Abstract
Brucellosis is a zoonotic disease with significant global health implications, particularly in endemic regions such as Saudi Arabia. It is characterized by clinical manifestations that mimic both infectious and noninfectious diseases, making diagnosis challenging. Congenital brucellosis, a rare entity, can be transmitted transplacentally from a bacteremic mother or through exposure to maternal secretions during delivery. This report describes the case of a neonate with extremely low birth weight and severe respiratory distress syndrome, who was ultimately diagnosed with congenital brucellosis caused by Brucella melitensis. Despite treatment with trimethoprim–sulfamethoxazole (TMP/SMX) and rifampin, the infant developed abdominal distension and hypotension, showed a clinical picture consistent with sepsis, and succumbed on day 28 of life. This report underscores the importance of early recognition and management of congenital brucellosis to improve outcomes and highlights gaps in antenatal screening and preventive strategies. Diagnosis was confirmed by isolating B. melitensis from blood culture using standard microbiological methods. The patient was treated with trimethoprim–sulfamethoxazole (8 mg/kg/day) and rifampin (10 mg/kg/day). Clinical signs such as persistent hypoxemia and abdominal distension developed before death on day 28. Antenatal screening gaps in endemic regions such as Saudi Arabia contributed to delayed diagnosis.
Introduction
Brucellosis is a zoonotic disease caused by Brucella species, which include Brucella melitensis, B. abortus, B. suis, and B. canis. The disease poses significant public health challenges globally, particularly in endemic regions such as Saudi Arabia, where the incidence is approximately 18 per 100,000 population per year, as reported by the Ministry of Health in 2011. 1
Transmission of brucellosis to humans typically occurs through contact with infected animals or their secretions, ingestion of unpasteurized dairy products, or inhalation of aerosolized bacteria. Although human-to-human transmission is rare, cases involving blood transfusion, 2 bone marrow transplantation, 3 and breast milk 4 have been documented. Congenital brucellosis, although rare, occurs via vertical transmission during pregnancy and has variable outcomes depending on the timing of diagnosis and management. The disease is often overlooked due to its nonspecific and protean manifestations, leading to delays in diagnosis.
This report details a case of congenital brucellosis presenting at birth with severe respiratory distress. The case emphasizes the need for heightened clinical suspicion, particularly in endemic areas, and stresses the importance of comprehensive maternal history and antenatal care. The reporting of this study conforms to the Case Report (CARE) guidelines. 5
Case report
A male neonate, born at 22 weeks + 6 days of gestation, presented with severe respiratory distress syndrome immediately after birth. His 35-year-old mother, gravida 3, experienced premature rupture of membranes (PROM) at 22 weeks, with severe oligohydramnios and unknown, untreated brucellosis. Cesarean section was performed due to a history of cesarean delivery and maternal complications. The neonate’s APGAR (Appearance, Pulse, Grimace, Activity, and Respiration) scores were 5 and 7 at 1 and 5 min, respectively. Growth parameters were as follows: weight, 590 g; length, 29 cm; and head circumference, 21 cm.
The infant was intubated and placed on mechanical ventilation in the neonatal intensive care unit (NICU) of Alhada Armed Forces Hospital, Saudi Arabia. The father provided written informed consent for the initiation of any necessary medical treatment for his newborn during the NICU stay.
The infant received surfactant therapy (Survanta) and was started on dopamine and dobutamine infusions. Despite treatment, the clinical condition remained critical with persistent hypoxemia. Suspected early-onset sepsis prompted the initiation of empiric antibiotics (ampicillin and gentamicin) as per the NICU protocol.
Initial investigations revealed leukocytosis (white blood cell count of 30 × 103/μL), thrombocytopenia (platelet count of 141 × 103/μL), and elevated C-reactive protein (CRP) level at 8.5 mg/L. Chest radiography showed bilateral fine miliary infiltrates with normal lung volume. Blood and urine cultures were obtained, with the latter proving negative.
On the fifth day, gram-negative coccobacilli were isolated from blood culture and identified as B. melitensis (Figure 1). The patient was diagnosed with congenital brucellosis and started on trimethoprim–sulfamethoxazole (TMP/SMX) and rifampin. After treatment, blood cultures were negative. However, on day 21, the neonate developed abdominal distension and clinical signs of sepsis. Despite the addition of meropenem and vancomycin, the infant’s condition continued to deteriorate in the form of hemodynamic instability and abdominal distension. On day 28, the neonate suffered cardiac arrest and could not be resuscitated.

Gram-negative coccobacilli detected in blood culture and identified as Brucella melitensis.
B. melitensis was identified from blood culture on day 5 using Gram staining and confirmed via standard biochemical tests. Follow-up blood cultures were obtained on days 7 and 10, both of which were negative after antibiotic initiation. TMP/SMX (8 mg/kg/day) and rifampin (10 mg/kg/day) were administered. Meropenem (20 mg/kg every 8 h) and vancomycin (15 mg/kg every 12 h) were started on day 21. The patient received ventilatory support (mechanical ventilation), inotropes (dopamine and dobutamine), and parenteral nutrition.
Discussion
Brucellosis is caused by gram-negative, nonmotile, nonencapsulated coccobacilli. B. melitensis is the most pathogenic species for humans, frequently associated with severe diseases.6,7 Congenital brucellosis, although rare (Tables 11,7,8 and 29–16) is a recognized complication of maternal infection and poses diagnostic challenges due to its nonspecific clinical manifestations, which overlap with other neonatal infections.7,17
Previously reported cases of congenital brucellosis from Saudi Arabia.
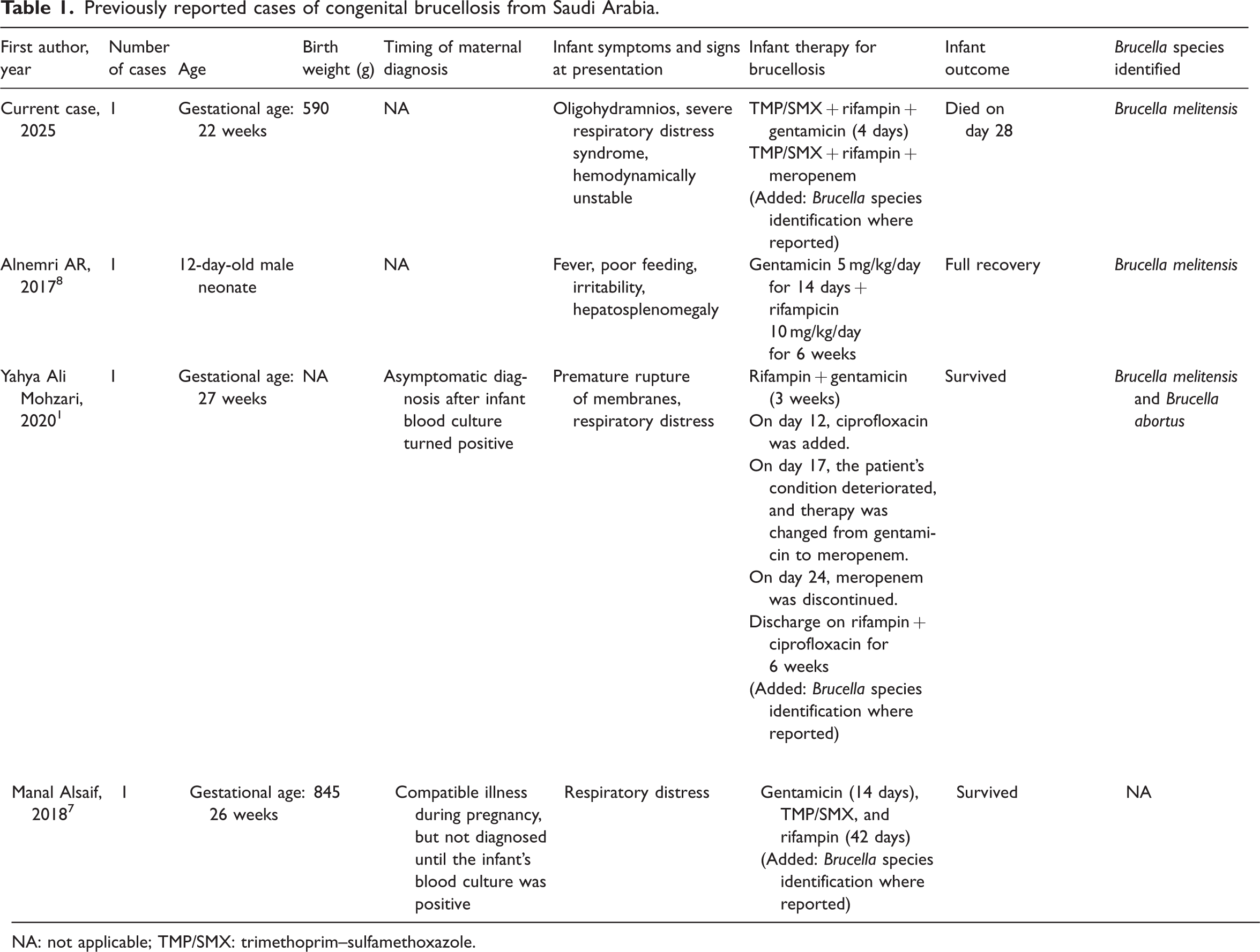
NA: not applicable; TMP/SMX: trimethoprim–sulfamethoxazole.
Previously reported cases of congenital brucellosis worldwide.

GA: gestational age; NA: not applicable; TMP/SMX: trimethoprim–sulfamethoxazole; CSF WBC: cerebrospinal fluid white blood cell.
The clinical spectrum of congenital brucellosis includes respiratory distress, sepsis, hepatosplenomegaly, hyperbilirubinemia, hypotension, and enterocolitis-like syndromes.7,17 Neonates may acquire the infection transplacentally or during delivery through exposure to maternal secretions.9,17 In the present case, the neonate exhibited severe respiratory distress as the initial and predominant symptom, which is uncommon and easily misdiagnosed as bacterial sepsis.
Saudi Arabia remains endemic for brucellosis despite public health efforts such as livestock vaccination and importation regulations.18,19 Cultural practices, including the consumption of raw camel milk, and resistance to culling infected animals contribute to the persistence of the disease. 18 Routine serological testing for brucellosis during pregnancy is not a standard practice in many endemic regions, leading to missed diagnoses and adverse perinatal outcomes. 15
Adverse pregnancy outcomes in brucellosis include spontaneous abortion, intrauterine fetal demise, PROM, and preterm delivery. 1 The pathophysiology is believed to involve placental infection despite the absence of erythritol, a sugar that facilitates Brucella growth in ruminant placentas.20,21 The immature immune system of preterm neonates, as seen in this case, further compromises the ability to elicit an adequate antibody response against Brucella.8,9
Thrombocytopenia in brucellosis is multifactorial, involving bone marrow suppression, hypersplenism, disseminated intravascular coagulation, and immune-mediated platelet destruction.6,8 In the current case, neonatal prematurity compounded the severity of hematological abnormalities. Furthermore, the rapid progression of symptoms despite targeted therapy highlights the challenges in managing congenital brucellosis in extremely preterm infants.
Prompt recognition of congenital brucellosis relies on a high index of suspicion, particularly in endemic areas. Definitive diagnosis is established by isolating Brucella species from blood cultures. 8 Early initiation of appropriate antimicrobial therapy is critical to improving outcomes. 1 TMP/SMX and rifampin remain the cornerstones of treatment, often supplemented with aminoglycosides or carbapenems in severe cases. 13 Multidisciplinary care involving neonatologists, infectious disease specialists, and microbiologists is crucial in managing such cases effectively.
Maternal brucellosis likely contributed to PROM and oligohydramnios through placental inflammation and reduced fetal perfusion. In endemic areas, a lack of routine serological screening during pregnancy contributes to undetected infections, increasing the risk of adverse neonatal outcomes. Routine maternal screening could have identified brucellosis and facilitated its treatment earlier, possibly preventing preterm delivery and infection transmission.
Conclusion
Congenital brucellosis, although rare, should be considered in neonates presenting with sepsis-like symptoms, particularly in endemic regions. Comprehensive maternal history, including dietary and occupational exposures, is essential for early diagnosis. This case highlights the importance of routine antenatal screening in endemic areas to prevent vertical transmission and associated morbidity and mortality. Greater awareness, improved diagnostic protocols, and preventive strategies are needed to mitigate the burden of congenital brucellosis.
Footnotes
Acknowledgements
We would like to thank our patient and his family for their kind participation in this work.
Authors’ contributions
All authors contributed substantially to writing the manuscript, reviewing the literature, the concept and design, acquisition and interpretation of data, drafting the article, and revising it critically for important intellectual content. All authors provided final approval of the version to be published.
Data availability
All data and materials related to the study are included in the current manuscript.
Declaration of conflicting interests
All authors declare no competing interests related to the study.
Funding
No funds were available for the current research.
