Abstract
Objective
This study aimed to investigate the effect of Homer1a on caspase-6/tumor necrosis factor-alpha signaling pathway in an inflammatory pain model. We sought to determine whether Homer1a could alleviate nociceptive hypersensitivity by inhibiting the activation of the caspase-6/tumor necrosis factor-alpha signaling pathway.
Methods
An inflammatory pain model was established by unilateral plantar injection of carrageenan in rats. The hypothesis was tested using behavioral and immunological approaches.
Results
In rats with carrageenan-induced hind paw inflammation, pain hypersensitivity was exacerbated, accompanied by an upregulation of Homer1a and activation of the caspase-6/tumor necrosis factor-alpha signaling pathway. Intrathecal administration of the caspase-6 inhibitor Z-valine–glutamic acid–isoleucine–aspartic acid–fluoromethyl ketone significantly attenuated microglial activation, tumor necrosis factor-alpha release, and thermal hypersensitivity but had no effect on Homer1a expression. Lentiviral overexpression of Homer1a reduced pain hypersensitivity and concurrently inhibited the caspase-6/tumor necrosis factor-alpha signaling pathway.
Conclusions
The present study demonstrates that Homer1a activation suppresses the caspase-6/tumor necrosis factor-alpha signaling pathway and alleviates thermal hypersensitivity in a rat model of inflammatory pain. These findings suggest that the Homer1a/caspase-6 signaling axis may represent a promising therapeutic target for inflammatory pain management.
Background
The Homer protein family is a major constituent of the postsynaptic density (PSD). All members of this family contain an Ena/VASP Homology 1 (EVH1) domain at the N-terminal region. With the exception of Homer1a, all other Homer proteins also feature coiled-coil (CC) structures and leucine zipper motifs at the C-terminal region.1,2 The EVH1 domain binds to proline-rich motifs in various proteins, including metabotropic glutamate receptor (mGluR), transient receptor potential cation channels (TRPC), inositol 1,4,5 trisphosphate receptor (IP3R), and Shank proteins. 3 Homer isoforms containing the CC domain can form homo- or heteromeric complexes, thereby linking mGluRs to other PSD proteins. Homer1a is a short, activity-dependent splice variant that retains the conserved EVH1 domain, enabling it to bind to the same target proteins as CC-Homers (e.g. Homer1b/c). However, due to the lack of the C-terminal multimerization domain, Homer1a cannot form multimeric complexes. Instead, it acts competitively by binding to CC-Homer target proteins and disrupting complex assembly. 4 As a result, Homer1a functions as a dominant-negative regulator that modulates synaptic signaling complexes. However, it remains uncertain whether additional cytoplasmic factors interact with Homer1a to influence nociception.
Caspases (CASPs) are a family of intracellular cysteine proteases activated by proteolytic processing and dimerization. Initiator CASPs (CASP2, CASP8, CASP9, and CASP10) are typically activated by proximity-induced dimerization, whereas executioner CASPs (CASP3, CASP6, and CASP7) require proteolytic removal of the prodomain and cleavage into large and small subunits to form an active heterodimer. 5 Substantial evidence implicates CASP6 in neurodegenerative diseases such as Alzheimer’s disease (AD) and Huntington’s disease (HD). 6 Beyond its role in neurodegeneration, CASP6 performs nonapoptotic physiological functions in the central nervous system. For instance, CASP6 released from axonal terminals has been shown to regulate microglial tumor necrosis factor-alpha (TNF-α) secretion, synaptic plasticity, and inflammatory pain. 7
In HD, activation of extrasynaptic N-methyl-D-aspartate receptors (NMDARs) elevates intracellular calcium levels, leading to CASP and calpain activation. 8 In AD, suppression of the BK channel induces neuronal hyperexcitability, which triggers excessive glutamate release and mitochondrial calcium (Ca2+) overload, resulting in CASP activation and excitotoxic neuronal damage. 9 Conversely, the rapid induction of the immediate-early gene Homer1a (IEG-Homer1a) disrupts interactions between CC-Homers and NMDARs/Shank or mGluRs, thereby reducing calcium mobilization and extracellular signal-regulated kinase 1/2 (ERK1/2) phosphorylation. 10 This inhibition of calcium influx may consequently affect CASP activation. Based on these theoretical foundations, we hypothesize that Homer1a regulates inflammatory pain via the CASP6/TNF-α signaling pathway.
Materials and methods
Animals
The experiments were performed on a total of 106 male Sprague–Dawley rats (200–250 g, 8 weeks old) obtained from the Experimental Animal Center of Xuzhou Medical University (Xuzhou, Jiangsu Province, China). The rats were housed in a temperature-controlled room (24°C ±1°C) with humidity maintained at 55% ±10% and a 12-h light/dark cycle (8:00 a.m. to 8:00 p.m.), with ad libitum access to food and water. All animal experiments were conducted in accordance with a protocol approved by the Laboratory Animal Ethics Committee of Xuzhou Medical University (202403S139) and the Guide for Care and Use of Laboratory Animals. 11 Efforts were made to minimize animal suffering and reduce the number of animals used. The reporting of this study conforms to the Animal Research: Reporting of In Vivo Experiments (ARRIVE) 2.0 guidelines. 12 All rats were randomized to different experimental groups, and blinding was implemented whenever possible. Investigators who performed and analyzed the tests were blinded to the experimental conditions of the animals.
Carrageenan-induced inflammation
Carrageenan (degraded λ-carrageenan, Sigma-Aldrich, USA) was dissolved in saline to form a 2% solution, which was stored at room temperature for 24 h. Under light isoflurane anesthesia, 100 µL of this solution was injected subcutaneously into the center of the ventral surface of the left hind paw using a 30-g needle. The carrageenan injection was unilateral. 13
Lentivirus transfection
Lentivirus expressing Homer1a-enhanced green fluorescent protein (EGFP) (LV/H1a, 3 × 108 TU/mL) was constructed by GenePharma. Rats were anesthetized with 4% isoflurane, and 30 µL of LV/H1a viral stock containing 9 × 106 virions or 4.5 µL LV/control viral stock plus 25.5 µL of phosphate-buffered saline (PBS) was infused intrathecally into the spinal canal of lumbar segments L4–L6. After a 2-min infusion period, the needle was left in place for an additional 2 min and then withdrawn slowly. Rats were allowed to recover for 2 weeks before behavioral testing. Following behavioral testing, animals were deeply anesthesized with 400 mg/kg 10% chloral hydrate and transcardially perfused with saline followed by paraformaldehyde. Forty-micrometer sections of the lumbar enlargement were examined to assess transduction efficiency and vector spread based on EGFP expression.
Measurement of pain threshold
The paw withdrawal thermal latency (PWTL) was measured using a thermal apparatus (BME410A, Institute of Biological Medicine, Academy of Medical Science, China). Rats were placed in clear plastic cages on an elevated glass plate and allowed to acclimate for 30 min before testing. A radiant heat source with constant intensity was placed underneath the glass and aimed at the mid-plantar area. A digital timer automatically recorded the duration from the onset of heat stimulation to paw withdrawal. The baseline PWTL of rats was maintained at approximately 15 s by adjusting the intensity of the light. Rats with a baseline PWL of less than 6 s were excluded from the study to avoid false-positive results (approximately 10% of the animals were excluded during screening). A cutoff time of 25 s was set to prevent tissue damage. Each animal was tested three times at 5-min intervals.
Western blotting
All rats were deeply anesthetized with 5% isoflurane until loss of righting reflex, followed by rapid cervical dislocation. The lumbar spinal cords were removed, frozen in liquid nitrogen, and stored at −80°C. Tissue samples were homogenized in radioimmunoprecipitation assay (RIPA) buffer containing protease inhibitor and phosphatase inhibitor cocktails. The lysates were centrifuged at 12,000 r/min for 15 min at 4°C, and protein concentration was determined using the bicinchoninic acid (BCA) Protein Assay Kit (Beyotime Institute of Biotechnology, Shanghai, China). Protein samples (40 µg total protein per lane) were separated using 12% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS–PAGE) and transferred onto polyvinylidene difluoride membranes (Millipore, USA). Membranes were blocked with 5% nonfat dry milk for 2h and then incubated overnight with primary antibodies: mouse anti-Homer1a (1:200; Santa Cruz Biotechnology), rabbit anti-aCASP6 (1:2000; Novus Biologicals), rabbit anti-total CASP6 (1:1000; Cell Signaling Technology), goat anti-Iba1 (1:1000; Abcam), and mouse anti-β-actin (1:1000; Santa Cruz Biotechnology). After washing, the blots were incubated for 2 h at room temperature with horseradish peroxidase (HRP)–conjugated secondary antibody (1:5000; ZSGB-Bio) and developed using enhanced chemiluminescence (ECL) solution (Beyotime). Specific bands were identified based on apparent molecular sizes, and band intensities were analyzed using ImageJ software.
Immunohistofluorescence
Rats were anesthetized with 5% isoflurane and subjected to sternotomy, followed by intracardiac perfusion with 200 mL of saline and 200 mL of 4% ice-cold paraformaldehyde (PFA; 4% in 0.1 M phosphate buffer). The lumbar spinal cord tissues (L4–L6) were dissected, post-fixed in 4% PFA overnight, and subsequently allowed to equilibrate in 30% sucrose in phosphate buffer overnight at 4°C. Transverse sections (40 μm) were cut on a cryostat and stored in phosphate buffer. After washing in PBS, the sections were incubated in PBS containing 5% normal donkey serum and 0.3% Triton X-100 (PBST) at room temperature for 2 h and then incubated with primary antibody, goat anti-Iba1 (1:400; Abcam), in PBST overnight at 4°C. To remove excess antibody, slices were washed three times at 5-min intervals with PBS and incubated with Alexa Fluor 488–conjugated secondary antibody (1:200; Life Technologies) for 2 h at room temperature. Images were captured using a high-resolution digital confocal microscope (Olympus) connected to a computer.
ELISA
Rat ELISA kits for TNF-α were purchased from R&D Systems. Protein samples were prepared as described for western blotting. For each assay, a 100-μL protein sample was used, and a standard curve was included in each experiment.
Statistical analysis
The size of the experimental groups was determined based on our previous experience and relevant literature. 14 In this study, the experimental groups were compared against a well-defined control group. The experimental unit for statistical analysis was a single animal. Data were presented as mean ± SEM from three separate experiments. Statistical analysis was performed using Prism 5.0 (GraphPad) software. One-way analysis of variance (ANOVA) or Student’s t-test was used to analyze data from different groups. A Mann–Whitney U test was used when the variances of the two populations were not equal. Two-way ANOVA with one repeated factor (time) was used for PWTL data analysis. A p-value of <0.05 was considered statistically significant.
Results
Upregulation of Homer1a, CASP6, TNF-α, and microglial activation in the carrageenan-induced pain model
Intraplantar injection of 2% λ-carrageenan induced thermal hypersensitivity, peaking at 3 h post-injection (Figure 1(a)). Western blot analysis showed elevated levels of Homer1a and active CASP6 (aCASP6) in the spinal cord dorsal horn (SCDH) during pain hypersensitivity, with no change in pro-CASP6 (pCASP6) expression (Figure 1(b) and (c)). Enzyme-linked immunosorbent assay (ELISA) indicated a significant increase in TNF-α levels between 6 and 8 h after injection (Figure 1(d)). Microglial morphology shifted from ramified to amoeboid, accompanied by a marked increase in ionized calcium-binding adaptor molecule 1 (Iba1)–positive cells at 6–24 h (Figure 2(a) and (b)).

Upregulation of Homer1a, CASP6, and TNF-α in the carrageenan-induced pain model.
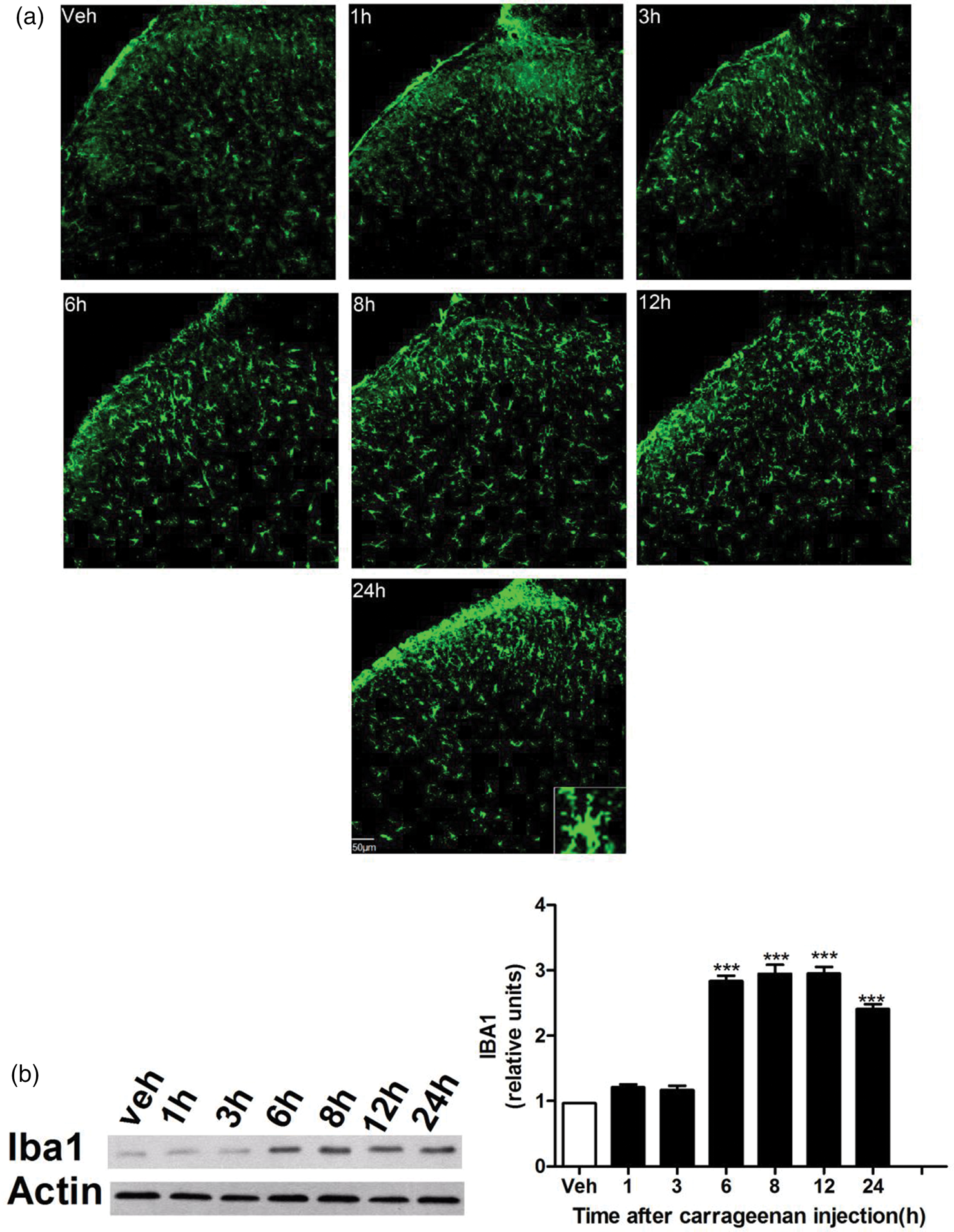
Morphologic and expression changes of microglia in the carrageenan-induced pain model.
CASP6 inhibition attenuates pain and inflammatory responses
To investigate the role of active CASP6 in carrageenan-induced inflammation, the CASP6 inhibitor Z-valine–glutamic acid–isoleucine–aspartic acid–fluoromethyl ketone (ZVEID-FMK, 10 µg dissolved in 4% dimethyl sulfoxide (DMSO); ApexBio) was administered intrathecally 2 h prior to carrageenan injection. 7 At the dosage used, no obvious toxicity was observed; animals exhibited normal behavior and unimpaired locomotion. Intrathecal ZVEID administration significantly reduced thermal hyperalgesia from 3 to 12 h after carrageenan challenge (Figure 3(a)). Based on previous findings indicating that CASP6 activation begins within 1 h and peaks at 8 h post-carrageenan injection, the 8-h time point was selected for immunoblotting. ZVEID treatment suppressed the levels of aCASP6, Iba1, and TNF-α (Figure 3(b) and (c)) but did not affect Homer1a expression (Figure 3(b)).
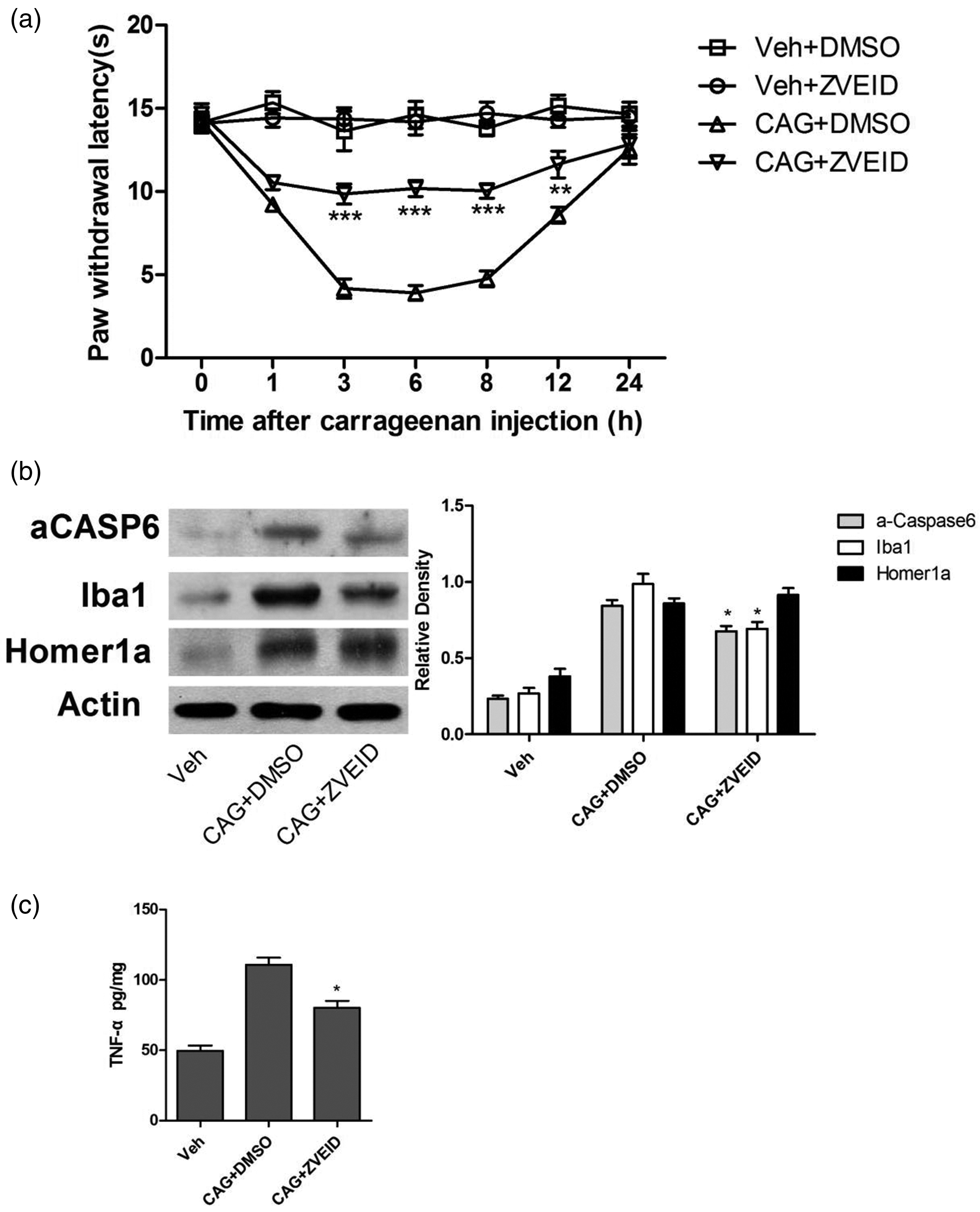
Caspase6 inhibition by ZVEID attenuates pain and inflammatory responses. (a) Compared with PWL in the control group treated with DMSO, PWL to thermal stimuli significantly increased from 3 h to 12 h following carrageenan (**p < 0.01; ***p < 0.001, CAG+DMSO vs. CAG + ZVEID, n = 6; two-way ANOVA) and (b–c) CASP6 signaling was inhibited by ZVEID; however, Homer1a activity induced by carrageenan was not affected. Data from at least three independent experiments are summarized in the graphs on the right. (*p < 0.05, CAG + DMSO vs. CAG + ZVEID, n = 3; Student’s t-test). ZVEID: Z-valine–glutamic acid–isoleucine–aspartic acid; PWL: paw withdrawal latency; DMSO: dimethyl sulfoxide; CAG: carrageenan; CASP6: caspase-6.
Homer1a overexpression alleviates pain and suppresses CASP6 signaling
To examine the role of Homer1a, an overexpression adenovirus (lentivirus Homer1a (LV/H1a)) was administered in a rat model of inflammatory pain. Homer1a levels were significantly elevated following LV/H1a treatment, but not in the LV/control group (Figure 4(b)). LV/H1a administration effectively attenuated inflammation-induced thermal hyperalgesia (Figure 4(a)). Similarly, the 8-h time point was chosen to evaluate whether LV/H1a could inhibit CASP6 signaling activation. In carrageenan-treated rats, LV/H1a significantly reduced the expression of aCASP6, Iba1 (Figure 4(b)), and TNF-α (Figure 4(c)), whereas no such effects were observed in the vehicle control group. These results indicate that intrathecal LV/H1a had no significant effect in normal rats but markedly alleviated inflammatory hyperalgesia and decreased aCASP6, Iba1, and TNF-α expression under inflammatory conditions.

Homer1a overexpression alleviates pain and suppresses CASP6 signaling. (a) Intrathecal injection of LV/H1a significantly reversed inflammation-induced thermal hyperalgesia (*p < 0.05; ***p < 0.001, CAG + LV/control vs. CAG+LV/H1a, n = 6; two-way ANOVA). (b–c) Homer1a, aCASP6, and Iba1 were assessed using western blotting, while TNF-α was assessed using ELISA. The expression of aCASP6, Iba1, and TNF-α was lower in CAG rats treated with LV/H1a than in those treated with LV/control. Data from at least three independent experiments are summarized in graphs on the right. (##p < 0.01, vehicle+LV/control vs. vehicle+LV/H1a; *p < 0.05, **p < 0.01, CAG+LV/control vs. CAG+LV/H1a, n = 3; Student’s t-test).
Discussion
The present study reveals that Homer1a prevents the development of pain hypersensitivity in rats with inflammatory pain and disrupts pain-related signaling pathway in the SCDH. The major findings are as follows: 1. A rapid increase in Homer1a and CASP6 signaling expression in SCDH neurons is associated with the development and maintenance of pain-related behaviors. 2. The CASP6 inhibitor ZVEID significantly attenuates thermal hyperalgesia and carrageenan-induced activation of CASP6 signaling but does not affect Homer1a expression. 3. Lentivirus-mediated overexpression of Homer1a following intrathecal injection reduces pain hypersensitivity and inhibits CASP6 signaling activation. These findings suggest a previously unrecognized mechanism by which Homer1a regulates inflammatory pain in the SCDH.
Emerging evidence indicates that Homer1a, a scaffold protein located at the PSD, regulates synaptic structure and postsynaptic signal transduction. By competing with Homer1b/c for binding sites, Homer1a disrupts multimeric complexes such as mGluR1/5-Homer1b/c-Shank and mGluR1/5-Homer1b/c-IP3R, shifting mGluR1/5 activation from a ligand-dependent to a ligand-independent mode. The resulting mGluR1/5-Homer1a complexes can inhibit NMDAR activity via the β subunit of mGluR1/5, promoting their subsequent endocytosis. A decrease in the levels of protein kinase reduces phosphorylation of AMPA receptor subunits, leading to their endocytosis due to the instability of nonphosphorylated forms. 15 Although many studies have focused on the role of Homer1a in central nervous system disorders such as AD, drug addiction, HD, and traumatic brain injury, its function in acute inflammatory pain remains less explored. Using a carrageenan-induced acute inflammatory pain model, we observed a rapid upregulation of Homer1a, which appears to participate in the regulation of neuronal hyperalgesia and pain-related behaviors. Furthermore, we investigated the correlation between altered Homer1a expression, pain behavior, and central sensitization. Our data demonstrate that overexpression of Homer1a alleviates inflammatory pain behaviors, likely by suppressing excitatory glutamatergic transmission and intracellular signaling pathways in dorsal horn neurons activated by peripheral inflammation, thereby inhibiting the development and maintenance of central sensitization.
CASP6, a member of the intracellular cysteine protease family, is widely recognized for its roles in apoptosis and neurodegeneration. Recent studies suggest that CASP6 also modulates synaptic transmission and pain. In a formalin-induced inflammatory pain model, Berta et al. detected activated CASP6 in the cerebrospinal fluid after plantar injection, indicating extracellular secretion of the CASP. 7 Our results show that intrathecal administration of the CASP6 inhibitor ZVEID-FMK significantly alleviates pain symptoms. Given its large molecular weight (∼700 kDa), ZVEID-FMK is unlikely to cross the blood–brain barrier and act intracellularly, supporting the hypothesis that it targets extracellular CASP6. Secreted CASP6 may activate microglia via p38 phosphorylation, triggering TNF-α production. TNF-α then binds to presynaptic tumor necrosis factor receptors (TNFR), enhancing glutamate release and excitatory synaptic transmission, ultimately leading to central sensitization. 16
In our experiments, ZVEID-FMK inhibited the CASP6/TNF-α signaling pathway without affecting inflammatory stress-induced Homer1a activation. Subsequently, adenovirus transfection was employed to overexpress Homer1a, which was found to suppress the activation of CASP6/TNF-α. Therefore, we propose that inflammatory stimuli trigger CASP6 release from central terminals of primary sensory neurons, inducing microglial activation reflected by p38 phosphorylation and TNF-α production. TNF-α enhances glutamate release, facilitating excitatory synaptic transmission. Increased formation of Homer1a-mGluR complexes inhibits NMDAR activation and AMPA receptor phosphorylation, thereby attenuating nociceptive signal amplification.
This study has several limitations. First, based on current research, it remains unclear whether a direct relationship exists between Homer1a and CASP6 or whether aCASP6 cleaves Homer proteins. This will require additional future studies for validation. Second, sex differences are known to influence pain research and microglial biology. 17 Our use of only male rats in this initial study was intended to control variables and maintain consistency with prior literature in this field; investigating sex-specific effects represents an essential future direction. Third, growing evidence suggests that spinal p38 phosphorylation may be a more reliable indicator of microglial activation and pain-related modulation than traditional markers such as Iba1. 18 Future studies should aim to identify and validate more accurate markers of microglial activation dynamics. Finally, this study is limited to investigating the role of the Homer1a/CASP6 axis in inflammatory pain. Therefore, the generalizability of this mechanism to other clinically relevant pain states, such as neuropathic pain (NP) and bone cancer pain (BCP), remains unexamined. Accumulating evidence has revealed that epigenetic mechanisms, including histone modifications, DNA methylation, and noncoding RNAs, play a critical role in the maintenance and progression of BCP by regulating pro- and antinociceptive gene expression.19,20 This insight raises a compelling possibility: targeting epigenetic pathways to modulate Homer1a expression may represent a promising therapeutic strategy. Such an approach could pave the way for novel epigenetics-based treatments aimed at controlling the onset and progression of diverse pain conditions.
Conclusion
This study demonstrates that Homer1a activation suppresses CASP6 signaling, significantly reduces microglial activation and TNF-α release, and alleviates thermal hypersensitivity in a rat model of inflammatory pain. These findings suggest that the Homer1a/CASP6 signaling axis may represent a promising therapeutic target for the treatment of inflammatory pain in patients.
Footnotes
Acknowledgments
We deeply appreciate the guidance of Prof Ming Yan during the experimental design and manuscript preparation. We also thank the anonymous reviewers for their constructive comments. Finally, we acknowledge the unwavering support from our families and colleagues.
Authors’ contributions
KJ designed the study and analyzed the data. WQZ, XXS, and MYY performed the experiments. KJ prepared the manuscript. All authors read and approved the final manuscript.
Data availability statement
All data supporting the findings of this study are included within the article.
Declaration of conflicting interests
The authors declare that they have no competing interests.
Funding
This work was supported by the Suzhou Science-Education Health Enhancement Project (Grant QNXM2024025).The funders had no role in the study design, data collection, data analysis, interpretation, or writing of this manuscript.
