Abstract
Cardiac involvement in idiopathic inflammatory myopathies is typically subclinical; however, it is an important predictor of poor prognosis. Although data are limited, including two small cohorts and several case reports, evidence has demonstrated that cardiac involvement in anti-melanoma differentiation–associated gene 5 antibody–positive dermatomyositis is a significant independent predictor of mortality. Here, we summarized the characteristics of two patients with anti-melanoma differentiation–associated gene 5 antibody–positive dermatomyositis who developed fatal cardiac complications, one at early stage and the other during long-term follow-up of the disease. Despite aggressive treatment of the primary condition and intensive life support, both patients ultimately succumbed to their cardiac events. These cases highlight the importance of cardiac evaluation and monitoring in patients with anti-melanoma differentiation–associated gene 5 antibody–positive dermatomyositis.
Keywords
Introduction
Cardiac involvement is common in idiopathic inflammatory myopathies (IIMs), with a prevalence ranging from 4% to 26%, and is associated with heterogeneous manifestations. 1 Severe cardiac involvement is one of the most important factors of mortality in the anti-melanoma differentiation–associated gene 5 antibody–positive dermatomyositis (anti-MDA5+ DM) subgroup, as reported in a few studies.2–4 In the present study, we report two cases of patients with anti-MDA5+ DM who presented with fatal cardiac complications during the very early inflammatory phase and persistent disease stage, respectively. This report aimed to highlight the importance of comprehensive cardiac monitoring in patients with anti-MDA5+ DM.
Case presentation
Case 1
A female patient, approximately 70 years of age, was admitted to Sir Run Run Shaw hospital, Zhejiang University School of Medicine in February 2023. She presented with a 2-month history of polyarthralgia involving the shoulders, elbows, and metacarpophalangeal (MCP) joints and a 2-week history of finger rashes with ulceration. She denied experiencing chest tightness, cough, or fever. She had a 4-year history of well-controlled hypertension. History of diabetes, cardiovascular and cerebrovascular diseases (except hypertension), family diseases, alcohol consumption, and smoking were denied.
The patient’s vital signs were normal on admission. Physical examination revealed tenderness of her shoulders, elbows, and MCP joints. Erythematous eruptions with ulceration were observed on her MCP joints and elbows, consistent with Gottron’s sign. Velcro-like crackles were heard bilaterally over the lower lung zones. Muscle strength was normal in all limbs. Cardiac and abdominal physical examinations were unremarkable. The initial laboratory findings are summarized in Table 1. Strong positivity for serum anti-MDA5 antibody (semiquantitative titer, +++) was detected using a specific line immunoassay (Euroimmun, Germany). Interstitial lung changes and inflammation predominantly in the bilateral lower lobes were observed on chest high-resolution computed tomography (HRCT) (Figure 1(a)). A pulmonary function test (PFT) could not be completed due to the patient’s poor cooperation. Arterial blood gas (ABG) analysis on room air demonstrated values within normal physiological ranges. Magnetic resonance imaging (MRI) suggested inflammatory myopathy despite normal electromyography (EMG). The initial 12-lead electrocardiogram (ECG) demonstrated regular sinus rhythm at 97 beats per min with mild anterior ST-T changes, and echocardiography (Echo) revealed mild left ventricular diastolic dysfunction.
Clinical findings of the patients on admission.

RP-ILD: rapidly progressive interstitial lung disease; NT-proBNP: N-terminal prohormone of brain natriuretic peptide
Bold text denotes values outside the normal range.
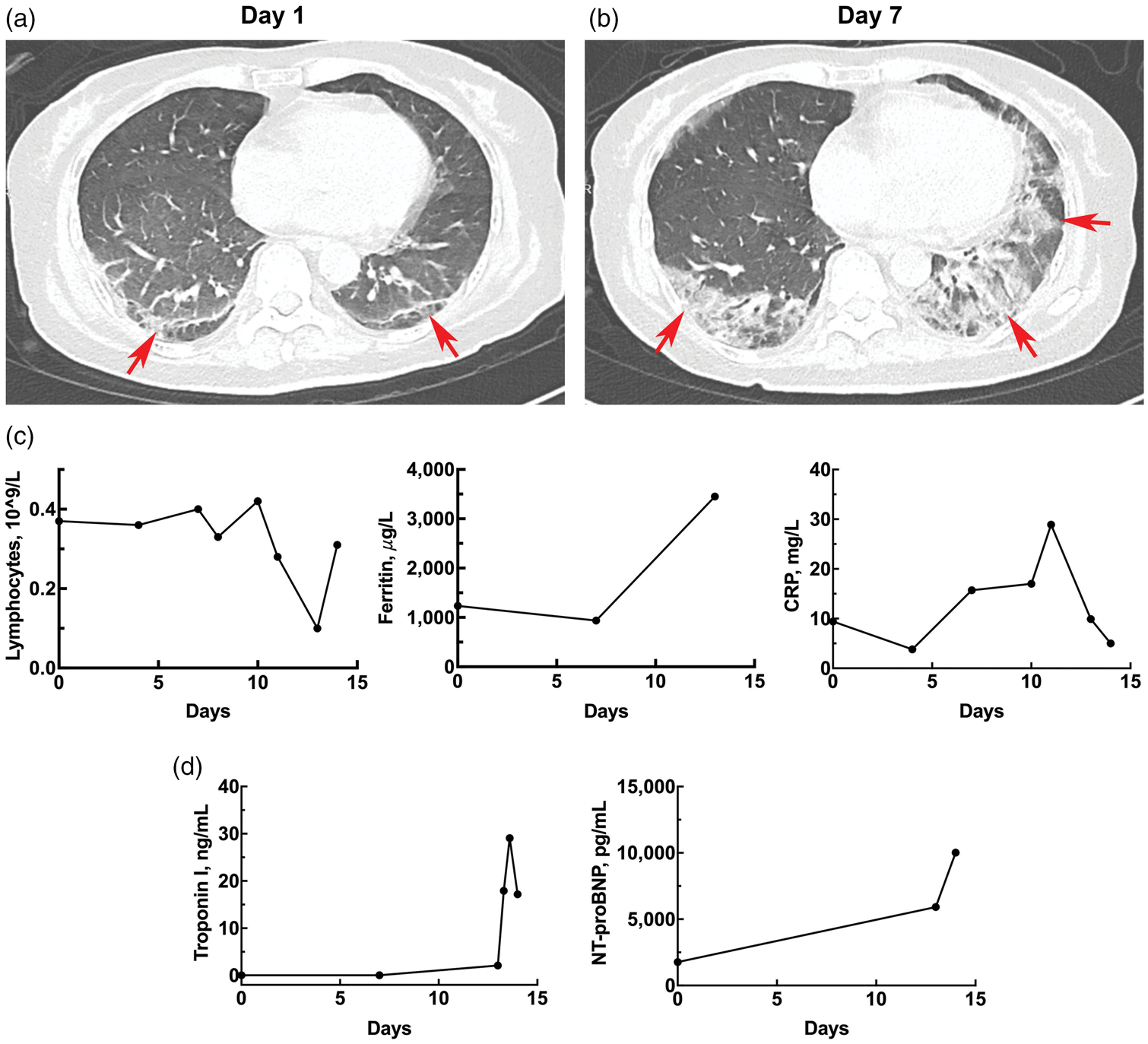
Longitudinal changes in pulmonary HRCT findings and laboratory markers for case 1. (a) Scattered subpleural-predominant inflammation and interstitial changes in both lower lungs on day 1 (arrows); (b) significantly progressed patchy opacities, linear densities, and interstitial abnormalities in both lower lungs on day 7 (arrows); (c) dynamic changes in peripheral blood lymphocyte count, ferritin level, and C-reactive protein level and (d) dynamic changes in the peripheral blood levels of troponin I and NT-proBNP.
The patient was diagnosed with anti-MDA5+ DM complicated with interstitial lung disease (ILD). Despite receiving high-dose methylprednisolone therapy (160 mg/day), the patient developed progressive respiratory failure requiring high-flow nasal cannula (HFNC) oxygen support 5 days after treatment initiation. Follow-up chest HRCT indicated rapidly progressive ILD (RP-ILD) (Figure 1(b)). We added cyclophosphamide (CTX; 400 mg/week), tacrolimus (2 mg/day), and immunoglobulin therapy (IVIG; 20 g/day for 5 days). However, the patient’s respiratory function continued to deteriorate (HFNC increased from 45 (fraction of inspired oxygen (FiO2), 50%) to 50 L/min (FiO2, 65%). Blood tests revealed persistent lymphopenia, hyperferritinemia, and moderate increase in the C-reactive protein level, suggesting a poor prognosis (Figure 1(c)). One week later, the patient developed dyspnea with oxygen saturation dropping to 85%, and her respiratory rate increased to 40 beats per min. The cardiac monitor showed transient ventricular fibrillation, and ECG revealed abnormal Q waves with ST-segment elevation in the high lateral, inferior, and extensive anterior walls. The patient was transferred to the intensive care unit following endotracheal intubation. Subsequent diagnostic workup demonstrated markedly increased levels of troponin I and N-terminal prohormone of brain natriuretic peptide (NT-proBNP) (Figure 1(d)). Left ventricular regional wall motion abnormality was observed on Echo. Acute myocardial infarction (AMI) was the primary consideration, and severe myocarditis was included in the differential diagnosis. Coronary angiography was strongly recommended for definitive diagnosis and intervention; however, the patient’s family declined the procedure. Moreover, the patient could not undergo cardiac MRI due to her constant need for ventilator support. Dual antiplatelet therapy and statin were administered as alternative therapy, and plasmapheresis was conducted daily for treating primary anti-MDA5+ DM. Ultimately, the patient was discharged against medical advice due to circulatory failure and subsequently died.
Case 2
A male patient, aged approximately 50 years, was admitted to the intensive care unit at Sir Run Run Shaw hospital, Zhejiang University School of Medicine in May 2022, with a 1-week history of cough with pink frothy sputum. His symptoms had worsened, accompanied by progressive chest tightness, dyspnea, orthopnea, and edema of the lower limbs and face; however, no fever was reported. He had been diagnosed with amyopathic dermatomyositis (ADM) 2 years ago based on fever, arthritis, Gottron’s sign with ulceration, and positive serum anti-MDA5 antibodies. Chest HRCT findings had been unremarkable at that time. At the time of ADM diagnosis, treatment was initiated with a combination of methylprednisolone (40 mg/day), CTX, and hydroxychloroquine. However, his erythematous eruptions with ulceration on fingers were poorly controlled. Prior to hospitalization, his recent treatment included prednisone (25 mg/day), CTX (200 mg every 2 weeks), and hydroxychloroquine (200 mg/day). He had no history of hypertension, diabetes, cardiovascular and cerebrovascular diseases, or other complications. History of family diseases was denied. He was a nondrinker and nonsmoker.
Physical examination on admission showed high blood pressure (164/96 mmHg), increased heart rate (130 beats per min), normal body temperature, and normal respiratory rate. Bilateral lung crackles were audible. Cardiac examination revealed tachycardia without murmurs. Erythema with ulceration was observed over his MCP and proximal interphalangeal joints (PIPs), with some ulcers healed and crusted. Other physical examination results were unremarkable. The laboratory results are listed in Table 1. ABG analysis indicated mild hypoxemia (partial pressure of arterial oxygen (PaO2), 75.8 mmHg). Chest HRCT revealed bilateral, multifocal pulmonary infiltrates with mild pleural effusions (Figure 2). NT-proBNP (55,000 pg/mL) level was markedly elevated. ECG revealed sinus tachycardia (112 beats per min) with left ventricular high voltage, and Echo demonstrated left ventricular hypertrophy with reduced left ventricular systolic function (Ejection Fraction (EF), 38.5%). He was diagnosed with acute left heart failure, and treatment was intensified with diuretics and antihypertensive agents. For the treatment of the underlying dermatomyositis (DM), we added prednisone dosage (50 mg once daily). Although cardiac MRI was essential for identifying the etiology of the patient’s heart failure, the examination could not be performed owing to hemodynamic instability. Despite intensive therapy, the patient died of circulatory failure 5 days after hospital admission.

Chest HRCT findings for case 2 at hospital admission. (a) Scattered multiple patchy opacities and nodules in both lungs (blue arrows) and (b and c) bilateral small pleural effusions (red arrows) visualized on both lung window (b) and mediastinal window (c). HRCT: high-resolution computed tomography.
The reporting of this study conforms to Case Report (CARE) guidelines. 5 The timeline of diagnosis, assessments, treatments, and outcomes of both cases are illustrated in Figure 3. Both patients provided written informed consent for treatment on admission. The legal guardians of both patients provided written consent for publication. We have de-identified all patient details.

Timeline of diagnosis, treatments, and outcomes in both patients. MDA5: melanoma differentiation–associated gene 5; DM: dermatomyositis; CTX: cyclophosphamide; HCQ: hydroxychloroquine; ILD: interstitial lung disease; HFNC: high-flow nasal cannula; IVIG: immunoglobulin therapy.
Discussion
Anti-MDA5+ DM is one of the most serious subgroups of DM, characterized by a high incidence of RP-ILD; severe typical rashes often accompanied by ulcers, and serologic positivity for anti-MDA5 antibody. Anti-MDA5+ DM is associated with the poorest prognosis among all DM subgroups, with a 1-year mortality rate of 50.3%. 6 Over the past decade, extensive studies have focused on the exploration of anti-MDA5+ DM–associated RP-ILD, as respiratory failure remains the leading cause of mortality in these patients. It is evident that cardiac events are another non-negligible cause of mortality, accounting for 12.5% of deaths in anti-MDA5+ DM patients. 3 Therefore, the identification of cardiac involvement in patients with anti-MDA5+ DM is crucial for clinicians.
The major cardiac manifestations in IIMs include heart failure, myocardial infarction, myocarditis, and arrhythmia, with the highest occurrence during the first 5 years after diagnosis.1,7,8 Data from a cohort of 1145 DM/polymyositis patients indicated that the incidence of coronary heart disease (CHD) in these patients was 2.21–3.73 times higher than that in the general population during an 8-year follow-up period. 9 However, studies on cardiac involvement in anti-MDA5+ DM patients remain limited. Prognostic data of 12 anti-MDA5+ DM patients with cardiac involvement demonstrated that 6 of the 8 deceased patients had concurrent heart failure. 3 Myocarditis, arrhythmia, atrioventricular block, and pericarditis in anti-MDA5+ DM patients have been reported in several case reports.10–12
In this study, we demonstrated two contrasting fatal trajectories (early acute cardiovascular event versus delayed cardiomyopathy), underscoring the unpredictable nature of cardiac complications. Both patients presented with prominent skin ulcers and subsequently experienced fatal cardiovascular events at 2 months and 2 years after DM symptom onset, respectively. These two contrasting cases not only highlight the necessity of cardiac monitoring during the acute inflammatory phase in anti-MDA5+ DM patients but also underscore the critical need for long-term cardiac surveillance in this patient population. Data from a large Chinese cohort has also indicated that the risk of cardiac-related death increases with prolonged disease duration from 3 years since disease onset in IIM patients. 13
We believe that the cardiovascular events in our patients were attributed to the anti-MDA5+ DM disease process. First, both patients had no obvious preexisting cardiovascular disease or cardiac risk factors prior to DM diagnosis. Second, both patients presented with Echo abnormalities, indicating DM-related cardiac involvement. Left ventricular diastolic dysfunction is the most commonly reported Echo manifestation in IIM patients during the early stage of disease.14,15 Significant deterioration of left ventricular diastolic dysfunction and newly appeared subclinical left ventricular systolic dysfunction were observed in IIM patients 2 years after diagnosis. 16 Another study, using global longitudinal strain to evaluate heart function, also reported that subclinical left ventricular dysfunction is common in IIM patients. 17
In our first patient, left ventricular diastolic dysfunction was detected using Echo at DM diagnosis. The final diagnosis of this patient (AMI or acute myocarditis) was not confirmed because coronary angiography could not be performed. However, the cardiac event occurred during high inflammatory phase of anti-MDA5+ DM, suggesting a correlation between the event and underlying inflammatory process.
For the second patient, treatment-related cardiovascular side effects must also be considered as he had been under treatment for 2 years. Glucocorticoids are reportedly associated with a time- and dose-dependent increased risk of cardiovascular events owing to their effect on blood pressure, glycemia, and lipid metabolism. 18 Additionally, the cardiotoxicity of CTX and hydroxychloroquine is well recognized. We suggest that the patient’s cardiac involvement is more likely attributable to recurrent pathologic process of the disease. First, the patient presented with significant left ventricular hypertrophy with diffused, reduced left ventricular systolic function, which indicated inflammatory cardiomyopathy rather than ischemic changes. Second, over the 2-year disease course, the patient experienced persistent cutaneous ulcerations, which reflected a recurrent inflammatory and fibrotic process. Based on early autopsy findings, inflammation and fibrosis are also primary pathological manifestations of cardiac involvement in patients with IIMs.19,20 Third, the patient did not exhibit other treatment-related adverse reactions, such as glucocorticoid-related hypertension, diabetes, or hyperlipidemia; CTX-related myelosuppression or renal toxicity; or hydroxychloroquine-related retinopathy. Therefore, we speculated that the recurrent inflammatory insults and subsequent maladaptive repair processes led to cardiac structural remodeling, culminating in left ventricular systolic dysfunction and eventual heart failure. Unfortunately, neither coronary angiography nor cardiac MRI were performed because of the patient’s hemodynamic instability, preventing further confirmation of the definite causes of heart failure. Nevertheless, this case highlights the crucial role of cardiac monitoring in patients with anti-MDA5+ DM who have a with long disease course.
The primary limitation of this study is the absence of coronary angiography or cardiac MRI results, which could have helped clarify the causes of cardiac events in the patients. We attribute this limitation primarily to the following reasons. In these two real-world patients, further diagnostic investigations were hindered by objective factors: the first patient experienced abrupt clinical deterioration coupled with family’s refusal of emergency coronary angiography, whereas the second patient presented with hemodynamic instability. In addition, insufficient awareness among clinicians regarding the need for cardiac screening and monitoring in IIM patients resulted in the absence of cardiac MRI screening in the first patient at the time of diagnosis and in the second patient throughout the 2-year disease course. Nevertheless, the presentation of these two patients and the discussion of their limitations will help raise awareness among clinicians and readers about the importance of routine cardiac screening and monitoring in patients with IIMs.
Conclusion
Our study highlighted that fatal cardiac complications can occur in anti-MDA5+ DM patients during the early inflammatory phase and throughout the long-term persistent stage of the disease. These patients should be subjected to comprehensive cardiac evaluation and rigorous cardiac surveillance. Future research should focus on the precise evaluation and management of cardiac involvement in anti-MDA5+ DM patients as well as on elucidating the underlying mechanisms.
Footnotes
Acknowledgment
Not applicable.
Author contributions
N.S., C.L., and Y.H. collected and analyzed the clinical data. N.S. and X.X. prepared the figures and tables. W.Z. reviewed the data of cardiac assessments. N.S. wrote the manuscript. W.Z. and Y.H. supervised the study.
Availability of data and materials
The datasets of our cases are available from corresponding author upon reasonable request. Patient identifiers have been removed to ensure confidentiality.
Declaration of conflicting interests
The authors declare no competing financial interests.
Ethics statement for the use of human data
Since this study involved clinical data from two patients without intervention, formal ethical approval was waived by the Sir Run Run Shaw Hospital Ethics Committee.
Funding
This work was supported by grants from the National Natural Science Foundation of China (82302020) and the Natural Science Foundation of Zhejiang province (LMS25H100001).
