Abstract
Objectives
With advances in antiretroviral therapy, the life expectancy of individuals living with human immunodeficiency virus infection has significantly improved. Consequently, non–acquired immunodeficiency syndrome-related conditions, particularly chronic liver disease, have become increasingly prominent causes of mortality.
Methods
A retrospective analysis was conducted using mortality data from the Centers for Disease Control and Prevention Wide-Ranging Online Data for Epidemiologic Research database spanning 1999 to 2020. We identified deaths among adults aged 25 years and older in which both human immunodeficiency virus infection and chronic liver disease were listed as causes of death. Data were grouped by gender, age, race, and region. Age-adjusted mortality rates were calculated per 1 million adults in the United States. Annual percent changes were computed using Joinpoint regression.
Results
From 1999 to 2020, there were 13,447 deaths in the United States in which both human immunodeficiency virus infection and chronic liver disease were listed as causes of death, with an overall age-adjusted mortality rate of 2.49. Males had higher age-adjusted mortality rates (3.85) than females (1.08). Non-Hispanic Black population had a higher age-adjusted mortality rate (8.71) than Hispanics (3.3) and non-Hispanic whites (2.29).
Conclusion
Mortality in which both human immunodeficiency virus infection and chronic liver disease were listed as causes of death has declined, but racial disparities persist, particularly among non-Hispanic Black individuals. Addressing these disparities will require policies that improve care access and reduce liver disease-related risks among people living with human immunodeficiency virus infection.
Keywords
Introduction
With the increasing use of highly active antiretroviral therapy (HAART), mortality rates due to human immunodeficiency virus (HIV) infection have declined in recent years. However, individuals living with HIV infection are now at increased risk of mortality due to non–HIV-related comorbidities, including non–acquired immunodeficiency syndrome (AIDS)-associated malignancies, cardiovascular disease, liver disease, substance use, and suicide. In particular, chronic liver disease (CLD) accounts for 13% to 18% of deaths among individuals with HIV infection.1,2 Contributing factors to CLD in this population include liver damage directly associated with HIV infection, antiretroviral treatment toxicity, co-infection with viral hepatitis, alcoholism, and metabolic dysfunction-associated liver disease (MASLD). Patients co-infected with HIV and hepatitis B virus (HBV) or hepatitis C virus (HCV) are more likely to develop liver fibrosis and cirrhosis. These conditions lead to an increased risk of progression to hepatocellular carcinoma (HCC). 3 Recent data suggest that 30% of HIV infection patients are co-infected with HCV, with 40%–69% experiencing steatosis due to MASLD in this subpopulation. 2
While many of these non–HIV-related comorbidities may arise from chronic inflammation and HIV itself, there is also likely an additional contribution from health conditions and social determinants that are not directly linked to HIV status. 4 Identifying the demographic and regional distribution of CLD-related mortality in HIV cases can help identify populations at the highest risk so that timely targeted interventions can be provided. In this study, we investigated the trends in mortality due to HIV infection and liver disease in the United States (US) from 1999 to 2020, stratifying data by year, sex, race/ethnicity, urbanization status, and geographic region.
Methods
This study adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 5
Data regarding deaths that took place in the US related to HIV infection and CLD were extracted from the Centers for Disease Control and Prevention Wide-Ranging Online Data for Epidemiologic Research (CDC WONDER) database. 3 The Multiple Cause-of-Death Public Use record death certificates were studied to identify records in which both HIV infection and CLD were mentioned as either contributing or underlying causes of death on nationwide death certificates. This database has previously been used to determine trends in mortality of HIV infection and CLD in patients aged ≥15 years.6 –8 HIV infection patients were identified as per the International Classification of Diseases 10th Revision Clinical Modification (ICD-10-CM) codes B20-B24, and for the purpose of this study, CLD included HCV infection (B17.1, B18.2), HBV infection (B16, B17.0, B18.0, B18.1), ALD (K70), and MASLD (K75.8, K76.0). This study was exempt from local Institutional Review Board approval because the CDC WONDER database contains publicly available, anonymized data.
Co-reported HIV infection and CLD deaths, population sizes, and location of death (including medical facilities (outpatient, emergency room, inpatient, death on arrival, or status unknown), home, hospice, and nursing home/long-term care facility) were extracted for each calendar year from 1999 to 2020. Demographic characteristics (sex, race/ethnicity, age) and regional information (urban-rural and state) were extracted from 1999 to 2020. Race/ethnicities were defined as non-Hispanic (NH) White, NH Black or African American, Hispanic or Latino, NH American Indian or Alaskan Native, and NH Asian or Pacific Islander patients. These race/ethnicity categories have been previously used in analyses from the CDC WONDER database and rely on data reported on death certificates. For urban-rural classifications, the 2013 National Center for Health Statistics Urban-Rural Classification Scheme was used to divide the counties into urban (large metropolitan area; population ≥1 million), medium/small metropolitan area (population 50,000–999,999), and rural (population <50,000) categories. 9 Regions were classified into the following four categories: northeast, midwest, south, and west, according to the Census Bureau's definitions.
Crude and age-adjusted mortality rates (AAMRs) per 1,000,000 population were determined. AAMRs were calculated for each calendar year from 1999 to 2020, and the overall AAMR for the study period was determined by averaging these yearly rates. All the AAMRs are reported with their corresponding 95% confidence intervals (CIs). Crude mortality rates were determined by dividing the number of HIV- and CLD-related deaths by the corresponding US population in that year. AAMRs were calculated by standardizing HIV- and CLD-related deaths to the year 2000 in the US population. 10 Due to limitations of the CDC WONDER database, population-level denominators for people living with HIV infection were not available. Therefore, all AAMRs in this study are reported per 1 million US adults and do not reflect within-population rates among individuals with HIV infection. The Joinpoint Regression Program (Joinpoint V 4.9.0.0, National Cancer Institute) was used to determine trends in AAMRs using annual percent change with 95% CI. 11 This software identifies significant variations in AAMRs over time by fitting log-linear regression models where temporal shifts occurred. Annual percent changes (APCs) were considered to increase or decrease if the slope describing the change in mortality significantly differed from zero using 2‐tailed t-testing. Statistical significance was set at P < 0.05.
Results
Between 1999 and 2020, a total of 13,447 deaths occurred due to HIV infection and CLD in the US. A large majority (62%) of deaths occurred in inpatient medical facilities, while 14.9% occurred at the deceased’ homes (Supplementary Table S1). Deaths at nursing homes and hospices accounted for 7.9% and 6% of fatalities, respectively. Further, 0.3% of patients were dead on arrival at a medical facility. The place of death was unknown for 0.37% of patients.
Annual mortality trends
The AAMR declined significantly from 4.91 (95% CI: 4.62–5.21) in 1999 to 1.72 (95% CI: 1.57–1.87) in 2020 (Supplementary Table S2, Figure 1). The decrease in AAMR was particularly steep from 1999 to 2001 (APC: −21.3, 95% CI: −30.1 to 3.2) but decelerated thereafter until the end of the study period (APC: −1.8, 95% CI: −19.3 to 8.8).

Overall and sex-stratified, age-adjusted mortality rates per 1,000,000 for deaths with both HIV infection and CLD listed in adults in the United States, 1999–2020. CLD: chronic liver disease; HIV: human immunodeficiency virus.
CLD deaths among individuals with HIV infection and CLD
A sensitivity analysis examining CLD as the underlying cause of death with HIV infection as a contributing cause identified 1453 deaths from 1999 to 2020 (overall AAMR: 0.27, 95% CI: 0.26–0.29) (Supplementary Figure 1). The AAMR for CLD deaths among individuals with HIV infection approximately halved from 0.42 (95% CI: 0.34–0.51) in 1999 to 0.22 (95% CI: 0.17–0.29) in 2003 (APC: −13.58, 95% CI: −22.66 to −3.43), demonstrating a fluctuating nonsignificant trend thereafter until 2020 (APC: 0.47, 95% CI: −0.85 to 1.81).
HIV-related deaths among individuals with HIV infection and CLD
Conversely, when HIV infection was the underlying cause and CLD a contributing cause, 9818 deaths were identified (overall AAMR: 1.79, 95% CI: 1.76–1.83) (Supplementary Figure 1). The AAMR for HIV-related deaths in individuals with concomitant CLD showed a steep, consistent decline from (4.09, 95% CI: 3.82–4.36) in 1999 to (0.86, 95% CI: 0.75–0.97) in 2020 (APC: −5.36, 95% CI: −6.50 to −4.21).
Mortality trends stratified by gender
Males had consistently higher AAMR than females throughout the study period (overall AAMR males: 3.85, 95% CI: 3.78–3.93; females: 1.08, 95% CI: 1.04–1.12). (Supplementary Table S3, Figure 1). Among females, the AAMR decreased from 1.98 (95% CI: 1.72–2.24) in 1999 to 0.79 (95% CI: 0.64–0.94) in 2020. The AAMR declined sharply from 1999 to 2001 (APC: −20.4, 95% CI: −30.7 to −1.6), after which it continued to fall at a slower rate (APC: −1.8, 95% CI: −19.3 to 8.8). Males exhibited a parallel trend. The AAMR decreased from 7.94 (95% CI: 7.41–8.48) in 1999 to 2.73 (95% CI: 2.46–3.00) in 2020. The AAMR decreased sharply from 1999 to 2001 (APC: −21.6, 95% CI: −29.7 to −4.9) followed by a relatively gradual decline until 2020 (APC: −2.7, 95% CI: −3.8 to 0.4).
Mortality trends stratified by race
Throughout the study duration, the highest AAMRs were observed among the NH Black or African American population, followed by that in Hispanics and NH Whites (overall AAMR NH Black: 8.71, 95% CI: 8.48–8.94; Hispanic: 3.30, 95% CI: 3.17–3.44; NH White: 1.35, 95% CI: 1.32–1.39). The temporal trend in AAMR was similar across the three groups, with a sharp dip in AAMR between 1999 and 2001, followed by a slower decline until 2020 (Supplementary Table S4, Figure 2).
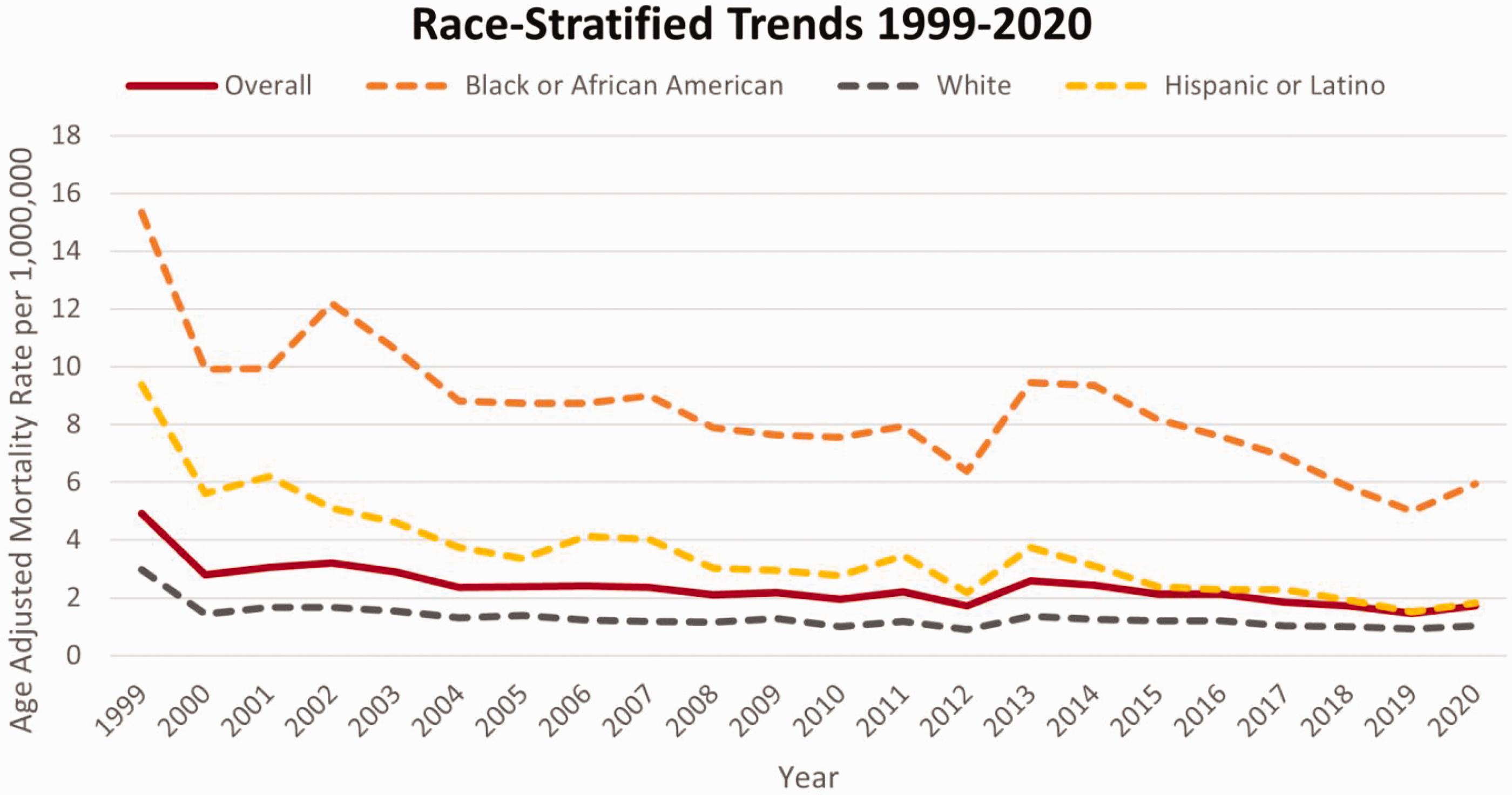
AAMRs per 1,000,000 for deaths with both HIV infection and CLD listed, stratified by race in adults in the United States, 1999–2020. AAMR: age-adjusted mortality rate; CLD: chronic liver disease; HIV: human immunodeficiency virus.
Mortality trends stratified by urbanization
Patients in metropolitan areas exhibited consistently higher AAMRs throughout the study period (overall AAMR metropolitan: 2.66, 95% CI: 2.61–2.71; non-metropolitan: 1.20, 95% CI: 1.12–1.28). In metropolitan areas, the AAMR decreased from 5.55 (95% CI: 5.20–5.89) in 1999 to 1.81 (95% CI: 1.64–1.98) in 2020. A particularly sharp decrease in AAMR was observed from 1999 to 2001 (APC: −22.2, 95% CI: −30.6 to −3.7), followed by a slower decline until 2020 (APC: −2.7, 95% CI: −4.4 to 2.8). In comparison, the AAMR in non-metropolitan areas remained relatively stable throughout the study period (AAPC: −0.8, 95% CI: −2.5 to 1.1) (Supplementary Table S5, Figure 3).

AAMRs per 1,000,000 for deaths with both HIV infection and CLD listed, stratified by urban–rural status in adults in the United States, 1999–2020. AAMR: age-adjusted mortality rate; CLD: chronic liver disease; HIV: human immunodeficiency virus.
Mortality trends stratified by state and census region
The northeast region experienced the highest AAMRs (overall AAMR: 3.93, 95% CI: 3.81–4.06), followed by the south region (overall AAMR: 2.53, 95% CI: 2.46–2.60), west region (overall AAMR: 2.31, 95% CI: 2.23–2.40), and midwest region (overall AAMR: 1.07, 95% CI: 1.01–1.13) (Supplementary Table S6, Figure 4).
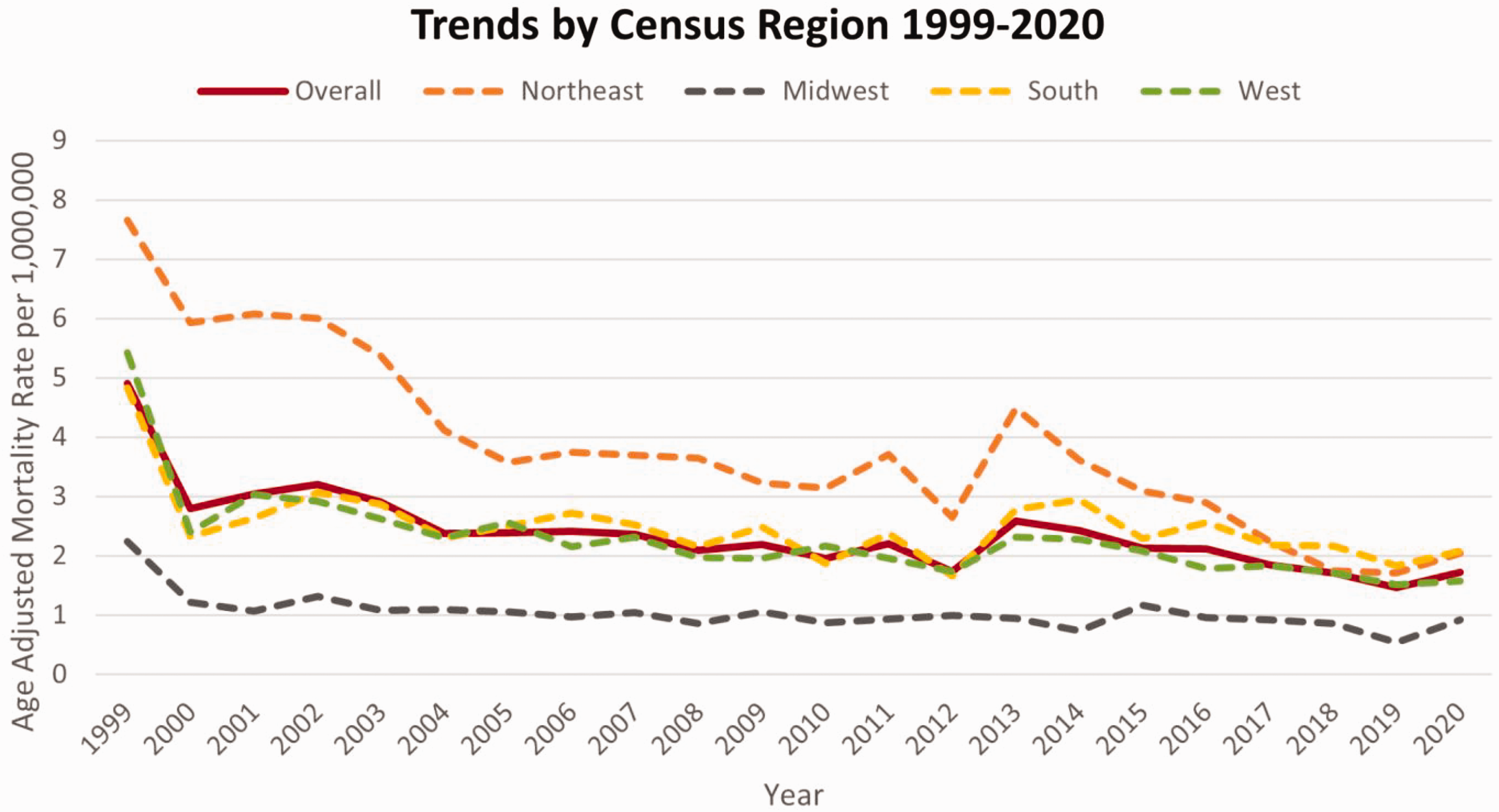
AAMRs per 1,000,000 for deaths with both HIV infection and CLD listed, stratified by the United States Census Region, 1999–2020. AAMR: age-adjusted mortality rate; CLD: chronic liver disease; HIV: human immunodeficiency virus.
The AAMR varied considerably between states, ranging from 16.62 (95% CI: 14.16–19.08) in the District of Columbia to 0.64 (95% CI: 0.44–0.89) in Iowa (Supplementary Table S7, Figure 5). States in the top 90th percentile included the District of Columbia, Maryland, New York, Rhode Island, and Connecticut, while those in the lower 10th percentile included Iowa, Wisconsin, Maine, Idaho, and Hawaii.

AAMRs per 1,000,000 for deaths with both HIV infection and CLD listed, stratified by state in the United States, 1999–2020. AAMR: age-adjusted mortality rate; CLD: chronic liver disease; HIV: human immunodeficiency virus.
Discussion
Our findings indicate a significant decline in mortality where both HIV infection and CLD were listed as causes of death, from 1999 to 2000, with notable geographic and demographic disparities. The sharp initial decline from 1999 to 2001, followed by a slower reduction in mortality rates, likely reflects the widespread uptake of HAART, notably the transition to effective triple-drug regimens. 12 The widespread adoption of HAART in the late 1990 s significantly improved survival rates for individuals with HIV infection by reducing the occurrence of opportunistic infections and coexisting conditions, such as liver disease.13,14
Gender-based disparities
Although both males and females demonstrated declining AAMRs over time, males consistently exhibited higher rates. This disparity may be partly attributed to higher rates of HCV co-infection and lifestyle-related risk factors among men.15,16 HCV/HIV co-infection accelerates liver fibrosis, potentially increasing the risk of liver-related complications. Interestingly, prior studies have not consistently found significant sex-based differences in liver-related mortality, suggesting that additional factors may contribute to the observed disparities in AAMRs between males and females.17,18 For instance, in a cohort of prospectively monitored hemophilia patients, Goedert et al. 19 reported a 16-year cumulative incidence of end-stage liver disease (ESLD) of 14% in men with HIV infection compared to 2.6% in those without HIV infection.
Substance use may also contribute to the elevated mortality burden observed among individuals with both HIV infection and CLD. Injection drug use, in particular, is a well-established risk factor for HCV co-infection, which significantly accelerates liver disease progression in people living with HIV infection.20 –22 Excessive alcohol consumption can further exacerbate liver injury in the context of viral hepatitis and HIV co-infection.23,24 These factors may partly underlie the differences in AAMRs stratified by gender. While studies have shown that women with HIV infection are more likely than men to undergo testing, they may face barriers to consistent HAART adherence due to stigma, negative healthcare experiences, limited partner support, and long clinic wait times. 25 Conversely, men with HIV infection, especially those with a history of substance use, may experience delayed linkage to care and reduced healthcare engagement, often driven by stigma and perceived discrimination.26,27
Racial disparities
NH Black individuals had the highest AAMRs and the slowest rate of decline than NH White and Hispanic individuals, despite an overall decrease in mortality. These disparities reflect persistent structural barriers, including socioeconomic inequalities, limited access to healthcare, and medical mistrust. 28 NH Black individuals face significant challenges, including lower rates of viral suppression, reduced likelihood of receiving timely HAART and direct-acting antiviral medication, and systemic racism within healthcare systems, all of which contribute to increased liver-related mortality.29 –31
Additionally, Black communities are disproportionately affected by social determinants of health that worsen health outcomes, including food insecurity, housing instability, and lower income levels. Metabolic comorbidities such as obesity and diabetes further exacerbate liver disease progression in these populations. 32
Interpreting mortality disparities across gender and racial groups is inherently complex, as multiple social, structural, and behavioral factors may exert opposing effects. For example, poorer healthcare access and lower testing rates among NH Black individuals and women could underestimate HIV infection or CLD as causes of death as reported on death certificates, potentially leading to underestimation of the true mortality burdens in these groups. Conversely, delayed diagnosis and limited access to timely or effective treatment may also contribute to worse outcomes. Among women, reduced HAART adherence due to stigma or systemic barriers could increase mortality risk, while among men, lower healthcare engagement might result in poorer outcomes. These competing and intersecting factors complicate the direct interpretation of trends, underscoring the need for individual-level studies that can more precisely account for testing, treatment uptake, and access to care.
Regional disparities
Regional disparities in mortality rates revealed that the population in the northeast region had the highest AAMRs, while that in the midwest region had the lowest. These variations may be linked to differences in HIV prevalence, healthcare infrastructure, and the availability of specialized liver disease care. 33 The District of Columbia, which reported the highest AAMR, has struggled with high HIV prevalence for an extended period, with 1.7% of the population currently diagnosed with HIV infection. 34 Limited access to healthcare, particularly among low-income and Black populations, contributes to the region's high mortality rate.
Higher rates of co-infection and HIV prevalence in urban areas, potentially linked to increased substance abuse, poverty, and crime rates, may explain why mortality rates in urban areas are nearly double than those in rural areas. 35 However, underreporting or delayed diagnosis due to disparities in healthcare access in rural regions may also impact these findings. 36
Impact of risk factors
Several factors contribute to the progression of liver disease in individuals living with HIV infection, including alcohol consumption, metabolic disorders, and viral co-infections. Studies indicate that excessive alcohol intake accelerates liver fibrosis, making it a significant contributor to liver disease. 37
MASLD has become an emerging concern in people with HIV infection, particularly those receiving HAART, driven by treatment-related metabolic disturbances.38,39 Metabolic disorders such as obesity and diabetes, conditions that have become increasingly prevalent among people living with HIV infection in recent decades, also significantly contributed to liver disease progression by promoting fibrosis and worsening MASLD outcomes. 40 Obesity is becoming increasingly prevalent in individuals with HIV infection and exacerbates liver damage by promoting liver steatosis, while diabetes, often comorbid with HIV infection, leads to insulin resistance and inflammation. 41
Individuals with HIV infection are also at a higher risk of developing complications from chronic viral hepatitis, particularly ESLD and HCC although their epidemiology has also evolved with improvements in screening and treatment. Factors associated with liver fibrosis in these individuals include higher CD4+ counts, elevated serum alanine aminotransferase (ALT) levels, HCV genotype 3 infection, alcohol abuse, older age, and liver steatosis.42 –45
Finally, long-term HAART use, especially beyond 6 years, increases the risk of advanced liver fibrosis due to drug toxicity. Certain HAART drugs, such as protease inhibitors and nucleoside analogs, can cause metabolic issues, including insulin resistance, dyslipidemia, and mitochondrial damage. These effects contribute to hepatic steatosis and steatohepatitis, which may exacerbate pre-existing liver conditions in individuals with chronic hepatitis. 46 These changes in the prevalence of risk factors may help explain the observed trends in AAMRs among individuals with HIV infection and CLD.
HIV-related deaths have declined significantly due to the introduction of HAART. According to the World Health Organization, over 600,000 people die due to HIV-related causes, primarily because they are unaware of their condition, are not undergoing treatment, or start treatment too late. 37 Moreover, a study from South Africa presented at the 12th International AIDS Society Conference on HIV Science (IAS 2023) showed that individuals who interrupted HAART therapy for more than 6 months and later resumed it were 2 to 3 times more likely to die than those who were consistent with their treatment. 38 Furthermore, research from the UK revealed that 87.1% of AIDS-related deaths occurred in individuals diagnosed late, with 28.5% never being linked to HIV outpatient care. 39
Limitations
This study has some limitations. A key limitation of this study is the inability to calculate liver disease mortality rates specifically within the population of individuals living with HIV infection because as the CDC WONDER database does not provide HIV-specific population denominators. As a result, the observed declines in mortality where both HIV infection and liver disease are listed may reflect changes in the size or demographic composition of the broader HIV population rather than a definitive reduction in liver disease burden among those with HIV infection. These findings should therefore be interpreted as population-level trends rather than within-group estimates. Additionally, mortality data may be subject to misclassification and underreporting, particularly among populations with limited access to healthcare. 47
Conclusion
These findings highlight the need for targeted policies to reduce racial and geographic disparities in HIV- and CLD-related mortality. Key interventions include expanding access to direct-acting antivirals for HCV treatment, improving healthcare infrastructure in high-impact areas, and addressing social determinants of health. Community-based therapies that focus on reducing stigma, improving healthcare trust, and increasing treatment adherence among Black individuals are particularly important. Additionally, interventions aimed at reducing alcohol use and managing metabolic comorbidities may help lower mortality rates. Future studies that leverage surveillance cohorts or registries with known HIV denominators are warranted to more precisely evaluate liver-related mortality in this population. Research is also needed to explore the long-term toxicities of HAART and the growing burden of MASLD in individuals with HIV infection.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251363782 - Supplemental material for Trends and disparities in human immunodeficiency virus and chronic liver disease mortality in the United States: A nationwide analysis
Supplemental material, sj-pdf-1-imr-10.1177_03000605251363782 for Trends and disparities in human immunodeficiency virus and chronic liver disease mortality in the United States: A nationwide analysis by Fariha Hasan, Mariam Shahabi, Amna Kaleem Ahmed, Ayesha Arshad, Husnain Ahmad, Zubia Zaman, Oasima Muner, Tommy Nguyen, Hassam Ali, Dushyant S Dahiya and Shahid M Malik in Journal of International Medical Research
Footnotes
Acknowledgements
None.
Author contributions
Conception and design: FH, MS, AKA
Analysis and interpretation of the data: FH, MS, AKA, AA, HA, ZZ, OM, TN, HA, DSD, SMM
Drafting of the article: FH, MS, AKA, HA, AA, ZZ
Critical revision of the article: FH, MS, HA, DSD, TN, SMM
Consent to participate
Not applicable.
Consent for publication
Not applicable. This study does not include any individual data requiring consent for publication.
Data availability statement
Data analyzed in this study were obtained from publicly available sources.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical considerations
This study was exempt from local Institutional Review Board approval as the CDC WONDER database contains publicly available, anonymized data.
Funding
This study has not received funding from any source.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
