Abstract
Objective
Occult hepatitis B virus infection, characterized by the presence of hepatitis B virus DNA in the absence of detectable hepatitis B surface antigen, poses a significant risk to blood donation safety. This study aimed to determine the prevalence of occult hepatitis B virus infection and identify demographic and clinical predictors among blood donors in Saudi Arabia who were negative for hepatitis B surface antigen.
Methods
A retrospective cross-sectional study was conducted using donor records to evaluate hepatitis B virus and other transfusion-transmissible infection markers. Demographic data were extracted, and multivariable logistic regression was performed to identify predictors of hepatitis B core antibody and hepatitis B virus DNA positivity.
Results
Among the 49,502 donors negative for hepatitis B surface antigen, 27 had occult hepatitis B virus infection, representing a prevalence of 0.054%. Occult hepatitis B virus infection was more frequent among donors aged 31–40 and >51 years and was significantly associated with non-Saudi nationality, Middle Eastern and Asian origin, and syphilis coinfection. Syphilis was also significantly associated with prior hepatitis B virus exposure, as indicated by hepatitis B core antibody positivity. Notably, 5 of the 27 patients with occult hepatitis B virus infection (18.5%) were negative for hepatitis B core antibody, indicating that these cases would have been missed using serological screening alone.
Conclusion
The prevalence of occult hepatitis B virus infection among blood donors in Saudi Arabia is low but clinically relevant. These findings underscore the limitations of serological screening alone and support the incorporation of nucleic acid testing to enhance transfusion safety.
Keywords
Introduction
Despite significant advancements in healthcare, including the widespread implementation of vaccination programs, hepatitis B virus (HBV) infection remains a major global health concern. An estimated 254 million people live with chronic HBV infection, and 1.1 million deaths were reported in 2022 due to HBV-related complications such as cirrhosis and hepatocellular carcinoma. 1 In Saudi Arabia, recent data have estimated the current prevalence of HBV to be approximately 1.7%. 2
HBV belongs to the Hepadnaviridae family and is a circular, partially double-stranded, enveloped DNA virus. 3 A unique feature of HBV is its ability to persist in host hepatocytes as covalently closed circular DNA, a stable episom that serves as a template for viral replication. This allows HBV to evade immune clearance and contribute to infection persistence. 4
HBV is primarily transmitted through bloodborne, sexual, and vertical (mother-to-child) routes. Among these, transfusion-transmitted infection (TTI) is one of the most preventable routes and can be efficiently controlled by using effective blood donor screening protocols. 5 Clinically, HBV infection is classified into acute, chronic, and occult forms based on viral persistence, immune response, and detectability. These categories are well-established in clinical practice guidelines and the literature.4,6,7 When occult HBV infection (OBI) is not routinely screened, it poses a risk of transmission through blood transfusion and may lead to viral reactivation in immunocompromised individuals, highlighting the need for more sensitive detection strategies.
Existing HBV screening strategies used by blood banks commonly rely on the detection of hepatitis B surface antigen (HBsAg) and, in some settings, hepatitis B core antibody (anti-HBc). However, in countries with a high anti-HBc prevalence, many blood bank services have adopted universal HBV DNA testing instead of anti-HBc screening because deferring all anti–HBc-positive donors may lead to unnecessary loss of safe donations. 8 Although HBsAg serves as a marker of active infection, anti-HBc, especially in the absence of HBsAg, indicates prior exposure to HBV and the presence of OBI. However, relying on serological markers alone may result in missed detection of infection cases, particularly during the window period and in cases of anti-HBc–negative OBI, where HBV DNA persists despite HBsAg negativity. To address this gap, nucleic acid testing (NAT) has been mandated as part of donor blood screening in many countries, including Saudi Arabia.9,10
Although the HBV prevalence in Saudi Arabia has declined since the implementation of the national HBV vaccination program in 1989, data on the burden and characteristics of OBI at the national level remain scarce. 11 Moreover, limited studies have systematically examined the demographic or clinical predictors associated with OBI markers such as anti-HBc and HBV DNA positivity.
In addition to assessing the prevalence of HBV infection in the screened blood donor population, this study primarily aimed to determine OBI prevalence and characterize OBIs among HBsAg-negative blood donors. Furthermore, demographic, regional, and coinfection-related predictors of anti-HBc and HBV NAT positivity were evaluated.
Method
Study design and setting
This retrospective study was conducted at King Fahad Medical City (KFMC) in Riyadh, Saudi Arabia. Blood donor data were collected from the electronic medical records of the KFMC blood bank between January 2022 and December 2023. The study population comprised all individuals aged 18–60 years who donated blood during the study period and for whom complete serological and molecular screening data were available. All eligible donors were included consecutively; therefore, the sample size was determined by the total number of donors within this timeframe. Donors with missing or incomplete records were excluded from the analysis. Screening included tests for HBsAg, anti-HBc, and HBV DNA using NAT, along with additional screening for TTIs, including human immunodeficiency virus (HIV), hepatitis C virus (HCV), syphilis, and malaria. The study protocol was reviewed and approved by the Institutional Review Board of KFMC (approval no. 24-254). This was a retrospective analysis of donor data; therefore, the requirement for informed consent was waived. All donor records were fully de-identified before analysis to ensure that no individual could be identified in any way. All procedures were conducted in accordance with the principles of the Declaration of Helsinki (1975, as revised in 2024) and local ethical guidelines. The reporting of this study was conducted in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines for observational studies. 12
Data extraction and variables
Demographic information, including age, sex, nationality, and region of origin, was extracted from donor records. Age was categorized into the following four groups: 18–30, 31–40, 41–50, and >51 years. Sex, nationality (Saudi Arabian or non-Saudi Arabian), and region of origin (Middle Eastern, Asian, African, European, American, or Other) were recoded as categorical variables. Regions with sparse data were either collapsed into broader categories or excluded during regression modeling. Malaria and syphilis statuses were grouped into positive, negative, or indeterminate categories based on test results. Only individuals with definitive positive or negative results were included in the multivariable analysis. Similarly, serological and NAT results for HCV and HIV were used to classify infection status as either positive or negative.
OBI was defined as the presence of HBV DNA in individuals who tested negative for HBsAg. Among OBI cases, further stratification was performed according to anti-HBc status. Patients who were positive for both HBV DNA and anti-HBc were classified as OBI+, while those negative for anti-HBc were classified as OBI−.
Serological and NAT assays
Serological screening for TTIs was performed using chemiluminescent microparticle immunoassays on the Alinity s platform (Abbott Laboratories, USA) as per the manufacturer’s instructions. For HBsAg detection, the Alinity s HBsAg assay was used, with a manufacturer-reported specificity of 99.97% (95% confidence interval (CI): 99.92%–99.99%) and sensitivity of 100% (95% CI: 99.27%–100.00%). Testing for anti-HBc was conducted utilizing the Alinity s Anti-HBc assay, with a specificity of 99.90% (95% CI: 99.77%–99.97%). HIV screening was performed utilizing the HIV Ag/Ab Combo assay, which screens for HIV-1 p24 antigen as well as antibodies against HIV-1 and HIV-2, with a reported specificity of 99.97% (95% CI: 99.93%–99.99%) and sensitivity of 100%. HCV antibodies were screened using the anti-HCV II assay, covering genotypes 1–6, with a specificity of 99.95% (95% CI: 99.92%–99.98%) and sensitivity of 100%. Screening for human T-lymphotropic virus I (HTLV-I)/HTLV-II was performed using the HTLV-I/II assay, with a manufacturer-reported specificity of 100.00% (95% CI: 99.93%–100.00%) and sensitivity of 100%. Syphilis screening was performed using the syphilis assay to detect antibodies against Treponema pallidum, with a reported specificity of 99.95% (95% CI: 99.86%–99.99%) and sensitivity of 100%. Malaria testing was performed utilizing the DiaMed Malaria Antibody enzyme immunoassay (Bio-Rad, France) for antibodies against Plasmodium species, showing >95% sensitivity and >98% specificity in comparative evaluations with reference panels, including those from the National Institute for Biological Standards and Control (NIBSC, UK). HIV-1, HIV-2, HCV, and HBV NAT testing were performed using the Cobas® MPX test (Roche Diagnostics, Switzerland) on the Cobas 6800/8800 system. This multiplex real-time polymerase chain reaction platform targets several genomic regions per virus, allowing simultaneous identification and differentiation of the four pathogens in a tested sample. NAT was performed using ethylenediaminetetraacetic acid (EDTA) plasma obtained from peripheral blood donations. According to manufacturer data, the assay achieved 100% sensitivity for all targeted agents in validation studies. The 95% limit of detection for HBV DNA using the Cobas® MPX test on the Cobas® 6800/8800 system was 1.4 IU/mL in EDTA plasma (95% CI: 1.2–1.7 IU/mL), according to the manufacturer’s information. All the described assays are part of the routine blood donor screening protocol at KFMC and are conducted in accordance with the regulations of the Saudi Ministry of Health.
Statistical analyses
Data were imported and analyzed using R statistical software (version 4.0), utilizing the dplyr (version 1.1.4), readxl (version 1.4.0), gtsummary (version 1.7.2), broom (version 1.0.5), and forcats (version 1.0.0) packages.
Descriptive statistics were generated using the “tbl_summary” function of the gtsummary package. Categorical comparisons were performed using Fisher’s exact test, with simulated p-values to account for small cell sizes. Overall distributions and subgroup counts were reported using the “add_overall” and “add_n” functions, respectively. 13 Two multivariable logistic regression models were constructed using generalized linear modeling with a binomial distribution and logit link. The first model assessed predictors of anti-HBc positivity as an indicator of previous HBV exposure, while the second model evaluated predictors of HBV NAT positivity as a marker of active or occult infection. Both models included age group, nationality, region, malaria status, and syphilis status as independent variables. Reference levels were set as 18–30 years for age, Saudi Arabian for nationality, Middle Eastern for region of origin, malaria-negative for malaria status, and syphilis-negative for syphilis status. Region-of-origin categories with no events or an insufficient sample size were removed or collapsed into broader groups to maintain model stability. Factor reference levels were defined using the “relevel” function of the forcats package, and collapsing of sparse levels was conducted using the “fct_collapse” function.
Model fitting was performed using the “glm” function, and model outputs were extracted using the “broom::tidy” function to obtain odds ratios (ORs), CIs, and p-values. Final tables were generated using the “tbl_regression” function with exponentiated coefficients. In cases where perfect or near-perfect separation was detected, especially for variables with very low event counts, additional level merging or filtering was applied to enable model convergence and interpretability.
Results
Demographic and clinical differences between OBI and non-OBI donors
Among the 49,502 HBsAg-negative individuals, 27 (0.054%) were identified as having OBI, defined as HBV DNA positivity on NAT. The flow of donor screening, classification by HBsAg and NAT results, and subsequent stratification of OBI cases based on anti-HBc status are shown in the study design diagram in Figure 1. These individuals were compared with 49,475 HBsAg-negative and HBV DNA–negative donors (non-OBI group) (Table 1). The OBI status was significantly associated with age group (p < 0.001), with the highest prevalence observed among individuals aged 31–40 (37%) and >51 (33%) years. A significant association was also noted for nationality (p < 0.001), with OBI being more common among non-Saudi Arabian donors (59%). Region of origin was significantly associated with OBI status (p = 0.002); most OBI patients were of Middle Eastern (59%) or Asian (37%) origin. Syphilis coinfection also showed a statistically significant association with OBI status (p = 0.021); however, no significant associations were observed for sex, malaria status, or markers of HIV and HCV infection. Of the total donor population, 44 (0.09%) were HBsAg-positive; their demographic characteristics are summarized in the supplementary table.
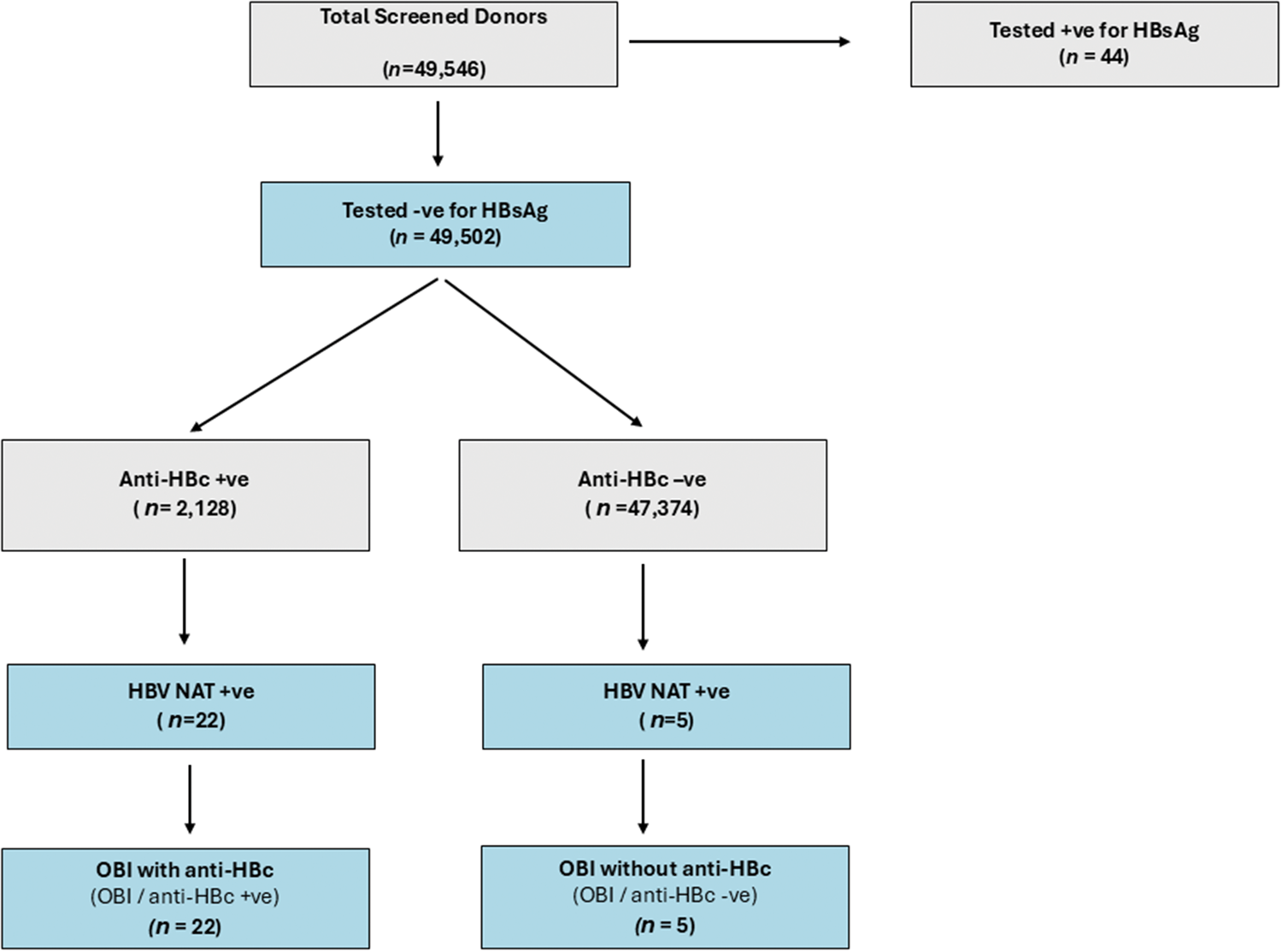
Flow diagram illustrating the classification of blood donors based on HBV serological and NAT screening. Of the 49,546 individuals tested, 44 were HBsAg-positive and excluded from the OBI analysis. Anti-HBc testing was performed among the 49,502 HBsAg-negative donors; 2128 were positive for anti-HBc and 47,374 were negative for anti-HBc. NAT screening for HBV DNA was then performed for both groups. In total, 27 donors were HBV DNA–positive based on NAT and were classified by OBI status as follows: 22 had OBI with anti-HBc (OBI/anti-HBc+) and 5 had OBI without anti-HBc (OBI/anti-HBc−). HBV: hepatitis B virus; NAT: nucleic acid testing; HBsAg: hepatitis B surface antigen; OBI: occult hepatitis B virus infection; anti-HBc: hepatitis B core antibody.
Demographic and clinical characteristics of HBV NAT–positive (OBI) and HBV NAT–negative (non-OBI) donors among HBsAg-negative individuals.

Percentages are based on the total number of HBsAg-negative donors (N = 49,502).
HBV: hepatitis B virus; NAT: NAT: nucleic acid testing; OBI: occult hepatitis B virus infection; HBsAg: hepatitis B surface antigen; HIV: human immunodeficiency virus; HCV: hepatitis C virus.
OBI prevalence and anti-HBc stratification
Among the 27 identified OBI cases, the majority (n = 22; 81.5%) were positive for anti-HBc (OBI-HBc+), while 5 (18.5%) were negative for anti-HBc (OBI-HBc−) (Table 2). OBI-HBc positivity was most commonly observed among donors aged 31–50 years (64%), whereas OBI-HBc negativity was more evenly distributed across age groups, with a higher proportion observed in the 41–50 years group (40%). The difference in age distribution was not statistically significant (p = 0.2).
Demographic and clinical characteristics of OBI cases stratified by anti-HBc status: OBI with anti-HBc (OBI/anti-HBc+) and OBI without anti-HBc (OBI/anti-HBc−).

Percentages represent the proportion of each subgroup of the total HBsAg-negative donor population (N = 49,502).
OBI: occult hepatitis B virus infection; anti-HBc: hepatitis B core antibody; HIV: human immunodeficiency virus; HCV: hepatitis C virus.
Compared with OBI-HBc− (40%) patients, a significantly higher proportion of OBI-HBc+ patients were non-Saudi Arabians (64%, p < 0.001). Furthermore, 55% of OBI-HBc+ patients were from the Middle East and 45% were from Asia, while 80% of OBI-HBc− patients were from the Middle East (p < 0.001).
Syphilis coinfection was observed in only one individual from the OBI-HBc+ group (4.5%; p = 0.019). Malaria coinfection was more common in OBI-HBc− patients (20%) than in OBI-HBc+ patients (4.5%); however, this difference was not statistically significant (p = 0.2). No HIV- or HCV-positive cases were detected in either group.
Predictors of anti-HBc positivity
A multivariable logistic regression model was employed to identify factors associated with anti-HBc positivity among HBsAg-negative individuals (Table 3). According to the analysis, age was the strongest independent predictor. Compared with individuals aged 18–30 years, those aged 31–40, 41–50, and >51 years had significantly higher odds of anti-HBc positivity (OR = 2.93, 95% CI: 2.45–3.53; OR = 7.42, 95% CI: 6.24–8.90, and OR = 13.8; 95% CI: 11.5–16.6, respectively; p < 0.001). Furthermore, the results of our analysis revealed that non-Saudi Arabians had higher odds of anti-HBc positivity than their Saudi Arabian counterparts (OR = 1.39, 95% CI: 1.24–1.56; p < 0.001).
Multivariable logistic regression analysis of the predictors of anti-HBc positivity among HBsAg-negative individuals.

anti-HBc: hepatitis B core antibody; HBsAg: hepatitis B surface antigen; OR: odds ratio; CI: confidence interval.
Compared with individuals from the Middle Eastern region, those from the Asian and African regions had significantly higher odds of anti-HBc positivity (OR = 2.13, 95% CI: 1.86–2.45 and OR = 1.61, 95% CI: 1.26–2.04, respectively; p < 0.001 for both). Coinfection with malaria or syphilis was associated with higher odds of anti-HBc positivity (OR = 1.65, 95% CI: 1.36–2.00; p < 0.001 and OR =4.27, 95% CI: 1.34–11.3; p = 0.007, respectively).
Predictors of HBV NAT positivity
Another multivariable logistic regression model was used to identify predictors of HBV NAT positivity among HBsAg-negative individuals (Table 4). Age remained a strong and independent predictor, with those aged 31–40, 41–50, and >51 years having significantly higher odds of HBV DNA detection (OR = 11.2, 95% CI: 3.34–69.3; OR = 17.8, 95% CI: 5.30–111; and OR = 26.8, 95% CI: 7.58–170, respectively; p ≤ 0.001 for all). Syphilis-positive individuals also had markedly increased odds of HBV NAT positivity (OR = 23.0, 95% CI: 1.26–116; p = 0.003). No statistically significant associations were observed for nationality, region of origin, or malaria status in this model.
Multivariable logistic regression analysis of the predictors of HBV NAT positivity among HBsAg-negative individuals.

HBV: hepatitis B virus; NAT: nucleic acid testing; HBsAg: hepatitis B surface antigen; OR: odds ratio; CI: confidence interval.
Discussion
OBI is an important but often under-recognized factor in HBV epidemiology. In this study, which involved 49,546 blood donors screened using both serological and molecular assays, we identified a low overall prevalence of OBI (0.05%) among HBsAg-negative donors.
Additionally, 44 individuals (0.09%) tested positive for HBsAg, indicating active HBV infection. This percentage is substantially lower than that reported in the western and eastern regions of the country, where the prevalence of active HBV infection among blood donors reached 0.4% and 3.24%, respectively.14,15 The comparatively lower prevalence of HBV infections among blood donors observed in this study may reflect the cumulative success of sustained vaccination efforts, early implementation of preventive strategies, and robustness of public health infrastructure in the central region. These findings also underscore notable regional disparities in HBV infection rates across Saudi Arabia, suggesting the need for public health interventions to consider local epidemiological contexts.
Although the prevalence of OBI in this study (0.05%) is low, it is consistent with the rates reported in low-endemic populations and has significant implications for blood safety and clinical monitoring. 16 Similar OBI prevalence rates have been reported among Saudi Arabian blood donors, including 0.057% in the Aseer and 0.05% in the Eastern regions, supporting the reliability of our findings.15,17 The key concern is that these cases may not be detected using standard HBsAg-based screening alone, highlighting the critical role of NAT in improving the sensitivity of HBV detection among blood donors. The prevalence of anti-HBc positivity (4.3%) and OBI (0.054%) observed in this study were higher than those reported for low-endemic countries in a recent international review, where the prevalence of anti-HBc positivity ranged from 0.42% to 1.19% and that of OBI was 0.0024%. 8
There are several slightly different definitions of OBI used in clinical practice and research; however, they all agree that the key diagnostic criterion for OBI is the detection of HBV DNA in the absence of HBsAg. The World Health Organization defines OBI as the presence of HBV DNA in the liver, with or without detectable DNA in the blood, in individuals who test negative for HBsAg using current assays. 18 In practice, liver biopsy is rarely performed; therefore, NAT is more commonly used for detection. The definition of OBI provided by the Centers for Disease Control and Prevention, particularly for studies involving blood donors, is as follows: HBV DNA-reactive, HBsAg-nonreactive, and anti-HBc-positive, regardless of the antibody to hepatitis B surface antigen (anti-HBs) status. 19 The European Association for the Study of the Liver (EASL) distinguishes seropositive OBI (anti-HBc positivity with or without anti-HBs positivity) from seronegative OBI (negative for both markers) and states that HBV DNA levels are usually very low in these cases. 7 The Taormina expert group has also defined OBI as HBV DNA positivity in the absence of HBsAg and classified cases as “true OBI” when they result from low-level replication or “false OBI” when they are caused by mutations that impair HBsAg detection. 20 In summary, these definitions emphasize the importance of combining molecular testing with serology to prevent the underdiagnosis of HBV infection in clinical and blood screening contexts.
In our study, all 27 OBI patients were HBsAg-negative and HBV NAT–positive. Among them, 22 (81.5%) were anti-HBc-positive and classified as OBI/anti-HBc+, while 5 (18.5%) had no detectable anti-HBc and were classified as OBI/anti-HBc−. This finding is consistent with that of previous reports showing that a minority of OBI cases lack anti-HBc and therefore would be missed using serological screening alone. 16 This highlights the critical role of molecular testing in identifying seronegative OBIs. HBV transmission from an anti-HBc-negative donor has also been reported. 8 The anti-HBc distribution among NAT-positive individuals in our cohort aligns with global data, which indicate that most but not all OBI patients show serological evidence of past HBV exposure.
The results of the logistic regression models further clarified the population characteristics associated with both HBV exposure and OBI. Anti-HBc positivity was strongly associated with older age, particularly age ≥41 years. This association may reflect cumulative exposure to HBV over time because older individuals have had a longer exposure to the risk of acquiring HBV infection. The gradual decline in immune protection with aging may also contribute to the increased serological evidence of past HBV exposure observed in older age groups. 21 Non-Saudi Arabian nationality as well as African and Asian regional origin were associated with higher odds of anti-HBc positivity. These associations are consistent with the high HBV endemicity reported in regions such as South-East Asia and sub-Saharan Africa, where at least 8% of the population are chronic HBV carriers, and 70%–95% show past or present evidence of infection. 22 Differences in historical vaccination coverage, healthcare access, and perinatal transmission rates across population groups may further contribute to these disparities. In addition to geographic and demographic factors, the role of coinfections in HBV transmission risk was evaluated. Syphilis coinfection emerged as a significant predictor of both anti-HBc and HBV NAT positivity. This likely reflects shared sexual transmission pathways between syphilis and HBV because individuals with a history of syphilis are at increased risk of HBV infection. 23 Notably, region of origin and nationality were strong predictors of anti-HBc positivity; however, they were not statistically significant predictors of HBV NAT positivity. This may be due to the small number of NAT-positive cases, which may have limited the power to detect these associations.
Although OBI prevalence was very low in this study, it remains a serious concern due to the potential for transmission through blood transfusion and organ transplantation, reactivation under conditions of immunosuppression leading to severe or even fulminant hepatitis, acceleration of progression in chronic liver disease, and continued risk of hepatocellular carcinoma development. 16 The detection of OBI patients without anti-HBc (OBI/anti-HBc−) in this study challenges the assumption that anti-HBc is an efficient surrogate for OBI. These findings support the value of incorporating NAT into HBV screening programs to enhance the safety of blood transfusions and organ transplantation. Quantitative levels of anti-HBc have been reported as potential predictors of OBI in a recent study. 24 Although this marker was not available in our dataset, its future application in blood donor screening may support risk stratification alongside existing serological and molecular assays. However, based on our findings, we do not recommend universal anti-HBc screening at this stage. We support the continued use of NAT as a core component of HBV screening; however, we acknowledge that implementation strategies must consider local prevalence, resource availability, and cost-effectiveness.
Although NAT is currently the most effective tool for identifying OBI cases missed using serology, its sensitivity remains limited by detection thresholds. Donors with extremely low HBV DNA levels may therefore escape detection. Transfusion-transmitted HBV infections have been reported in such cases despite negative NAT results. 25 Consequently, the true burden of OBI may have been underestimated in this study. Current HBV screening tools may fail to detect OBI in some cases, and emerging biomarkers such as hepatitis B core-related antigen and pre-genomic RNA have shown the potential to improve detection rates. However, these assays are still under investigation and are not yet suitable for routine blood donor screening. 26
Although this study has several strengths, including a large cohort of blood donors and a detailed analysis of demographic and coinfection predictors of HBV exposure and OBI, several limitations should be acknowledged. First, the small number of HBV NAT–positive individuals limited the statistical power to detect associations in certain subgroups. Second, HBV DNA detection was performed only using serum samples, rather than liver tissue samples, which is the gold standard for confirming occult infection. Third, the classification of OBI cases relied solely on anti-HBc status because additional serological markers such as anti-HBs and antibody to hepatitis B e antigen were unavailable, preventing a more comprehensive assessment of the immune profile in these cases. Fourth, sequencing of NAT-positive samples was not performed, limiting the exploration of viral diversity and potential genotype-specific patterns. In addition, the absence of unique donor identifiers prevented differentiation between first-time and repeat donations; therefore, some of the 27 OBI-positive donations may have originated from the same donor, potentially leading to a modest overestimation of OBI prevalence at the individual level. Finally, although the sample size was large, the data collection period spanned only 2 years. This relatively short duration, combined with the low prevalence of HCV and HIV infections in the cohort, may have limited the ability to detect meaningful associations between OBI and these infections.
Conclusion
This study provides valuable epidemiological evidence regarding OBI among blood donors in Saudi Arabia. It highlights the limitations of serological testing alone and provides evidence for a more nuanced, molecular-based approach to HBV screening. The inclusion of NAT in routine screening protocols may help improve detection, particularly among older adults and individuals with coinfections.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251387447 - Supplemental material for Occult hepatitis B virus infection and its demographic and clinical determinants among blood donors in Saudi Arabia: A retrospective cross-sectional study
Supplemental material, sj-pdf-1-imr-10.1177_03000605251387447 for Occult hepatitis B virus infection and its demographic and clinical determinants among blood donors in Saudi Arabia: A retrospective cross-sectional study by Abdulrahman F Alrezaihi, Lama Alzamil, Abdulrahman Alshalani, Jawaher Alsughayyir, Fahad S Alshehri and Abdulaziz M Almuqrin in Journal of International Medical Research
Footnotes
Acknowledgments
The authors thank the Ongoing Research Funding program, (ORF-2025-933), King Saud University, Riyadh, Saudi Arabia.
Author contributions
Conceptualization: A.M.A. and A. F. A.; methodology: A. F. A. and L A.; software: A. F. A.; validation: A.M.A. and A. F. A.; formal analysis: A. F. A. and J.A.; investigation: A. F. A and A. A.; resources: F.A.; data curation: A.A.; writing—original draft preparation: all authors; writing—review and editing: A.M.A.; visualization: A.M.A. and A. F. A.; supervision: A.M.A.; project administration: A.M.A. and A. F. A; funding acquisition: A.M.A. All authors have read and agreed to the submitted version of the manuscript.
Data availability statement
All data analyzed in this study are included in the manuscript.
Declaration of conflicting interests
The authors declare that they have no competing interests.
Funding
This project was funded by the Ongoing Research Funding program, (ORF-2025-933), King Saud University, Riyadh, Saudi Arabia.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
