Abstract
Objective
To investigate the possible correlation between histone deacetylase inhibition and radiosensitivity in PC-3 prostate cancer cells and to explore the possible mechanism involved.
Methods
PC-3 prostate cancer cells were treated with 0.40 μM CG-745 alone, radiation alone, or 0.40 μM CG-745 in combination with radiation. A CCK-8 kit was used to measure the proliferation of PC-3 cells. The colony formation assay was used to determine cell reproductive survival. Immunofluorescence analysis was used to detect the location of phosphorylated H2AX foci. Apoptosis and cell cycle distribution of PC-3 cells were assessed via flow cytometry analysis.
Results
CG-745 enhances the repressive effect of irradiation on PC-3 cell growth, as shown by the CCK-8 assay and colony formation assay. Compared with CG-745 or radiation alone, CG-745 combined with radiation significantly increased phosphorylated H2AX foci formation. The combination of CG-745 and radiation significantly increased the percentage of cells in the S phase and decreased percentage of cells in the G2 and G1 phases compared with treatment with CG-745 alone or radiation alone. Flow cytometry analysis showed that CG-745 promoted the PC-3 cell apoptosis induced by radiation.
Conclusions
The histone deacetylase inhibitor CG-745 enhanced radiation-induced DNA damage, cell cycle arrest, and cell apoptosis, thus increasing the radiosensitivity of PC-3 prostate cancer cells to X-ray irradiation.
Introduction
Histone deacetylases (HDACs) can regulate the expression of genes by regulating the acetylation of histones. 1 In tumor cells, the balance between histone acetyltransferases and HDACs is often disrupted. When histone acetylation is inhibited, it leads to the expression of oncogenes and the silencing of the expression of tumor suppressor genes. 2 High HDAC expression and histone hypoacetylation, which are related to posttranscriptional gene regulation, have been observed in tumor tissues, making them important therapeutic targets for many human cancers. 3 In acute promyelocytic leukemia, HDAC inhibition has no effect on normal cells and cells at the preleukemic stage. However, in tumor cells, HDAC inhibition leads to tumor-specific cell death through the TRAIL and FAS pathways. 4 Research has shown that by affecting the level of histone acetylation thus acting on multiple signal transduction pathways in tumor cells, HDAC inhibition can induce cell cycle arrest, cell differentiation, apoptosis, tumor suppression, and immune activation, resulting in strong anti-tumor activity.5–7 Many studies have shown elevated expression of HDAC1 in multiple cancers including prostate cancer. 8 The expression of class I HDACs, such as HDAC1 and HDAC2, escalates in prostate cancer and is higher in patients with castration-resistant prostate cancer, adversely affecting the disease’s prognosis. 9 CG-745 is a class I and class IIb HDAC inhibitor that has an inhibitory effect on HDAC1, 2, 3 and 6. 10 CG-745 has an anticancer effect in various cancer cells, including prostate cancer, pancreatic cancer, renal cell carcinoma, and colon cancer, both as a monotherapy and in combination with other anticancer therapies. 8
According to the cancer statistics in 2024, prostate cancer represents the most common malignant tumor in men in the United States, accounting for 29% of all new cancer cases in men and representing the second leading cause of cancer-related death in men, responsible for 11% of cancer-related deaths. 11 Radiotherapy plays an important role in the treatment of prostate cancer. Radiotherapy mainly leads to DNA damage, and repair after DNA damage is an important cause of the resistance to radiotherapy. Homologous recombination (HR) and non-homologous end joining (NHEJ) are the two main repair pathways for DNA double-strand breaks (DSBs).12,13 HDAC inhibition can also regulate the expression of multiple proteins involved in DNA damage reactions, thus inhibiting the HR and NHEJ pathways. 14 The combination of HDAC inhibitors and DNA-damaging agents induces a higher number and prolonged duration of phosphorylated histone H2AX (γH2AX) nuclear foci, which are classical markers of DSBs.15–17 Research has shown that inhibiting the expression of HDACs can improve the effects of drugs that cause DNA DSBs. 18
Considering the important functions of HDACs in the DNA damage response, we hypothesized that HDAC inhibition may reduce the capacity for DNA repair in prostate cancer cells after X-ray irradiation, thus increasing their radiosensitivity. The present study aimed to investigate the possible correlation between HDAC inhibition and radiosensitivity in PC-3 prostate cancer cells and explore its possible mechanism.
Materials and methods
Cell culture and drugs
PC-3 prostate cancer cells (RRID:CVCL_0035) were obtained from the Shanghai Institute for Biological Science (Shanghai, China) and cultured in RPMI-1640 medium (Gibco, Waltham, Massachusetts, United States) supplemented with 10% fetal bovine serum (Gibco, Waltham, Massachusetts, United States) at 37°C in a humidified atmosphere with 5% CO2. The HDAC inhibitor CG-745 was obtained from Selleck Chemicals (Shanghai, China).
Drug treatment and X-ray irradiation
PC-3 prostate cancer cells were treated with 0.40 μM CG-745 alone, radiation alone, or 0.40 μM CG-745 in combination with radiation. Radiotherapy was performed using an RS-2000 Pro X-ray irradiator (Rad Source Technologies, Buford, Georgia, United States) with 3 Gy of X-ray irradiation alone or in combination with CG-745. Radiation treatment was conducted immediately after CG-745 treatment.
CCK-8 assay
We used a CCK-8 kit (Beyotime, Shanghai, China) to measure the proliferation of PC-3 cells. PC-3 cells were plated in 96-well plates (2 × 104 cells/well) and treated with CG-745 alone, radiation alone, or CG-745 in combination with radiation. After that, all cells were cultured for 48 h. Subsequently, 10% CCK-8 solution was added to each well, and the cells were cultured for 1 h at 37°C. The absorbance was measured at 450 nm using a microplate reader. Cell survival was expressed as the absorbance of each condition relative to that of untreated controls.
Colony formation assay
The survival rate of clone formations was used to assess the potential for cell proliferation. PC-3 cells were seeded into 6-well plates and treated with CG-745 alone, radiation alone, or CG-745 in combination with radiation. After 2 weeks of incubation, the cells were washed with phosphate-buffered saline (PBS), fixed with 4% paraformaldehyde for 30 min, and then stained with crystal violet. Thirty minutes later, the plates were washed with PBS five times and clone formation was quantified.
Immunofluorescence analysis
Immunofluorescence analysis was used to detect the location of γH2AX foci. PC-3 cells were plated in 24-well plates (3 × 104 cells/well) and treated with CG-745 alone, radiation alone, or CG-745 in combination with radiation. After washing with PBS, the cells were fixed in 4% paraformaldehyde for 20 min. The fixed cells were permeabilized in 0.1% Triton X-100. The cells were then stained with a γH2AX primary antibody (Abcam, Waltham, Massachusetts, United States; 1:250) and with a secondary antibody (1:1000) at the next step. The nuclei were stained with 10 μg/mL 4′,6-diamino-2-phenylindole for 10 min at room temperature. Images were obtained using a fluorescence microscope (Zeiss, Oberkochen, Germany).
Cell cycle analyses
Cells were harvested by trypsinization and washed with PBS. After washing, the DNA was stained with propidium iodide (PI) solution containing RNase A (Multi Sciences, Hangzhou, China). The cell cycle distribution was monitored via flow cytometry (BD Biosciences, San Jose, California, United States).
Apoptosis assay
The apoptosis of PC-3 cells was assessed via flow cytometry analysis. PC-3 cells were collected and washed with PBS and then resuspended in binding buffer. Next, 5 μL Annexin V-FITC and 10 μL PI (Multi Sciences, Hangzhou, China) were added to the buffer and incubated for 5 min in the dark. Afterward, the samples were analyzed via flow cytometry (BD Biosciences, San Jose, California, United States).
Statistical analyses
Statistical analysis was performed using SPSS statistical software for Windows (SPSS, Chicago, Illinois, United States). The data are presented as the means ±SEMs. Two groups of comparisons were performed using Student’s t test for independent samples. P < 0.05 was considered significant.
Results
CG-745 enhances the repressive effect of irradiation on PC-3 cell growth
To detect the response of PC-3 cells to radiotherapy combined with CG-745, PC-3 cells were treated with CG-745 alone, radiation alone, or CG-745 in combination with radiation. The viability of PC-3 cells was measured using a CCK-8 assay, and the results indicated that cell viability was suppressed by CG-745 (79.97% ± 9.28%) or radiation alone (63.63% ± 9.88%), and radiation alone had a greater suppressive effect than CG-745 alone. The lowest cell viability was observed in the group treated with CG-745 in combination with radiation (32.23% ± 8.69%, P < 0.01, Figure 1(a)). Similar results were found in the colony formation assay. Compared with CG-745 (22.89% ± 1.39%) or radiation alone (15.33% ± 1.15%), CG-745 combined with radiation (10.00% ± 1.67%) also had a greater effect on suppressing cell proliferation (P < 0.01, Figure 1(b) and (c)).

CG-745 enhances the repressive effect of irradiation on PC-3 cells growth. (a) The viability of PC-3 cells was measured using a CCK-8 assay and (b, c) the cell proliferation of PC-3 cells was measured using a colony formation assay. *P < 0.05, **P < 0.01.
CG-745 enhances DNA damage in PC-3 cells after irradiation
To further explore the effect of CG-745 on irradiation-induced DNA damage, we used γH2AX foci formation as a surrogate DSB marker. Both CG-745 and radiation increased γH2AX foci formation in PC-3 cells after irradiation. In addition, significantly greater γH2AX foci formation was detected in the CG-745 combined with radiation group than in the CG-745 or radiation alone group (Figure 2).

Immunofluorescence analysis of γH2AX foci formation shows CG-745 enhances DNA damage in PC-3 cells after irradiation. DAPI: 4′,6-diamino-2-phenylindole; γH2AX: phosphorylated H2AX.
Cell cycle alterations in PC-3 cells
Flow cytometry analysis revealed that treatment with CG-745 or radiation alone increased the S cell content and decreased the G2 cell content in PC-3 cells, revealing that CG-745 or irradiation induced S phase cell cycle arrest. Compared with CG-745 or radiation alone, the combination of CG-745 and radiation significantly increased the percentage of cells in the S phase and decreased the percentage of cells in the G2 and G1 phases (P < 0.01, Figure 3).

Cell cycle alterations in PC-3 cells. (a) Cell cycle distribution was monitored via flow cytometry analysis. (b) Percentage of cells in the G1 phase. (c) Percentage of cells in the S phase and (d) percentage of cells in the G2 phase. *P < 0.05, **P < 0.01.
CG-745 promoted PC-3 cell apoptosis induced by radiation
Flow cytometry analysis revealed that PC-3 cell apoptosis was increased after treatment with CG-745 or radiation alone. The combination of CG-745 and radiation promoted PC-3 cell apoptosis more strongly than either CG-745 alone or radiation alone did (P < 0.01, Figure 4).
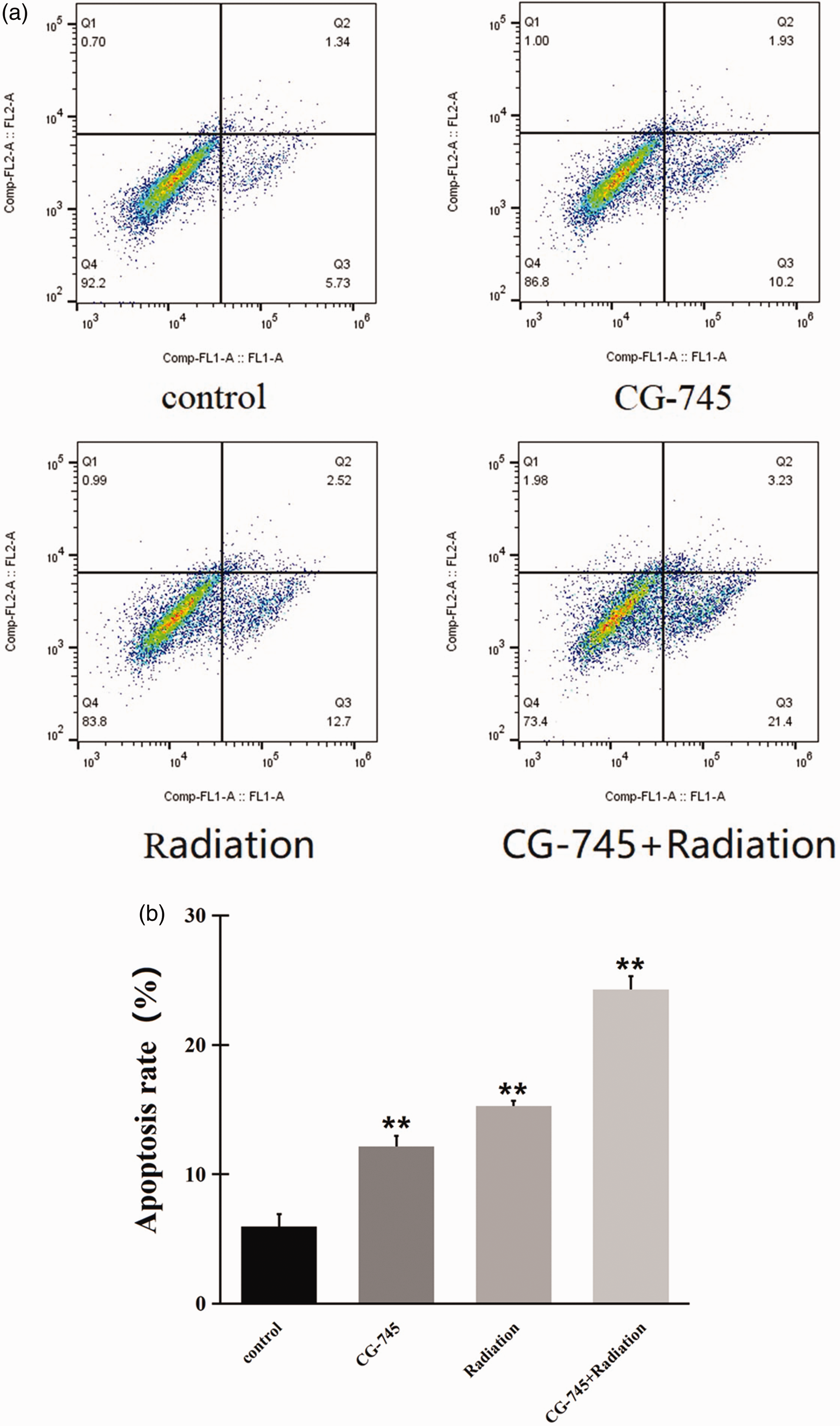
CG-745 promoted the PC-3 cell apoptosis induced by radiation. (a) Apoptosis of PC-3 cells was assessed via flow cytometry analysis and (b) the combination of CG-745 and radiation promoted PC-3 cell apoptosis more strongly than either CG-745 alone or radiation alone did. *P < 0.05, **P < 0.01.
Discussion
HDAC inhibitors have emerged as promising cancer therapeutic agents.19–22 CG-745, an HDAC inhibitor, has been reported to act against a variety of cancers in several studies.10,23–25 The present study investigated whether the use of CG-745 could increase the radiosensitivity of PC-3 cells to X-ray irradiation. The CCK-8 assay and colony formation assay revealed that CG-745 enhances the repressive effect of irradiation on PC-3 cell growth. These results suggested that CG-745 may have radiosensitizing effects on PC-3 cells.
Radiotherapy induces cell damage in cancer cells by causing DSBs. DNA DSBs are considered the most toxic type of DNA damage, causing cell death. 26 To investigate the DSBs in PC-3 cells after treatment with CG-745 and irradiation, γH2AX nuclear foci, the classical markers of DSBs, were detected via immunofluorescence analysis. Compared with CG-745 or radiation alone, CG-745 combined with radiation significantly increased γH2AX foci formation, indicating that CG-745 may increase the radiosensitivity of PC-3 cells through the DNA damage repair pathway.
Cell cycle arrest and apoptosis are typical impairments induced by radiotherapy.27,28 The cell cycle checkpoints, including G0/G1 phase checkpoints, S phase checkpoints, and G2/M phase checkpoints, are responsible for irradiation-induced DNA damage.29,30 Studies have shown that DNA-damaging agents often induce cell cycle arrest in the G1 or G2/M phases,31,32 and irradiation exposure can induce an S phase arrest. 33 In the present study, compared with CG-745 or radiation alone, the combination of CG-745 and radiation increased cell cycle arrest at the S phase and promoted apoptosis of PC-3 cells, indicating a synergistic effect on cell cycle distribution and apoptosis. These results demonstrate that the inhibition of S phase cell cycle arrest and the induction of apoptosis are critical mechanisms of the radiosensitization effect of CG-745 on PC-3 cells.
CG-745 has been reported to be a potent anticancer agent in many kinds of cancer. Combination therapy studies have proven that CG-745 can enhance the immune system’s anticancer effects and increase the anticancer efficacy of anti-PD-1 antibody. 10 In our study, CG-745 enhanced the effect of radiotherapy. These results indicate that CG-745 might have great potential in combination with other cancer therapies. Moreover, previous clinical studies have shown that HDAC inhibitors have limited effectiveness and unfavorable adverse effects. 34 However, in the initial human trial, CG-745 showed a favorable safety profile, and no dose-limiting toxicities were observed. 35
In summary, we confirmed that CG-745 enhanced radiation-induced DNA damage, cell cycle arrest, and cell apoptosis, thus increasing the radiosensitivity of PC-3 prostate cancer cells to X-ray irradiation. These findings indicate that the HDAC inhibitor CG-745 may be an effective radiosensitizer for cancer therapy. These results need validation in other cell lines and in vivo models. More experiments should be performed to clarify the complex mechanisms such as the DNA repair proteins and pathways involved. Further preclinical studies are needed before implementing clinical therapy for prostate cancer by using the combination of CG-745 and radiation in vivo.
Footnotes
Acknowledgments
None.
Author contributions
Wu and Liao established the study design. Wu performed the drug treatment and X-ray irradiation. Liao performed the CCK-8 assay, colony formation assay, immunofluorescence analysis, and flow cytometry analysis. Wu prepared the material for the figures. Liao conceived the study and wrote the main manuscript text. All authors reviewed the manuscript.
Consent for publication
All authors provided the consent for publication.
Data availability statement
The data used to support the findings of this study are available from the corresponding author upon request.
Declaration of conflicting interests
All authors declare no conflict of interest.
Ethical approval and consent to participate
Not applicable.
Funding
This study was partly supported by The Science and Technology Project of Taizhou City (XM20220596), The Traditional Chinese Medicine Project of Zhejiang Province (2022ZB396), and The Medical and Health Research Project of Zhejiang Province (2024KY1799).
