress markers, measures of endothelial function, or cytokine profiles (e.g. IL-6 and TNF-α), which could provide additional mechanistic insights into the observed association and should be incorporated in future studies despite the significant positive association observed in our results. Finally, although the NHANES employs a large, nationally representative sample, our subgroup findings—particularly those observed among individuals of Mexican American ethnicity—should be interpreted with caution and require confirmation in larger, ethnically diverse, and preferably longitudinal cohorts.
Conclusion
As a novel marker for assessing inflammation and lipid metabolism, NHR provides new insights into the interplay between systemic inflammation, dyslipidemia, and albuminuria. Further extensive prospective studies are essential to corroborate our findings
Footnotes
Acknowledgments
We would like to thank all the study participants.
Author contributions
PP and JW designed the research. PP, JW, and JW collected and analyzed the data as well as drafted the manuscript. KL revised the manuscript. All authors contributed to the article and approved the submitted version.
Consent for publication
Not applicable.
Clinical trial number
Not applicable.
Data availability
Declaration of conflicting interests
All authors declare that they have no competing financial interests.
Ethical statement
The portions of this study involving human participants, human materials, or human data were conducted in accordance with the Declaration of Helsinki and were approved by the National Center for Health Statistics (NCHS) Ethics Review Board. The patients/participants provided their written informed consent to participate in this study.
Funding
The study was funded by the Evaluation of Flight Adaptability and Individualized Treatment for Brain Disorders in High-Performance Fighter Pilots (BKJTS221J007) and Development of a Rapid Detection Technology for Brain Injury Biomarkers (21-163-12-ZT-006-002-16).
