Abstract
Herein, we present a rare and complex clinical case of a patient with urinary tract infection leading to ground Escherichia coli sepsis, severe diabetic ketoacidosis, and severe rhabdomyolysis. The patient was admitted to the hospital with fever, abnormal mental behavior, disturbances in the internal environment, and acute kidney injury and was thoroughly evaluated by physicians to determine the underlying causes of severe rhabdomyolysis, including Escherichia coli infection and metabolic disturbances. After treatment with rehydration resuscitation, anti-infective agents, and hemoadsorption combined with continuous venovenous hemodialyfiltration, the patient’s condition improved, renal function was restored, and hemodialysis treatment was discontinued.
Introduction
Acute kidney injury (AKI) is the most common complication of rhabdomyolysis (RM). 1 Statistically, AKI occurs in approximately 13%–50% of patients with RM; of whom, 26% require renal replacement therapy (RRT).1–3 The etiology of RM can be classified into genetic or acquired causes; genetic causes include lipid or carbohydrate metabolic diseases, while acquired causes are further divided into traumatic causes, including crush injuries, electrical injuries, compression, and vascular or orthopedic surgeries, and nontraumatic causes, including vigorous exercises, seizures, exposure to extreme heat, malignant hyperthermia and neuroleptic malignant syndrome, alcohol, infections, electrolyte imbalance, and drugs/toxins. 4 However, patients with RM and AKI caused by diabetic ketoacidosis (DKA) and Escherichia coli infection are rare, and the mortality rate of such patients is quite high. 5 Therefore, early diagnosis and effective treatment are important. The current report conforms to the Case Report (CARE) guidelines 6 ; we successfully treated a middle-aged woman with RM and AKI caused by E. coli infection and severe DKA using the Jafron HA380 device (Jafron Biomedical Co., Ltd., Zhuhai City, China) combined with continuous venovenous hemodialyfiltration (CVVHDF).
Case report
The patient was a female in her early 50s who presented with persistent fever and painful urination for more than 10 days. She was eventually hospitalized for delirium at the West China Hospital of Sichuan University (Chengdu, China) in October 2024 and was promptly admitted to the intensive care unit for treatment. Furthermore, she received endotracheal intubation, respiratory support, and metaraminol bitartrate for hemodynamic maintenance. She had a history of schizophrenia and diabetes.
On physical examination at the time of admission, she had a heart rate of 130 beats per minute and blood pressure level of 115/79 mmHg. She exhibited confusion with inappropriate questions and answers. Routine laboratory results were obtained at the time of presentation (Table 1). Computed tomography suggested a lung infection with atelectasis of the lower lobes of both lungs.
Abnormal laboratory data at admission.

WBC: white blood cell; CK: creatine kinase; Mb: myoglobin.
On admission, she was initiated on active fluid resuscitation and intravenous piperacillin sodium and tazobactam sodium (4.5 g every 8 h for 7 days) to control the infection, while insulin was continuously infused until the patient’s anion gap returned to normal and the DKA resolved. On the first day, 4.8 L of fluid was infused nasally and 3.5 L intravenously; however, the patient’s creatinine level increased from 227 to 267 µmol/L. At 24 h after admission, we initiated CVVHDF therapy through the Prismaflex CRRT System® (Baxter, Deerfield, IL, USA) with an AN69ST-150 filter (Gambro Renal Products, Lakewood, CO, USA). The rate of blood flow was 150 mL/min. Postdilution was performed using a bicarbonate replacement solution (Chengdu Qingshan Likang Pharmaceutical Co., Ltd., Chengdu, China) at a rate of 1000 mL/h for each replacement solution and dialysis solution. Anticoagulation was performed using citrate combined with low-molecular heparin. The blood filter was changed every 72 h unless coagulation occurred. After the above aggressive treatment, the patient’s hypernatremia gradually improved on day 3 of hospitalization, and the serum sodium level returned to normal.
However, the patient’s creatine kinase (CK) and myoglobin (Mb) levels were elevated. Therefore, on day 5 of hospitalization, we started adding HA380 therapy to CRRT for 10 h, which reduced the CK level from 42,762 to 37,528 U/L and Mb level from 45,640 to 28,745 ng/mL (Figure 1). Continuation of a second HA380 treatment in tandem with CRRT for 10 h on day 6 of hospitalization resulted in a decrease in the CK levels from 17,863 to 15,125 U/L and Mb levels from 26,450 to 12,094 ng/mL (Figure 1). On day 7 of hospitalization, she had no further fever, and metaraminol bitartrate dosage was reduced from 4 µg/kg/min on admission to 0.25 µg/kg/min (Table 2). Unfortunately, we could not observe the overall trend in the Mb levels because the upper limit of Mb measurement in our hospital was 3000 ng/mL. To assess the patient’s condition and treatment effectiveness, the samples were deliberately diluted before measuring the absolute value of Mb levels during the use of hemoadsorption (HA).
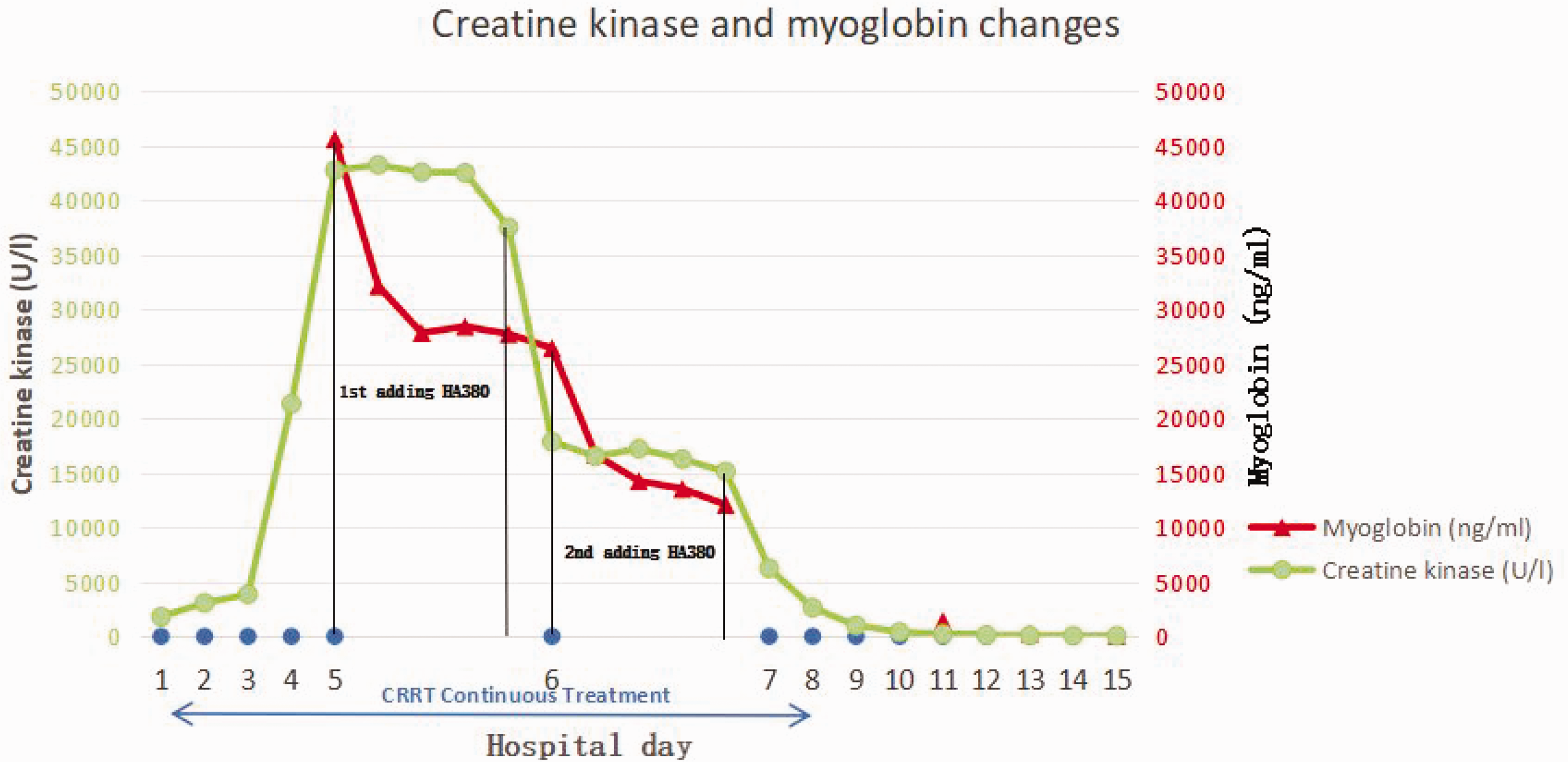
Decrease in Mb and CK levels at 10 h of HA380 therapy. CK: creatine kinase; Mb: myoglobin.
Changes in the patient’s condition during hospitalization.

RRT: renal replacement therapy; APACHE II: Acute Physiology and Chronic Health Evaluation II; SOFA: Sequential Organ Failure Assessment; MAP: mean arterial blood pressure; CK: creatine kinase; Mb: myoglobin; CVVHDF: combined with continuous venovenous hemodialyfiltration.
The patient’s condition subsequently improved, and she was transferred out of the intensive care unit on day 15 of hospitalization and discharged on day 26 of hospitalization. At the time of discharge, the patient exhibited normothermia, answered questions clearly, and was completely off hemodialysis treatment (creatinine level: 94 μmol/L, urine output: 3000 mL/d).
Discussion
Although RM has a diverse etiology, E. coli infection and DKA-associated RM are exceptionally rare. According to the literature, the mechanisms underlying infection-induced RM include tissue hypoxia, direct bacterial invasion of muscles, low oxidative and glycolytic enzyme activity, activation of lysosomal enzymes, and mechanisms involving endotoxins. 7 Many case reports have linked hypernatremia and severe dehydration to RM with a hyperosmolar state secondary to diabetes as the underlying mechanism of DKA.8,9 Denman reported that hypernatremia alone is a potential cause of RM. 10
Mb is the main causative agent of AKI in patients with RM.11,12 Mb excretion is reduced when AKI occurs, and it further exacerbates renal injury through indirect and direct mechanisms. 13 Therefore, it is important to promptly and effectively reduce Mb levels in the circulation. In our case, HA380 combined with CVVHDF was effective in eliminating Mb (Figure 1). According to previous studies, the screening coefficient for Mb via CVVHDF with AN69ST membrane ultrafiltration at a rate of 1 L/h was 0.21. 14 There are relatively few studies on the efficiency of HA380 on Mb clearance. However, according to a survey of CytoSorb, the median MB plasma clearance was 80%, 40%, 20%, 15%, and 12% at 10 min and 1, 3, 6, and 12 h, respectively, following the start of treatment with CytoSorb. 11 CytoSorb has a loading volume of 300 mL and an effective membrane area of approximately 40,000 m2, whereas HA380 has a loading volume of 380 ± 3 mL, with an effective membrane area of approximately 54,000–64,000 m2. 15 We suspect that HA380 is more efficient at eliminating Mb than CytoSorb, but studies comparing the two devices based on the clearance effect of Mb are lacking. In our case, the serum Mb levels decreased obviously during HA treatment with HA380; however, a large sample study is warranted for further confirmation.
The adsorption profile of HA380 was 5–60 KDa 15 ; however, CK, with a molecular weight of 82 kDa, also decreased during the use of HA380 in this patient (Figure 1). Therefore, a higher adsorption profile of HA380 than previously assumed should be considered and verified, with particular attention to side effects, such as albumin and platelet depletion. We found that two 10h HA380 treatments decreased the patient’s platelet count from 121 × 109/L to 102 × 109/L (Table 2); however, little change was observed in the albumin levels, which was 27.4 g/L at the beginning of the first HA380 treatment and 27.2 g/L at the end of the second HA380 treatment (Table 2). However, the study of HA380 for sepsis revealed that no other adverse events such as thrombocytopenia and albumin reduction were observed. 16
He et al. 16 demonstrated that HA380 significantly reduced interleukin (IL)-6, IL-8, and IL-10 levels, thus considerably reducing the need for vasopressor medications. Although there was no cytokine trend in this patient, we found that with aggressive anti-infective agents, the patient’s vasopressor drug dosage was 1 µg/kg/min before starting the first HA380 treatment and 0.25 µg/kg/min at the end of the second HA380 treatment (Table 2). After HA380 (day 7 of hospitalization), the patient stopped having a fever. However, an animal study demonstrated that 4 h of HA380 hemosorbent therapy during sepsis resulted in an approximately 25%–35% decrease in vancomycin and gentamicin levels in patients. 17 Therefore, HA380 is considered a suitable treatment option for infection-induced RM; however, the timing of initiating HA380 therapy and how often to change the adsorbent device for maximizing patient benefit remain unclear.
The European national consensus, which states that initiation of HA therapy should be considered when Mb levels are elevated to >10,000 ng/mL, regardless of comorbid AKI (class I recommendation), recommends that adsorption devices should be changed every 8–12 h (class II recommendation) until Mb concentrations are <5000 ng/mL (class III recommendation). 18 However, sufficient evidence is lacking. Our patient started HA much later and stopped it much earlier; however, the patient’s renal function fully recovered after approximately 1 month. Therefore, in patients with RM, in addition to the Mb level, the time of initiation or cessation of HA should be assessed by considering the patient’s urine output, degree of control of the primary disease, and other factors. A randomized controlled trial is warranted to further confirm the occurrence of RM during HA treatment initiation and discontinuation.
Finally, there are certain limitations of this case. First, due to the limitations of the myoglobin test on line in our hospital, we could not measure the exact value of the patient’s myoglobin at the time of admission. Second, the article included a single case; thus, further studies with larger sample sizes are warranted to validate our findings.
Conclusion
We successfully treated a patient with RM and AKI caused by E. coli infection and DKA. By actively and effectively treating the primary disease, CVVHDF plus HA not only lowered the patient’s Mb and CK levels but also reduced the patient’s vasopressor drug dosage, and the patient eventually exhibited complete renal function recovery.
Footnotes
Acknowledgements
The authors sincerely thank the patient and her family for their willingness to allow us to share the case.
Author contributions
X.C.Z. and X.L. designed the work; X.C.Z. and X.L. collected and analyzed the data; X.C.Z., X.L., Y.Y.Y., F.W., L.Z., and P.F. drafted the work or substantively revised it; and all authors read and approved the final manuscript.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Declaration of conflicting interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
Ethics statement
Consent was obtained from the patient’s relatives for the treatment regimen and publication of the case report. The study was conducted according to the principles of the Declaration of Helsinki (version 2013, October; ![]() ) following laws and medical research involving humans (WMO) and the requirements of China law regarding human-based research.
) following laws and medical research involving humans (WMO) and the requirements of China law regarding human-based research.
Funding
The study was funded by the 1.3.5 project for disciplines of excellence from the West China Hospital of Sichuan University (ZYGD23015).
