Abstract
Paradoxical embolism refers to thromboemboli originating from the venous system or right heart that traverse an intracardiac or pulmonary shunt into the systemic circulation, causing end-organ embolization. It is hypothesized to occur when the right atrial pressure exceeds the left atrial pressure. To date, patent foramen ovale is the most common intracardiac shunt associated with paradoxical embolism. During acute pulmonary embolism, acutely elevated right atrial pressure can reopen a functionally closed patent foramen ovale, facilitating paradoxical embolism into the systemic circulation. The cerebrum is the most frequently affected organ, resulting in neurological deficits, followed by peripheral arterial occlusion and mesenteric embolism; myocardial infarction is rarely reported. Herein, we describe the diagnosis and management of a patient in her early 60s who presented with acute cerebral embolism caused by paradoxical embolism complicating acute pulmonary embolism.
Keywords
Introduction
Paradoxical embolism (PDE) is a well-recognized clinical entity characterized by venous thrombi passing through an intracardiac defect or pulmonary shunt into the arterial circulation, leading to end-organ ischemia, most commonly stroke, but also involving other systemic arteries. 1 This process relies on right-to-left interatrial shunting (RLS), which occurs when increased right atrial pressure (e.g. during diastole, early systole, or maneuvers such as coughing, sneezing, or Valsalva) drives blood flow across congenital or acquired cardiac communications. 2 Among these, patent foramen ovale (PFO) is the most prevalent intracardiac shunt linked to PDE, accounting for a substantial proportion of cryptogenic strokes, particularly in younger adults. 3 In such cases, venous thrombi—often originating from deep vein thrombosis (DVT) or pulmonary embolism (PE)—serve as the primary embolic source, traversing the PFO to cause arterial occlusion.4,5
Despite its clinical significance, PDE remains underdiagnosed due to its nonspecific manifestations that overlap with those of other thromboembolic disorders. The coexistence of acute cerebral infarction and acute PE—two life-threatening conditions often linked to PDE—further complicates diagnosis because the causal relationship between venous and arterial events may be overlooked.6,7 Controversies persist regarding optimal management; although PFO closure is recommended in selected PDE patients to prevent recurrent embolism, 8 there is no consensus on patient selection criteria, intervention timing, and the role of long-term anticoagulation alongside surgical or percutaneous closure.8,9 Additionally, the true incidence of PDE in patients with concurrent PE and cerebral infarction remains unclear, limiting evidence-based guidelines for this high-risk subgroup.
In this report, we describe the case of a middle-aged patient presenting with acute cerebral infarction secondary to PDE, concurrent with acute PE. By detailing the diagnostic workup, including confirmation of PFO and documentation of venous thromboembolism, and multidisciplinary management, this case highlights the challenges of identifying PDE in complex clinical scenarios. It also contributes to the literature by illustrating the relevance of PFO closure in preventing recurrent events in patients with combined arterial and venous thromboembolic manifestations, informing clinical decision-making in similar cases.
Case presentation
A woman in her early 60s was admitted to the emergency department with a 40-min history of dyspnea, accompanied with severe cough, palpitations, nausea, and vomiting. She had a 5-year history of hypertension and hyperlipidemia. On arrival, her vital signs were as follows: blood pressure, 113/64 mmHg; heart rate, 132 beats/min; and oxygen saturation (SaO2), 89% on low-flow oxygen therapy.
The patient exhibited acute hypoxic respiratory failure. Admission electrocardiogram showed an S wave in lead I and a Q wave in lead III (S1Q3 pattern), consistent with acute cor pulmonale (Figure 1). Laboratory tests revealed hypoxemia and elevated levels of N-terminal pro–B-type natriuretic peptide (NT-proBNP), serum cardiac markers, and D-dimer (Table 1). Given the high clinical suspicion for PE, contrast-enhanced chest computed tomography (CT) was performed, which confirmed bilateral submassive PE with acute right ventricular dilatation (Figure 2). The patient was prepared for percutaneous pulmonary artery thrombectomy and inferior vena cava filter (IVCF) placement. During preparation, she developed sudden left lower limb weakness without numbness or slurred speech. Urgent brain magnetic resonance imaging revealed a new acute infarction in the right frontal lobe. She underwent intravenous thrombolysis with 50 mg recombinant tissue plasminogen activator (rt-PA) in the emergency department, following which left lower limb muscle strength recovered and chest tightness subsided.

ECG at admission. Admission ECG revealed S deflections (full arrows) in lead I and Q waves (empty arrows), which is consistent with the S1Q3 pattern of acute cor pulmonale. ECG: electrocardiogram.
Results of laboratory examinations.
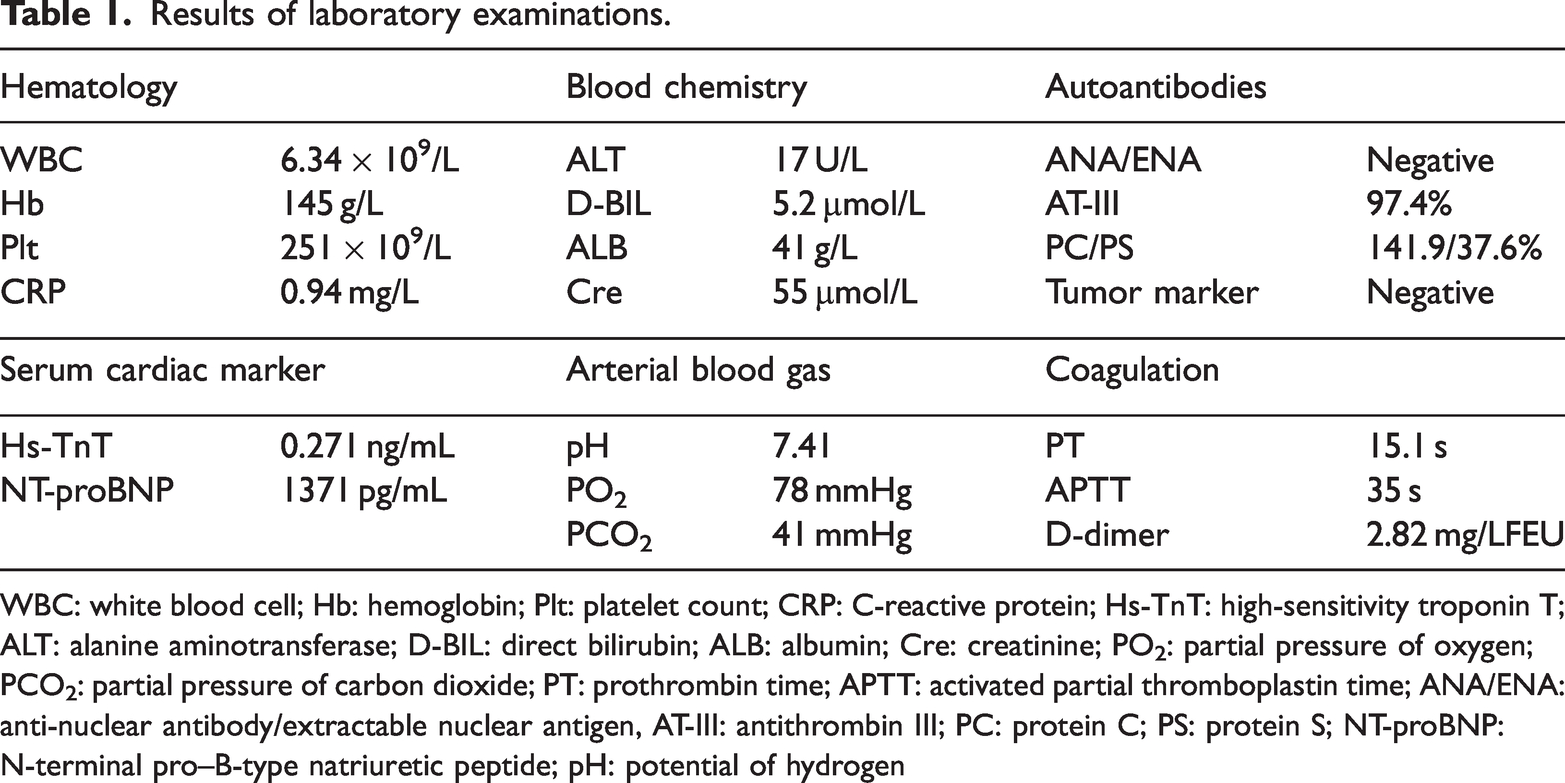
WBC: white blood cell; Hb: hemoglobin; Plt: platelet count; CRP: C-reactive protein; Hs-TnT: high-sensitivity troponin T; ALT: alanine aminotransferase; D-BIL: direct bilirubin; ALB: albumin; Cre: creatinine; PO2: partial pressure of oxygen; PCO2: partial pressure of carbon dioxide; PT: prothrombin time; APTT: activated partial thromboplastin time; ANA/ENA: anti-nuclear antibody/extractable nuclear antigen, AT-III: antithrombin III; PC: protein C; PS: protein S; NT-proBNP: N-terminal pro–B-type natriuretic peptide; pH: potential of hydrogen
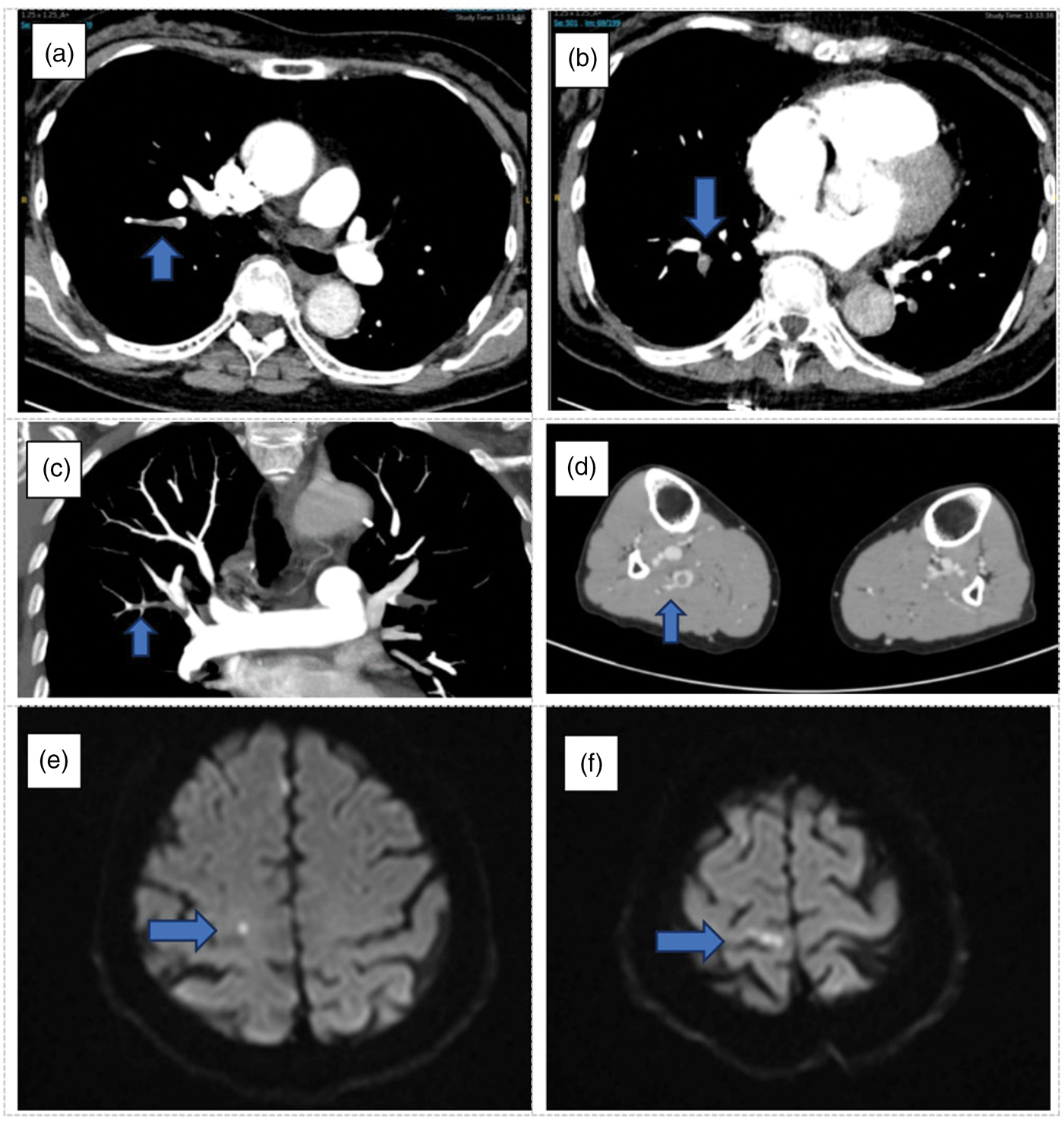
Admission pulmonary CTA and magnetic resonance imaging. (a and b) Pulmonary CTA at admission revealed multiple emboli (blue arrows) in the pulmonary arteries and branches. (c and d) Lower-extremity CT revealed right lower limb intermuscular vein thrombosis. (e and f) Cranial magnetic resonance imaging of the patient revealed acute multiple cerebral infarctions in the right frontal lobe. CTA: computed tomography angiography; CT: computed tomography.
Her hemodynamics improved after thrombolysis, as follows: blood pressure, 136/93 mmHg; heart rate, 80 beats/min; and SaO2, 95%. Due to decreased inferior vena cava blood flow, a Gunther tulip IVCF (Bard Vena Cava Filter; USA; Product Reference Number: DL950F) was placed. Saline contrast transthoracic echocardiography (via the left cubital vein) showed rapid opacification of the right heart chambers with simultaneous contrast appearance in the left heart, confirming RLS and a PFO (Figure 3).

Saline contrast echocardiogram and postoperative pulmonary CTA. (a and b) Bubble study revealed that >10 bubbles crossed the LA in 4 cardiac cycles (upper: bubbles in the LA and LV; lower: bubbles in the LV). (c and d) Pulmonary CTPA at the 1-month follow-up revealed no residual emboli.
The patient was transferred to the Vascular Surgery Ward, where anticoagulation treatment was continued with low–molecular-weight heparin (LMWH) and rivaroxaban. Follow-up thrombophilia screening, including antiphospholipid antibodies, antithrombin III deficiency, protein C and S deficiency, antinuclear antibody, extractable nuclear antigen, and antineutrophil cytoplasmic antibodies, yielded negative results; however, protein C, protein S, and antithrombin III levels were considered unreliable owing to the ongoing LMWH and rivaroxaban therapy. The final diagnosis was PE, PFO, lower-extremity DVT, and acute right frontal cerebral infarction. After her condition stabilized, she was discharged on long-term rivaroxaban (20 mg daily) for PE and DVT management.
The 1-month follow-up pulmonary CT angiography showed no residual emboli (Figure 3). The patient remains under long-term follow-up in the Vascular Surgery Department.
Discussion
This patient likely had an underlying hypercoagulable state, predisposing her to DVT. Her clinical trajectory—distal DVT → submassive PE → acute ischemic stroke—exemplifies the pathophysiological cascade of PDE, aligning with established mechanisms and contemporary guidelines. Venous embolization caused subacute PE and secondary pulmonary hypertension, leading to RLS through the PFO and subsequent PDE.
All intracardiac communications—including atrial/ventricular septal defects, cyanotic congenital heart defects, and pulmonary arteriovenous malformations—carry a risk of PDE. The most common associated defect is PFO, 6 present in 20%–25% of adults.7–9 Physiologically, the left atrial (LA)-to-right atrial (RA) pressure gradient prevents embolization through a PFO. However, submassive PE increases right ventricular (RV) afterload, transiently elevating RA pressure above LA pressure—a critical trigger for RLS through the PFO. 10 This reversal allows platelet aggregates, clots, or other particulate matter to travel from the systemic venous circulation to the brain. In this case, PE-induced transient RV pressure elevation caused RLS, leading to PDE; thrombi from the lower-extremity DVT traversed the PFO, occluding cerebral arteries and causing stroke.
PDE may present latently with mild symptoms, leading to frequent misdiagnosis. Timely diagnosis is critical to prevent potentially life-threatening complications. 5 In cases of concurrent venous thromboembolism and arterial embolism, PDE should be suspected; in addition to treating acute events, thorough investigation of the embolic source is essential to prevent recurrence.
Diagnosis of cardiac defects relies on transthoracic echocardiography, transesophageal echocardiography (TEE), and saline contrast echocardiography (SCE). 11 Transthoracic echocardiography is noninvasive and accessible; however, it is less sensitive than TEE for detecting PFO. In the present case, transthoracic ultrasound was inconclusive, and diagnosis was confirmed via SCE (involving injection of agitated saline to assess the intracardiac blood flow). 12 Saline transthoracic contrast echocardiography is the primary method for detecting intracardiac (interatrial) shunting and remains the standard for assessing RLS severity, aiding the identification of high-risk PFOs eligible for transcatheter closure.13–15
Not all PFOs carry the same stroke risk. Patients at a high risk of recurrent embolism require intracardiac shunt closure. Evidence indicates that patients with moderate/large PFOs or atrial septal aneurysms (ASAs) face a higher risk of recurrent stroke and derive greater benefit from PFO closure.16,17 A “large PFO” is defined as that with the appearance of >20 microbubbles in the left atrium within 3 cardiac cycles after RA opacification. 18 Other high-risk features include PFO diameter >3.4 mm (ranging up to 5.8 mm in some studies), highly mobile PFO valves, a well-developed Eustachian valve, Valsalva maneuver preceding the event, recurrent embolic history, and hypercoagulable states. 19 In patients with recent ischemic stroke due to PFO with large RLS or ASA, PFO closure plus long-term antiplatelet therapy significantly reduces stroke recurrence compared with antiplatelet therapy alone.20,21
Management decisions were guided by risk stratification and aligned with current PDE and PFO-related stroke guidelines. Thrombolysis with 50 mg rt-PA (weight-based) was administered within the 4.5-hour window for acute ischemic stroke, consistent with the American Heart Association/American Stroke Association guidelines. 22 An IVCF was temporarily placed to prevent recurrent PE during the post-thrombolysis period, guided by the patient’s clinical context, thrombotic burden, and consistency with the 2020 American Society of Hematology guidelines, despite the absence of proximal DVT and the presence of distal DVT (specifically right below-knee venous thrombosis). 23 Long-term anticoagulation with rivaroxaban was initiated due to suspected underlying thrombophilia.
Limitations
This case report has certain limitations. First, we did not perform a transesophageal echocardiography to evaluate for the presence of microshunts. Second, the role of antiphospholipid syndrome in potentiating PFO-related embolism requires further exploration because this association is not fully characterized in the current literature.
Conclusion
PDE is a rare but critical diagnosis to consider in patients presenting with concurrent venous thromboembolism and arterial embolism. In addition to treating acute events, thorough investigation of the embolic source is essential to prevent recurrence. This case highlights how venous thrombi can bypass the pulmonary vasculature via a PFO or atrial septal defect to reach systemic arteries. Clinicians should recognize this possibility and consider interatrial shunt closure for secondary prevention.
Footnotes
Acknowledgements
We would like to thank the patient and her family. We extend our thanks to the laboratory and radiology departments of the Beijing Tsinghua Changgung Hospital for facilitating the acquisition of the relevant materials.
Authors’ contributions
Yu Chen and Pengshu Zhang are responsible for writing the article. All authors have read and approved the manuscript.
Data availability statement
The data supporting the findings of this study are available within the article.
Declaration of conflicting interests
The authors report no conflicts of interest.
Ethics statement and informed consent
We obtained ethical board approval for publication. Written informed consent for the publication of patient data was obtained from the patient’s families. The reporting of this study conforms to the Case Report (CARE) guidelines. 24
Funding
Not applicable.
