Abstract
Objective
The purpose of this study was to assess whether circulating soluble ST2 independently predicts prognosis in patients with chronic heart failure.
Methods
This study was registered with the International Prospective Register of Systematic Reviews (PROSPERO) under the unique registration number CRD42023489018. Two researchers systematically searched PubMed, Embase, and Web of Science for all studies published up to 1 September 2024. To evaluate the quality of the study, the Newcastle–Ottawa Scale was used; Review Manager software was used for statistical analysis and construction of forest plots.
Results
The final analysis comprised 17 studies in total. This meta-analysis demonstrated that a high soluble ST2 level was a predictor of poor all-cause mortality (hazard ratio: 1.03, 95% confidence interval: 1.02–1.04, p < 0.00001), poor all-cause mortality/heart failure-related readmission (hazard ratio: 1.46, 95% confidence interval: 1.33–1.61, p < 0.00001), and higher cardiovascular mortality/heart failure-related hospitalization (hazard ratio: 1.50, 95% confidence interval: 1.30–1.74, p < 0.00001) in patients with chronic heart failure. Subgroup analyses were conducted based on ethnicity, sex, left ventricular ejection fraction, and follow-up duration for both all-cause mortality and all-cause mortality/heart failure-related readmission. Soluble ST2 demonstrated good prognostic value in all subgroups.
Conclusion
This study, based on current evidence, suggests that soluble ST2 has independent prognostic value in patients with chronic heart failure. The soluble ST2 biomarker performed well in predicting all-cause mortality/heart failure-related readmission and cardiovascular mortality/heart failure-related hospitalization. Further research is needed to validate its role in clinical practice.
Introduction
Heart failure (HF), which marks the culmination of many cardiac pathological states greatly elevates all-cause mortality (ACM), raises the risk of repeat hospitalization associated with HF, and places a heavy burden on the global health economy as the number of patients continues to rise. 1 HF is a serious medical condition owing to its high morbidity and lethality. Moreover, once HF progresses to the chronic stage, the biological plasticity of the heart, which may self-repair, contributes to disease irreversibility and treatment complexity. 2 Ventricular remodeling, as a key biological process in HF progression, reflects profound changes in the structure and function of the heart, wherein the conversion of cardiac fibroblasts to myofibroblasts not only accelerates cardiac fibrosis but also further deteriorates the pumping function of the heart. 3 In this context, the search for biomarkers that can accurately predict the prognosis of patients with HF from the time of treatment initiation has become crucial in the management of HF.4,5 HF can be categorized into two distinct phenotypes based on ejection fraction: (a) HF with preserved ejection fraction (HFpEF) and (b) HF with reduced ejection fraction (HFrEF).6,7 HFpEF is characterized by a preserved left ventricular ejection fraction (LVEF) >50%, typically associated with diastolic dysfunction, where the heart’s ability to relax and fill with blood is impaired. In contrast, HFrEF is defined by a reduced LVEF of <40%, primarily associated with systolic dysfunction, where the heart’s pumping ability is compromised. 8 These two forms of HF differ not only in their underlying pathophysiological mechanisms but also in clinical presentation, prognosis, and treatment response. The distinct nature of these conditions underscores the importance of identifying biomarkers such as soluble ST2 (sST2) that could serve as prognostic tools in both HFpEF and HFrEF populations. 9
sST2 is a circulating cytokine that acts as a decoy receptor for interleukin (IL)-33. It is produced when the ST2 receptor (also known as IL1 receptor-like (RL) 1) is cleaved from the cell surface, and it plays an important role in regulating the inflammatory and fibrotic processes within the heart. sST2 interferes with the cardioprotective effects of IL-33 by preventing its binding to the membrane-bound receptor ST2L.10,11 Therefore, sST2 is not only of interest as a biomarker of cardiac fibrosis and HF progression but also as a potential indicator for assessing HF prognosis, which deserves in-depth investigation. 12 Although the correlation between baseline sST2 levels and increased risk of HF has been widely reported in the literature, the inconsistencies in these studies with respect to the length of follow-up period limit the generalizability of the findings.9,13,14 Contrary to previous meta-analyses, we performed a multidimensional analysis that included subgroup evaluations based on ethnicity, sex, LVEF, and follow-up duration, offering a deeper understanding of the predictive potential of sST2. Through systematic assessment and meta-analysis of existing studies, this study comprehensively evaluated the impact of sST2 as a therapeutic target on both long-term and short-term prognoses of patients with chronic HF (CHF). Furthermore, the results of the subgroup analyses are suggestive and require further investigation for validation.
Methods
Protocol
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 15 and Assessing the Methodological Quality of Systematic Reviews 16 criteria were followed in the reporting of this work. This study was registered with the International Prospective Register of Systematic Reviews (PROSPERO, number CRD42023489018). This study did not involve new human or animal experiments; therefore, approval from an institutional ethics committee was not required.
Literature search and eligibility criteria
This systematic meta-analysis adhered to the Meta-analysis of Observational Studies in Epidemiology (MOOSE) guidelines. 14 A comprehensive database search was performed by two researchers (YZ and YL) to identify relevant studies in PubMed, Embase, and Web of Science on 1 September 2024. The search strategy for PubMed was as follows: (“left ventricular dysfunction” OR “cardiac insufficiency” OR “cardiac dysfunction” OR “Right-Sided Heart Failure”OR “Left-Sided Heart Failure” OR “Heart Decompensation” OR “Congestive Heart Failure” OR“Cardiac Failure” OR “heart failure”) AND (“suppression of tumorigenicity 2” OR “ST2”). We also screened similar meta-analyses and abstracts of scientific conferences held during the past 3 years, such as those by the American Heart Association, American College of Cardiology, European Society of Cardiology, and Congress of the European Association of Percutaneous Cardiovascular Interventions. The detailed search strategy is provided in Supplementary Table 1.
Inclusion criteria for studies were as follows: (a) HF patients aged ≥18 years; (b) baseline sST2 levels available; (c) adjusted hazard ratios (HRs) and 95% confidence intervals (CIs) were provided for the risk of cardiovascular mortality/HF-related hospitalization (CVM/HFH), ACM/HF-related readmission (HFR), and ACM; and (d) prospective cohort studies or post-hoc analysis of randomized controlled trials. We excluded retrospective studies, overlapping studies, and those lacking comprehensive patient information. There were no language restrictions in the studies included in this review; all the included studies were published in English. Two authors independently assessed the inclusion of studies; any disagreements were resolved through discussion. To assess the consistency between the authors, 20% of the studies in the full-text screening stage were randomly sampled and re-screened. The kappa statistic was calculated to assess the consistency in the inclusions by the two authors.
Data extraction and quality assessment
YZ and YL extracted the data and addressed any contentious issues that emerged through discussion. Author, publication year, study location, study design, sample size, demographic information (mean age and sex), follow-up duration, adjusted variables, HRs, 95% CI, and threshold values for sST2 level were among the data that were extracted from the studies. For studies lacking HRs or 95% CIs, essential data were derived from survival curves. Study quality was appraised independently by two researchers using the Newcastle–Ottawa Scale (NOS); studies with scores of 7–9 were deemed high quality.
Statistical analyses
Statistical analyses were performed using Review Manager software, version 5.3 (Cochrane Collaboration in Oxford, UK). HR served as the main effect size in this study. If the sST2 level was presented as a continuous variable, we extracted the dose–response HR—the corresponding hazard ratio per 1-U (1 ng/mL) increase in the sST2 level; alternatively, when the sST2 levels were categorized, HRs were calculated by comparing the values above the cutoff with those below the cutoff value. Adjusted HRs were prioritized for extraction as they better reflect the true risk levels accounting for potential confounders. Unadjusted HRs were used only when adjusted HRs were unavailable. We used Cochrane’s Q test and the I2 index to assess heterogeneity among studies. An I2 value >50% indicated significant heterogeneity; in such cases, we used a random-effects model to combine the effect sizes. If the I2 value was <50%, a fixed-effects model was used. Additionally, we reported the 95% CI of I2 to provide more information about the stability of the heterogeneity estimate. In addition to the I2 index, we calculated tau2 as an absolute measure of heterogeneity. Tau2 reflects the variance of the true effects among studies, and the larger its value, the stronger the heterogeneity among studies. We have reported the value of tau2 in the Results section and combined it with the I2 index to describe the heterogeneity among studies more comprehensively.
To improve the accuracy of CI estimation for the effect size in the random-effects model, we introduced the Hartung–Knapp adjustment method. We used the metafor package in R software for the meta-analysis and Hartung–Knapp adjustment. To assess the impact of individual studies on the combined effect size, we conducted a sensitivity analysis. We used funnel plots and Egger’s test to evaluate publication bias. The funnel plot is a scatter plot with the effect size as the abscissa and the reciprocal of the standard error as the ordinate. If the graph presents a symmetrical funnel shape, it suggests a small publication bias, and if the graph is asymmetrical, there may be publication bias. To verify the reliability of the assessment results, the kappa statistic was used to test inter-assessor agreement, and statistical analyses were performed using Statistical Package for Social Sciences software (version 26.0; IBM Corp., Armonk, NY, USA). The criteria for evaluating agreement were based on the classification proposed by Landis and Koch: a kappa value of 0.81–1.00 indicates almost perfect agreement; 0.61–0.80, substantial agreement; 0.41–0.60, moderate agreement; 0.21–0.40, fair agreement; and ≤0.20, slight-to-no agreement.
Results
Description of studies
Overall, 5 databases yielded a total of 1003 publications; these were not manually identified from the reference citations of pertinent studies. First, 789 duplicates were eliminated from the 1003 studies. The titles and abstracts were then evaluated, and 143 studies that cited duplicate publications, were nonclinical studies, or fulfilled one of the other exclusion criteria were excluded. The remaining 33 studies were read in depth, and 16 were excluded because they included “patients without HF” or fulfilled any other exclusion criterion. Finally, the meta-analysis contained information from 17 trials involving 5373 participants (Figure 1).13,14,17–30 The kappa statistic was calculated as 0.82, indicating almost perfect agreement.

Flowchart of the systematic search and selection process.
The included studies were conducted in multiple countries in North America, Europe, East Asia, and other continents and published from 2011 to 2021. Table 1 displays the baseline information collected from each included study, including the author name, country, study design, sample size, age, proportion of male participants, patients, adjusted type, and follow-up duration.
Characteristics of the included studies.
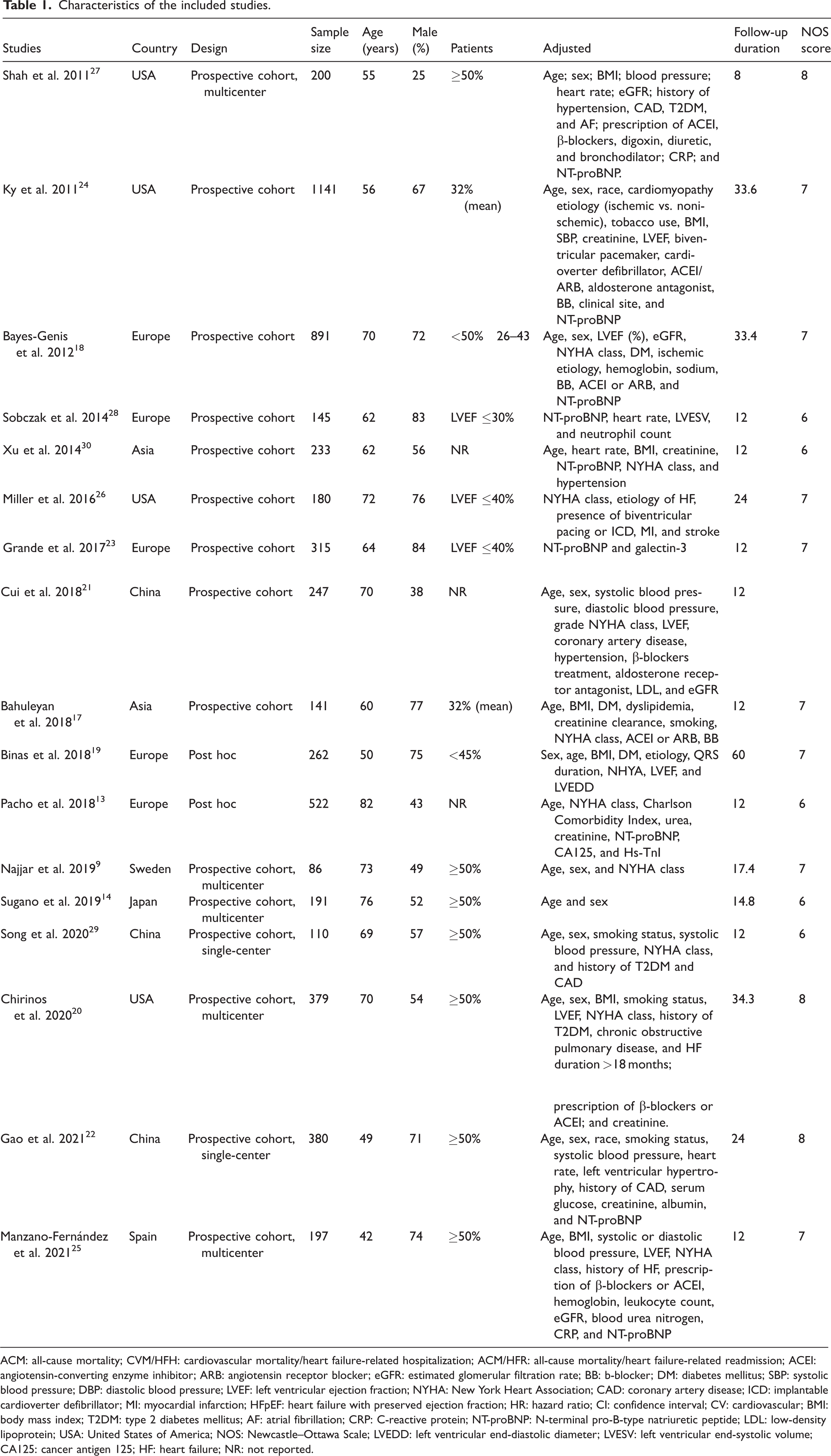
ACM: all-cause mortality; CVM/HFH: cardiovascular mortality/heart failure-related hospitalization; ACM/HFR: all-cause mortality/heart failure-related readmission; ACEI: angiotensin-converting enzyme inhibitor; ARB: angiotensin receptor blocker; eGFR: estimated glomerular filtration rate; BB: b-blocker; DM: diabetes mellitus; SBP: systolic blood pressure; DBP: diastolic blood pressure; LVEF: left ventricular ejection fraction; NYHA: New York Heart Association; CAD: coronary artery disease; ICD: implantable cardioverter defibrillator; MI: myocardial infarction; HFpEF: heart failure with preserved ejection fraction; HR: hazard ratio; CI: confidence interval; CV: cardiovascular; BMI: body mass index; T2DM: type 2 diabetes mellitus; AF: atrial fibrillation; CRP: C-reactive protein; NT-proBNP: N-terminal pro-B-type natriuretic peptide; LDL: low-density lipoprotein; USA: United States of America; NOS: Newcastle–Ottawa Scale; LVEDD: left ventricular end-diastolic diameter; LVESV: left ventricular end-systolic volume; CA125: cancer antigen 125; HF: heart failure; NR: not reported.
Quality assessment
Table 1 displays the NOS scores of the included studies. The overall characteristics and quality scores of the included studies were as follows: median score, 7 and range, 6–8; 14 studies were classified as high quality. Detailed scoring for each study is provided in Supplementary Table 2. The kappa value for data extraction was 0.86 and that for quality assessment was 0.83, indicating excellent objectivity and reliability of the assessment process with no systematic bias.
Publication bias
For the ACM, ACM/HFR, and CVM/HFH outcome measures, Egger’s test and funnel plots for the included studies showed no publication bias (ACM: p = 0.42; ACM/HFR: p = 0.106; CVM/HFH: p = 0.474) (Supplementary Figures 1 to 3).
Outcomes of the meta-analyses
ACM
A total of 8 studies, including 2538 participants, reported ACM,9,13,14,18,19,22,25,27 and the results of the meta-analysis suggested that elevated sST2 levels were associated with a higher risk of ACM (HR: 1.03, 95% CI: 1.02–1.04, p < 0.00001, I2 = 77% (95% CI: 62%–86%), tau2 = 0.003) (Figure 2). There was significant heterogeneity; however, a similar association was observed after applying the Hartung–Knapp adjustment (HR: 1.18, 95% CI: 1.02–1.37) (Supplementary Figure 4).

Forest plot of ACM. SE: standard error; df: degrees of freedom; CI: confidence interval; I2: I-squared; ACM: all-cause mortality.
Subgroup analyses of ACM
Subgroup analyses revealed that elevated sST2 levels were significantly associated with increased ACM in Western populations, but not in Asian populations. Stratification by sex, LVEF, and follow-up duration consistently demonstrated a significant association between higher sST2 levels and worse ACM outcomes across subgroups, although the degree of heterogeneity varied. These findings highlight potential ethnic and clinical variability in the prognostic value of sST2 (Supplementary Table 3).
ACM/HFR
In total, 8 studies, including 2651 patients, reported ACM/HFR,9,13,20,24,26,29,30 and according to the meta-analysis, higher sST2 levels were associated with increased ACM/HFR (HR: 1.46, 95% CI: 1.33–1.61, p = 0.00000, I2 = 61% (95% CI: 20%–82%), tau2 =0.047) (Figure 3). The association remained unaltered after Hartung–Knapp adjustment (HR: 1.60, 95% CI: 1.32–1.95) (Supplementary Figure 5).

Forest plot of all-cause mortality/heart failure-related readmission (ACM/HFR).
Subgroup analysis of ACM/HFR
Subgroup analyses demonstrated that elevated sST2 levels were significantly associated with an increased risk of ACM/HFR across diverse populations. This association remained robust regardless of ethnicity (Asian or Western), sex, LVEF, and follow-up duration, with all subgroups showing statistically significant HRs. These findings underscore the consistent prognostic value of sST2 across various clinical and demographic settings (Supplementary Table 4).
Cardiovascular mortality (CVM)/HFH
A total of 4 studies, including 848 patients, reported CVM/HFH17,21,23,28; higher sST2 levels were associated with increased CVM/HFH (HR: 1.50, 95% CI: 1.30–1.74, p = 0.00000, I2 = 76% (95% CI: 46%–91%), tau2 = 0.134) (Figure 4). The results obtained after Hartung–Knapp adjustment are presented in Supplementary Figure 6 (HR: 2.11, 95% CI: 1.33–3.34).

Forest plot of cardiovascular mortality/heart failure-related hospitalization (CVM/HFH).
Discussion
Clinicians tend to perform risk stratification in HF patients based on several clinical manifestations and laboratory biomarkers. 31 In terms of readmission rates and ACM in patients with chronic, recurrent HF, easily accessible and reproducible blood-level cardiac biomarkers offer the potential to improve the accuracy of predictive outcomes. Considering the degree of economic development in different countries and regions, although blood biomarkers do not allow for a comprehensive multidimensional assessment of the specific health condition of patients with CHF, their advantage in providing informative data for risk assessment in any situation is evident.4,32 From the perspective of evidence-based medicine, our study explored 17 studies from different countries and regions to preliminarily validate the relationship between sST2 level and prognosis of patients with CHF and provide evidence-based medical evidence for the use of sST2 as a clinical prognostic indicator.
Recently, there has been growing emphasis on the importance of identifying prognostic markers in HF.33,34 For assessing risk stratification in HF, the current widely recommended biomarker is brain natriuretic peptide (BNP), which is recommended in professional guidelines as being the most preferred biomarker for assessing the prognosis of acute and chronic patients. However, the specificity of natriuretic peptide is often altered by the presence of multiple confounding factors (e.g. multisystemic disease, aging, atrial fibrillation, and obesity), which further contributes to its reduced predictive confidence.35,36 ST2 belongs to the family of IL1RL1-like proteins that are produced by cardiomyocytes and fibroblasts. There are two isoforms of ST2: sST2 and ST2L. sST2 functions as a decoy receptor to disrupt the antifibrotic and antihypertrophic signaling pathways in cardiomyocytes when it binds to IL-33, which is released in response to cardiac damage and inflammation. Similar to natriuretic peptide, sST2 is sensitive to mechanical stretch; thus, it responds to load with dynamic levels in the short term, which quickly decrease following successful treatment. This dynamic behavior of sST2 makes it a valuable biomarker for monitoring acute cardiac stress and treatment efficacy. Elevated sST2 levels are often associated with increased fibrosis, inflammation, and worse clinical outcomes, highlighting its potential as a diagnostic and prognostic tool in HF. Modulation of sST2 levels could restore the balance of IL-33/ST2 signaling, opening up new therapeutic possibilities for preventing cardiac remodeling and improving long-term outcomes in HF patients.37–39 In addition to sST2, other biomarkers such as galectin-3 (Gal-3) have gained attention for their potential prognostic value in CHF.40,41 Gal-3 is a carbohydrate-binding protein involved in fibrosis, inflammation, and cardiac remodeling, which are key processes in the pathophysiology of HF. Several studies have demonstrated that elevated Gal-3 levels are associated with adverse outcomes in patients with CHF, including increased risk of hospitalization, mortality, and disease progression. 42 Unlike sST2, which reflects myocardial stress and injury, Gal-3 is more closely linked to the fibrotic response and inflammation within the heart. 43 Cell growth and differentiation, cell adhesion, apoptosis, and angiogenesis are only a few of the biological processes for which it has been demonstrated to extracellularly mediate signaling pathways. 4 Ventricular remodeling is mediated at the myocardial level by the inflammatory process of HF and macrophage activation, which encourages collagen deposition and myocardial fibroblast proliferation. 44 In contrast to BNP and sST2, Gal-3 has a stable structure. Therefore, stratification of patients through comprehensive assessment of multiple biomarkers at the hematological level in a minimally invasive manner is a future research direction. 44
Compared with previous studies,45–48 the strength of our study lies in its meta-analysis design that involved a systematic search and inclusion of published studies as well as the evaluation of the potential ability of sST2 as a predictive marker of prognosis in patients with CHF in terms of multiple clinical indicators, revealing that high sST2 levels are a valid predictor of poor prognosis. Meanwhile, the results of ACM and ACM/HFR were further analyzed in subgroups; ethnicity, sex, LVEF, and follow-up duration were used as the basis for creating the subgroups, which allowed multidimensional evaluation to strengthen the findings. In addition, our meta-analysis reaffirmed that in subgroup analyses of ACM by ethnicity, sST2 was not a valid predictor of ACM in Asian populations. An exploration of the reasons for this revealed that only two studies were included in this subgroup analysis. The smaller sample size appears to be the most important factor affecting the results, likely reflecting the limited use and detection of sST2 or population-specific factors inherent to Asian populations, which need to be further investigated.
This study strictly followed the PRISMA guidelines throughout all stages; however, certain limitations should be acknowledged. First, the included studies consisted solely of prospective cohort studies and post-hoc analyses; no randomized controlled trials (RCTs) were included; this may have limited the strength of the evidence. Second, the measurement methods for sST2 varied across studies, potentially introducing bias. To address this, we standardized the extraction of HRs and 95% CIs. For studies lacking these parameters, survival curves were used to obtain the necessary data. Third, there was considerable variability in the follow-up duration across the included studies, ranging from 8 months to approximately 34 months. This heterogeneity may have influenced the results. Despite the high quality of the included studies, no publication bias was detected, and subgroup analyses were performed to explore the heterogeneity of ACM. However, the specific sources of heterogeneity were not clearly identified.
Conclusion
Our study suggests that higher sST2 levels are associated with poorer clinical prognosis in patients with CHF. Higher sST2 levels may predict higher ACM, ACM/HFR, and CVM/HFH. Current evidence suggests that sST2 has independent prognostic value; however, further studies are needed to confirm these findings and fully justify its clinical utility.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251406969 - Supplemental material for The prognostic value of circulating soluble ST2 in patients with chronic heart failure
Supplemental material, sj-pdf-1-imr-10.1177_03000605251406969 for The prognostic value of circulating soluble ST2 in patients with chronic heart failure by Yue Zhang, Yunhong Liu, Yuanxia Yang, Lei Peng, Pengyu Zhong and Xiaoxin Jiang in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605251406969 - Supplemental material for The prognostic value of circulating soluble ST2 in patients with chronic heart failure
Supplemental material, sj-pdf-2-imr-10.1177_03000605251406969 for The prognostic value of circulating soluble ST2 in patients with chronic heart failure by Yue Zhang, Yunhong Liu, Yuanxia Yang, Lei Peng, Pengyu Zhong and Xiaoxin Jiang in Journal of International Medical Research
Footnotes
Acknowledgments
Not applicable.
Authors’ contributions
Yue Zhang and Yunhong Liu designed the study. Yuanxia Yang, Lei Peng, and Pengyu Zhong searched the literature, selected the papers, and extracted the data. Xiaoxin Jiang and Yue Zhang confirm the authenticity of all the raw data. Yue Zhang and Xiaoxin Jiang wrote the manuscript. All authors have read and approved the final version of the manuscript.
Consent for publication
Not applicable.
Clinical trial number
Not applicable.
Data availability
No datasets were generated or analyzed during the current study.
Declaration of conflicting interests
The authors confirm that there are no conflicts of interest to disclose.
Ethics approval and consent to participate
Since this research is a meta-analysis based on the published articles, the need for ethical approval and informed consent was waived.
Funding
This work was supported by the Shenzhen Medical Research Fund (SMRF): A2302048.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
