Abstract
Background
We aimed to investigate the clinical characteristics and influencing factors of severe influenza A virus pneumonia complicated by gram-negative bacterial infection in children.
Methods
We retrospectively analysed data from 43 paediatric patients with severe influenza A virus pneumonia who had been treated at our hospital. The patients were divided into observation (28 patients) and control (15 patients) groups based on whether they had a coinfection with gram-negative bacteria. The clinical data of the two groups were compared, and multivariate regression analysis was performed.
Results
The mean age in the observation group was 2.5 ± 1.53 years; 26 (92.86%) patients were aged ≤5 years, and 11 (39.3%) had underlying diseases. The mean age in the control group was 4.07 ± 2.11 years; 10 (66.67%) patients were aged ≤5 years, and 2 (13.33%) had underlying diseases. The differences in age and the proportion of children with underlying diseases were statistically significant (
Conclusion
Severe influenza A virus pneumonia complicated by gram-negative bacterial infection is more common in children aged <5 years. Younger age and the presence of underlying diseases increase the likelihood of gram-negative bacterial coinfection. Persistent fever, wheezing and significant elevations in C-reactive protein level, procalcitonin level, platelet count and monocyte count are important early warning signs for the clinical identification of gram-negative bacterial infection.
Introduction
Influenza, also known as the flu, is an acute respiratory infectious disease caused by the influenza virus and is capable of triggering global pandemics. During flu seasons, the incidence of influenza in children can reach 20%–30%.1–3 The predominant circulating strains of the influenza virus are influenza A (flu A) and influenza B, with influenza A exhibiting high antigenic variability and posing significant risks. 4 Pneumonia is the most common complication of influenza A in children, and the majority of severe cases and fatalities are attributed to pneumonia.5–7 Secondary bacterial coinfections are frequently occur following influenza virus infection, exacerbating disease severity and affecting prognosis. 8 Our preliminary research found that gram-negative bacteria (GNB) accounted for the majority of severe influenza pneumonia cases with bacterial coinfection in our hospital and were most commonly seen in cases of pneumonia with influenza A virus coinfection. 9 However, few domestic reports have described severe influenza A pneumonia complicated by GNB infection. Therefore, this study further analysed the clinical characteristics and influencing factors of hospitalised children with severe influenza A pneumonia and GNB coinfection to enhance the understanding of this disease and help determine appropriate prevention and treatment strategies for clinicians.
Patients and methods
Patients
This study adopted a retrospective research design and included children with severe influenza A virus pneumonia who were admitted to our hospital between 1 July 2019 and 30 June 2022. All patient details have been de-identified.
The inclusion criteria were as follows:
Aged 1 month to 12 years; Confirmed diagnosis of influenza A virus infection, defined as having an epidemiological history, presenting with influenza-like symptoms and testing positive for influenza A virus antigen; Meeting the diagnostic criteria for severe pneumonia.
Patients who met any one of the following conditions were considered to have severe pneumonia:
Poor general condition; Disturbance of consciousness; Cyanosis; Rapid breathing (age <2 months: respiratory rate (RR) ≥ 60 breaths/min; age ≥2 months to 1 year: RR ≥ 50 breaths/min; age >1–5 years: RR ≥ 40 breaths/min; and age >5 years: RR ≥ 30 breaths/min); Dyspnoea (moaning, nasal flaring and the ‘three depression’ sign, i.e. depression of the suprasternal fossa, supraclavicular fossa and intercostal space); Pulse oxygen saturation ≤92%; Refusal to eat or signs of dehydration; Plain chest radiography or computed tomography (CT) showing lung infiltration, pneumothorax, lung necrosis or lung abscess in ≥two-thirds of a lung; Extrapulmonary complications.
Patients who met the above inclusion criteria were enrolled and divided into two groups based on sputum culture results: those with negative cultures and no signs of bacterial infection were assigned to the control group, and those with GNB infections were included in the observation group. Patients with gram-positive bacterial infections were excluded. The criteria for a positive sputum culture were as follows: tracheal aspirate ≥105 colony-forming units (CFU)/mL or bronchoalveolar lavage fluid (BALF) ≥104 CFU/mL. Bacteria were observed on sputum smear staining, with evidence of leukocyte infiltration and phagocytosis.
The exclusion criteria were as follows:
Incomplete clinical data precluding statistical analysis; Gram-positive bacterial infections identified in cultures (our previous study found that among children with severe influenza–associated pneumonia and bacterial coinfection in our hospital, GNB were the predominant pathogens, while coinfection with gram-positive bacteria was relatively uncommon); Contaminated specimens or colonising bacteria, defined as sputum specimens with >10 squamous epithelial cells per high-power field (HPF) and <25 leukocytes per HPF, which were deemed unqualified; pathogens isolated from nonsterile sites without imaging evidence or clinical signs of infection were considered colonisers.
This study was approved by the Ethics Committee of our hospital (Approval number: XCSER [2023]28). All procedures conformed to the principles of the Declaration of Helsinki. As this was a retrospective analysis, the need for informed consent from patients and their families was exempted. This study followed the relevant Enhancing the QUAlity and Transparency Of health Research (EQUATOR) guidelines. 10
Methods
Data collection
Clinical data of paediatric patients were retrieved from the hospital’s electronic medical record (EMR) system, including clinical data, symptoms, signs, auxiliary examination results, treatment details, prognoses and patient outcomes. The differences between the two groups were compared.
Pathological examination methods
The pathological examination methods were as follows:
Statistical analysis
Data were statistically analysed using SPSS version 23.0 software (IBM Corp., Armonk, NY, USA). Normally distributed measurement data were expressed as mean ± standard deviation (SD) and compared between the two groups using the
Results
General data
Between 1 July 2019 and 30 June 2022, 49 paediatric cases of severe influenza A virus pneumonia were treated at our hospital. After excluding 6 patients with gram-positive bacterial coinfections, the study included 28 patients with GNB infections in the observation group and 15 patients without bacterial infections in the control group. In the observation group, there were 20 males (71.4%) and 8 females (28.6%), with a mean age of 2.5 ± 1.53 years. Among them, 26 cases (92.9%) were ≤5 years old, while 2 (7.1%) were >5 years old. Among the observed patients, 11 (39.3%) had underlying diseases: febrile seizures (n = 4), congenital heart disease (n = 3), developmental disorders (n = 2), epilepsy (n = 1), asthma (n = 1) and Kawasaki disease (n = 1), with 1 patient presenting with both epilepsy and Kawasaki disease. In the control group, there were nine males (60%) and six females (40%), with a mean age of 4.07 ± 2.11 years. Of these, 13 cases (86.7%) were ≤5 years old, while 2 (13.3%) were >5 years old. The control group included two cases (13.3%) of comorbidities (febrile seizure (n = 1) and growth retardation (n = 1)). Compared with the control group, the observation group had a lower mean age and a higher proportion of children aged ≤5 years with underlying diseases. Demographic comparisons are presented in Table 1.
Comparison of general data between the observation and control groups.
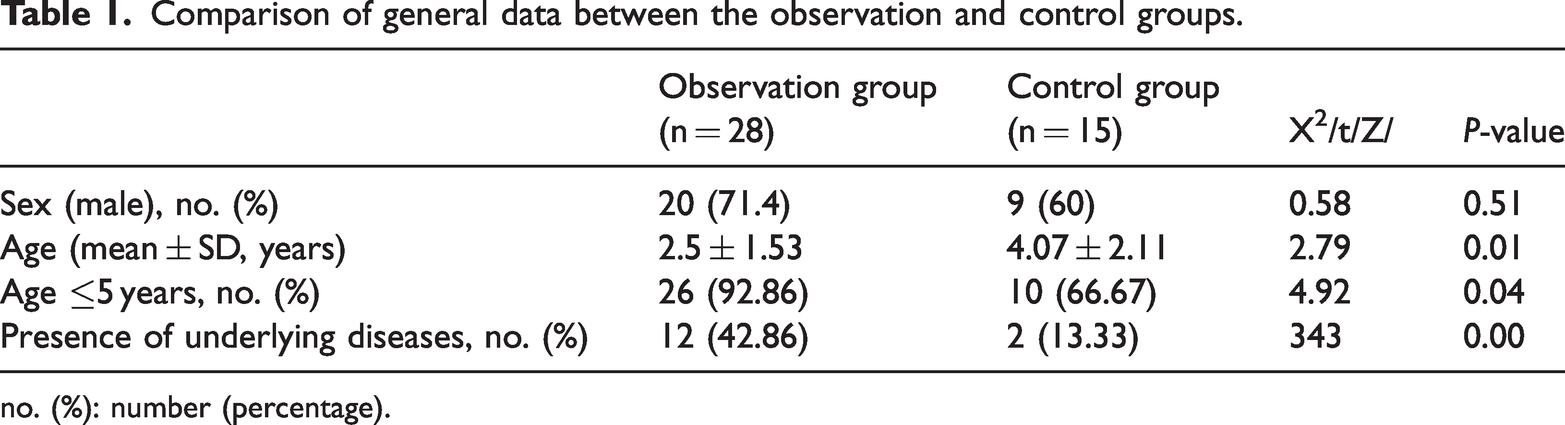
no. (%): number (percentage).
Clinical characteristics
All 28 patients in the observation group presented with high axillary fever, predominantly exhibiting continuous or remittent fever patterns. The median peak temperature reached 39.8°C (range, 39.5°C–40.0°C), with a median febrile duration of 6.5 (range, 5.75–6.5) days. Notably, 15 cases (53.57%) experienced prolonged fever lasting ≥7 days, 21 cases (75%) developed wheezing and 15 cases (53.57%) showed convulsions or consciousness disturbances. Radiological findings revealed pulmonary consolidation with atelectasis in 17 cases (60.7%). One case showed coinfection with respiratory syncytial virus, and three cases were coinfected with
Comparison of clinical characteristics between the observation and control groups.

M (P25, P75): median (interquartile range); no. (%): number (percentage); WBC: white blood cell; NEU: neutrophil; LY: lymphocyte; MO: monocyte; PLT: platelet; CRP: C-reactive protein; PCT: procalcitonin; MP: mycoplasma; ESR: erythrocyte sedimentation rate; CK: creatine kinase; CK-MB: creatine kinase MB isoenzyme; FDP: fibrinogen degradation products; LDH: lactate dehydrogenase.
Bacterial culture and sensitivity
Among the 28 paediatric patients in the observation group, a total of 30 culture-positive specimens were collected, including 28 oropharyngeal/nasal sputum samples and 2 BALF samples. Notably, two cases showed concurrent positivity in both sputum and BALF cultures. The isolated pathogens included
Treatment and outcomes
Both observation and control groups received neuraminidase inhibitor (NAI) therapy. In the observation group (n = 28), treatment regimens included oral/osmotic nasogastric oseltamivir (26 cases), intravenous peramivir (1 case) and sequential oseltamivir–peramivir therapy (1 case). Notably, only seven cases (25%) initiated NAI treatment within 48 h of symptom onset. All patients in the observation group received antibiotic therapy, with a median duration of 7 (range, 6–8) days. Advanced interventions included fiberoptic bronchoscopy with bronchoalveolar lavage (13 cases, 46.43%) and paediatric intensive care unit (PICU) admission (18 cases, 64.29%). The median hospitalisation duration was 13 (range, 9–15) days, with 22 cases (78.57%) hospitalised for ≥7 days. Mortality occurred in one patient aged ≤5 years with pre-existing epilepsy, who developed hemophagocytic syndrome. In the control group (n = 15), patients received oseltamivir (14 cases) or peramivir (1 case), with 7 cases (46.67%) initiating NAI therapy within 48 h. Fewer patients required bronchoscopy (four cases, 26.67%) or PICU care (two cases, 13.33%). The median hospitalisation duration was shorter at 7 (range, 6–10) days, with eight cases (53.33%) hospitalised for ≥7 days and no mortality. Comparative analysis demonstrated significantly higher rates of PICU admission (64.29% vs. 13.33%,
Comparison of treatment between the observation and control groups.

M (P25, P75): median (interquartile range); no. (%): number (percentage); PICU: paediatric intensive care unit.
Risk factors for severe influenza A virus coinfection with GNB
Intergroup comparisons were performed between the observation and control groups (Tables 1 to 3). Variables with statistically significant differences—including age of ≤5 years, underlying diseases and antiviral initiation within ≤48 h—were incorporated into the logistic regression model. The analysis identified age of ≤5 years, antiviral treatment initiation within ≤48 h and underlying diseases as risk factors for severe influenza pneumonia complicated by GNB infection. Specific details are shown in Table 4.
Risk factor analysis of severe influenza A pneumonia complicated by Gram-negative bacterial infection.

SE: standard error; OR: odds ratio; CI: confidence interval.
Discussion
This study provides an in-depth exploration of the etiological characteristics, risk factors and early identification markers of severe influenza A virus pneumonia complicated by GNB infections in children. In recent years, multiple domestic and international reports11–15 as well as our preliminary research
9
have demonstrated a significant shift in the pathogen spectrum of bacterial pneumonia in children—particularly in cases of severe influenza A virus pneumonia with bacterial coinfection—from traditional gram-positive predominance to gram-negative dominance. Our study, conducted in Xiamen, further confirms this epidemiological trend, with GNB accounting for 100% of cases in the observation group and
While elucidating this shift in the pathogen spectrum, the issue of antimicrobial resistance poses significant challenges for clinical management. Our study revealed that
Early identification of high-risk populations is crucial for timely intervention. This study clearly delineates two high-risk groups: young children (particularly those <5 years old) and those with underlying medical conditions. In the observation group, 92.86% of children were aged <5 years—significantly higher than that in the control group—and logistic regression analysis confirmed younger age as an independent risk factor for GNB coinfection (odds ratio (OR) = 2.36). This aligns with global data showing that children aged <5 years are at higher risk of severe influenza–related complications and mortality, often associated with bacterial coinfections.20,21 Their vulnerability stems from immature immune systems, making them more susceptible to influenza-induced disruption of immune barriers.15,16 Additionally, the proportion of children with underlying diseases in the observation group (42.86%) was significantly higher than that in the control group (13.33%). Logistic regression analysis identified underlying conditions as an independent risk factor for GNB coinfection (OR = 2.36), consistent with the findings of Yang et al. 20 and studies in Australia and Norway.22,23 Although the result is statistically significant with a high OR, the wide confidence interval (CI) for underlying diseases (95% CI, 1.28–87.2) suggests considerable uncertainty in the estimation. This wide CI may be attributed to the relatively small sample size in this study, which represents a limitation of our research. Despite this uncertainty, it is noteworthy that the lower bound of the CI (1.28) remains greater than 1, indicating that the underlying disease is an important risk factor (OR > 1). This finding alerts clinicians that the underlying condition may represent a highly significant risk factor and should be taken seriously. However, due to the uncertainty in the magnitude of the risk, caution is warranted when applying this information for precise risk assessment in individual patients. Future larger-scale studies—particularly prospective studies or meta-analyses targeting this specific patient population—are needed to narrow the CI and more accurately quantify the true effect size of this risk factor.
Early identification of severe influenza A pneumonia complicated by GNB infection in children is critical for improving clinical outcomes. Our study revealed that compared with children with uncomplicated severe influenza A pneumonia, those with GNB coinfection exhibited more alarming clinical and laboratory features: significantly prolonged fever duration (higher proportion of individuals with fever lasting ≥7 days), increased incidence of wheezing, markedly elevated inflammatory markers (CRP and PCT) and abnormal elevations in monocyte and platelet counts. Notably, disproportionate rises in CRP and PCT serve as sensitive indicators of bacterial infection, particularly GNB infection. The observed thrombocytosis may reflect platelet activation and recruitment during early sepsis,24,25 providing a potential warning sign for impending septic risk. Therefore, for high-risk children with severe influenza A pneumonia (particularly those aged <5 years or those with comorbidities), clinicians should maintain a high index of suspicion for GNB infection when encountering the following conditions: (a) persistent or recurrent high-grade fever; (b) new-onset or worsening wheezing; or (c) concurrent laboratory abnormalities, including significantly elevated CRP/PCT and abnormal platelet or monocyte counts. These indicators should prompt the consideration of empirical GNB coverage even before microbiological confirmation becomes available.
Based on these findings, we propose the following clinical recommendations. First, for children aged <5 years (particularly infants) and those with underlying medical conditions who develop severe influenza A pneumonia, clinicians should maintain a high level of suspicion for GNB infection and implement close monitoring of body temperature, respiratory status and inflammatory markers (CRP and PCT). Second, when children present with either clinical manifestations suggestive of GNB infection (e.g. persistent fever ≥ 7 days or wheezing) or laboratory warning signs (e.g. markedly elevated CRP/PCT levels and abnormal platelet/monocyte counts), empirical anti–gram-negative therapy should be initiated immediately without awaiting definitive microbiological confirmation. Considering the local pathogen distribution profile, empirical therapy should cover both
This study has several limitations. As a single-centre retrospective study, it only included cases from the period between July 2019 and June 2022, with a relatively small sample size (n = 28). The restricted case selection may introduce certain biases, potentially compromising the representativeness of the findings and statistical power. The definition of GNB coinfection was based on sputum and BALF culture results; however, the possibility of contamination or colonisation cannot be ruled out. Furthermore, as all cases were recruited from Xiamen in southern China, regional variations in genetic backgrounds, circulating influenza/bacterial strains and antibiotic prescribing practices may limit the external validity of our results. Future multicentre prospective cohort studies with larger sample sizes encompassing broader geographic regions are needed to more comprehensively characterise the epidemiology, better define risk factors and more accurately determine the prognostic implications of severe influenza A pneumonia with GNB coinfections in Chinese children. Such studies would also enable ongoing surveillance of pathogen distribution shifts and antimicrobial resistance patterns.
In conclusion, this study demonstrates that GNB (predominantly
Footnotes
Acknowledgements
We thank the patients, nurses and clinical staff who provided care for the patients.
Authors’ contributions
QYW, YL, ZQZ and JJH were involved in study design. QYW and JJH wrote the main manuscript and prepared the tables. FCW, SSY, YPG and YMW facilitated data collection. All authors reviewed the manuscript.
Availability of data and materials
All data generated during this study are included in this published article.
Clinical trial number
Not applicable.
Consent for publication
In this retrospective study, the need for written informed consent from the patients was waived, and the study was approved by the Ethics Committee of Xiamen Children’s Hospital (Approval No. XCSER [2023]28).
Declaration of conflicting interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
The study was approved by the Ethics Committee of Xiamen Children’s Hospital (Approval No. XCSER [2023]28).
Funding
This work was supported by the Natural Science Foundation of Xiamen Science and Technology Bureau (Grant No.: 3502Z20227147 and 3502Z20227301) and the 1125 Talent Project of Xiamen Children’s Hospital (Grant No.: 2020-1125-YQ-040).
