Abstract
Transient osteoporosis of the hip is a rare, temporary condition characterized by hip pain and functional limitations, which can significantly reduce the quality of life. Bone marrow edema of the proximal femur, identified through magnetic resonance imaging is the primary diagnostic feature of transient osteoporosis of the hip. While the exact cause of transient osteoporosis of the hip remains unclear, several potential pathophysiological mechanisms warrant further investigation to better understand this condition. In this case study, we present an unusual case of a patient with transient osteoporosis of the hip who was diagnosed with non-alcoholic fatty liver disease. A 42-year-old Caucasian man presented with left hip pain. After clinical and radiological evaluations, he was diagnosed with transient osteoporosis of the hip. Biochemical assessment revealed a persistent and isolated elevation of alanine aminotransferase. The patient underwent a treatment regimen involving 16 days of intramuscular neridronate along with physical therapy. At the 3- and 6-month follow-ups, he showed significant improvements in both clinical symptoms and radiological findings. In our study, we propose, for the first time, a potential association between liver disorders, specifically non-alcoholic fatty liver disease, and the onset of transient osteoporosis of the hip, exploring the possible pathophysiological mechanisms connecting these two conditions and discuss the rationale for both pharmacological and non-pharmacological treatments.
Keywords
Background
Transient osteoporosis of the hip (TOH) is a rare condition that typically affects middle-aged men or women in late pregnancy. 1 It is characterized by functional limitations and musculoskeletal pain, leading to restriction in daily activities. 2 Magnetic resonance imaging (MRI) is the gold standard for diagnosing TOH. It can detect changes in the signal intensity of the bone marrow, showing high signal on fluid-sensitive sequences such as T2/proton density with fat suppression and short tau inversion recovery (STIR), along with low signal intensity on T1-weighted images. 3
The pathophysiological mechanisms underlying TOH are not fully understood. Some researchers suggest that factors like increased bone turnover, venous hypertension, and/or microfractures may contribute to the development of bone edema. 1 Various risk factors are associated with TOH, including pregnancy, alcohol consumption, smoking, corticosteroid use, trauma, chronic inflammatory rheumatic diseases, vascular disorders, and metabolic conditions. 1
In this article, we present an atypical case of TOH, discussing the clinical manifestations, diagnostic process, and therapeutic approaches employed. Furthermore, we propose a new pathophysiological hypothesis.
We adhered to the CARE guidelines during the development of this manuscript. 4
Case presentation
A 42-year-old Caucasian man was referred to our outpatient rehabilitation service of Physical and Rehabilitation Medicine of University of Campania “Luigi Vanvitelli” in June 2021 with complaints of pain in his left lower limb, specifically in the trochanteric region and anteromedial thigh, lasting approximately 2 weeks. He was 1.78 m tall, weighed 78 kg (body mass index: 24.6 kg/m2), and worked as a sommelier. He reported consuming approximately 1 unit of alcohol daily and engaging in moderate physical activity. His medical history included a diagnosis of non-alcoholic fatty liver disease in 2019, for which he had been undergoing regular clinical and instrumental follow-up, including ultrasound examinations.
During the initial physical examination, the patient reported moderate left hip pain, rated 5/10 on the numeric rating scale (NRS), particularly when the hip was maximally flexed and during the flexion, adduction, and internal rotation (FADIR) test. However, there were no limitations in the range of motion during active and passive left hip flexion. Additionally, no pain or joint limitations were noted in the left knee, and muscle strength in the lower limbs was preserved. To investigate the possibility of femoroacetabular impingement, hip x-rays were requested in antero-posterior, lateral, and Dunn lateral views. The patient was prescribed acetaminophen 1000 mg as needed for pain management.
A few days later, the patient returned to our outpatient service without having undergone the radiography test. He now reported sudden and severe pain in the left lower limb (NRS 9/10) and difficulty in walking. Upon physical examination, active flexion of the left hip had worsened, limited to 70°, although passive flexion remained unaffected. Muscle strength assessment revealed weakness in the left hip flexor muscles, likely due to increased pain intensity during contractions. Tests for femoral nerve stretch and straight leg raise were performed to rule out irritation of the lumbosacral nerve roots.
To further investigate, MRI scans of the hips and laboratory tests were requested. Laboratory results were largely normal, except for a low red blood cells distribution width of 10.8% (normal range: 11.6%–14.6%) and an elevated alanine aminotransferase (ALT) level of 61 U/L (normal value: 47 U/L) (Table 1).
Laboratory results of complete blood cell count, complete blood chemistry, and serum protein electrophoresis.

25 (OH) vitamin D3: 25 hydroxy vitamin D3; CRP: C-reactive protein; ESR: erythrocyte sedimentation rate; HDL: high-density lipoprotein; PTH: parathyroid hormone; TSH: thyroid-stimulating hormone.
out of range.
The MRI revealed areas of high signal intensity on STIR sequences at the femoral head and neck (Figures 1(a) and (d)), leading to a diagnosis of TOH based on clinical and radiological findings. Treatment included intramuscular neridronate at a dose of 25 mg/day for 16 consecutive days, calcium citrate 500 mg twice daily for 1 month, and cholecalciferol 2000 IU/day for 1 month. Additionally, the patient underwent physical therapy modalities, including pulsed electromagnetic fields (PEMFs) stimulation using single-voltage pulses lasting 1.3 milliseconds at a frequency of 75 Hz for 8 hours per day (overnight) over a total of 45 days. The patient was also advised to adopt protected weight-bearing for 20 days to facilitate recovery.

Assessment of the healing process progression through magnetic resonance imaging (MRI). (a) Axial MRI performed in July 2021: high signal intensity areas at left femoral head and neck. (b) Axial MRI performed in October 2021: smaller extension of the intraspongious edema at left femoral head. (c) Axial MRI performed in January 2022: normalization of the signal of the left femoral head. (d) Coronal MRI performed in July 2021: high signal intensity areas at left femoral head and neck. (e) Coronal MRI performed in October 2021: smaller extension of the intraspongious edema at left femoral head and (f) Coronal MRI performed in January 2022: normalization of the signal of the left femoral head.
One month later, laboratory tests were conducted, which showed mostly normal results, except for a persistently elevated serum ALT levels of 66 U/L.
During a clinical follow-up in October 2021, the patient reported significant pain relief (NRS 2) and showed only mild limitation in external rotation of the hip, with a negative FADIR test. Muscle strength in the left hip flexor muscles had returned to normal. A follow-up MRI scan (Figure 1(b) and (e)) revealed “intraspongiosus edema in the posterior region of the left femoral head, with slightly reduced extent compared with the initial MRI.”
A subsequent MRI conducted in January 2022 (Figure 1(c) and (f)), indicated that the signal in the femoral head returned to normal compared with the previous scan. Figure 2 illustrates the timeline of diagnostic assessments and clinical progression.
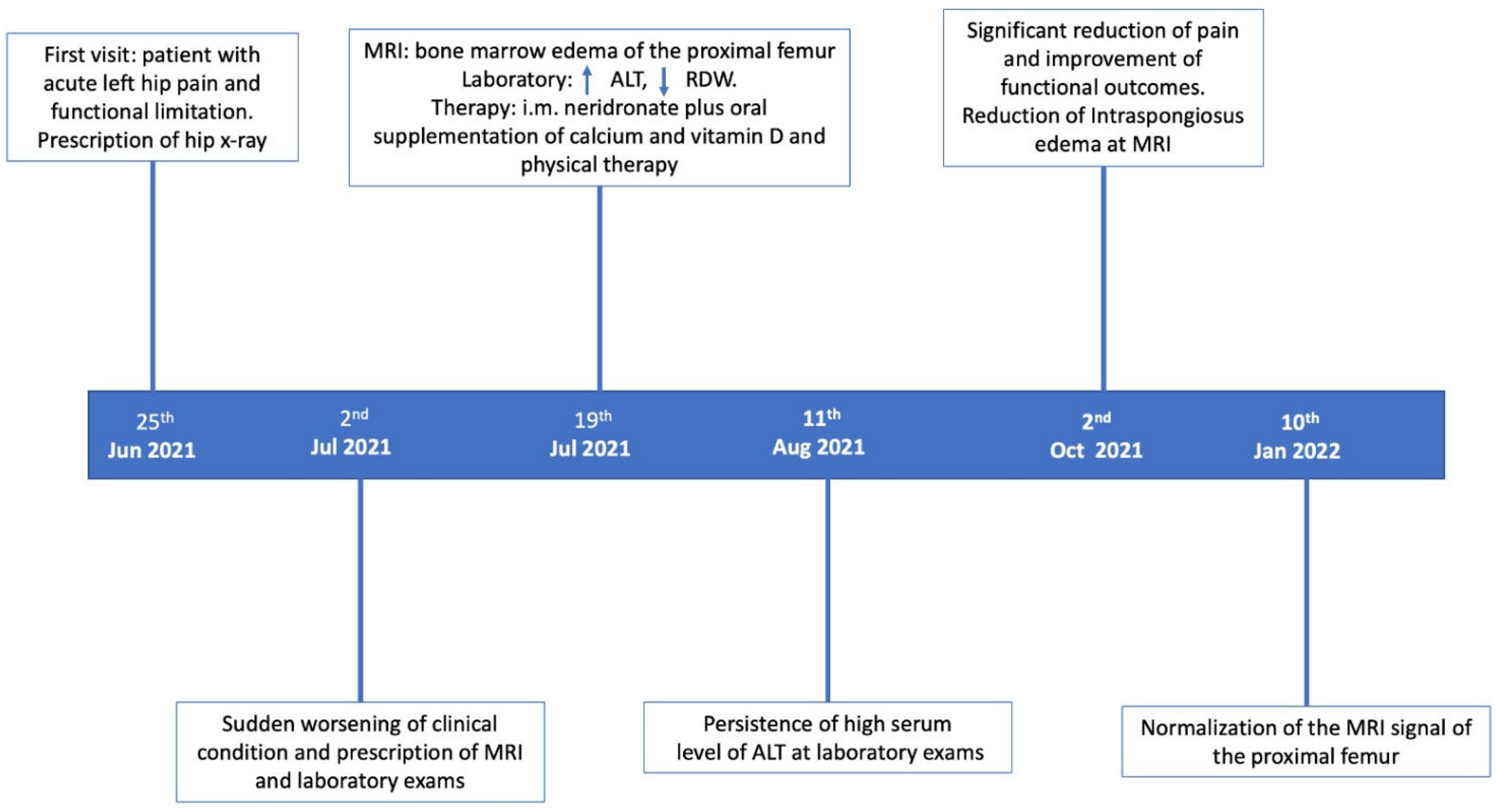
The patient’s journey.
Discussion
TOH is a rare condition with an unknown cause. 5 Several potential factors have been suggested as triggers for its development, including inflammation, infection, trauma, surgery, degenerative processes, ischemic injury, medications, and metabolic disorders. To our knowledge, in this article, we present the first reported case of TOH in a patient with NAFLD, in the absence of other potential causes of metabolic bone disorders (MBDs).
The exact pathogenic mechanisms linking hepatic disorders and TOH remain unclear. In 1984, Rozenbaum et al. 6 proposed a potential association between the hemodynamic changes observed in cirrhosis and pregnancy, and the development of TOH. Although liver disorders have been found to be linked to bone damage, no studies have explored a possible connection between liver disorders and TOH. A recent systematic review comprising seven studies, revealed that individuals with NAFLD are at a higher risk of developing osteoporosis compared with controls of both sexes, with an increased risk of nearly 50%. 7 Several mechanisms have been proposed to explain this association, including chronic inflammation, insulin resistance, and hyperlipidemia. Karmakar et al. 8 demonstrated that rats with liver disorders experienced a reduction in the urinary secretion of calcium, phosphate, creatine, ALP, and tartrate-resistant acid phosphatase when given a black tea extract. This extract contains various compounds, such as flavonoids which possess antioxidant, hypocholesterolemic, and hypolipidemic properties. The authors postulated that black tea could potentially protect against skeletal dysfunction induced by non-alcoholic steatohepatitis by modulating the balance of receptor activator of nuclear factor kappa ligand (RANKL) and osteoprotegerin (OPG) through a reduction in RANKL levels.
Clinical studies have also indicated a connection between MBDs and chronic liver diseases. In 2010, Guañabens et al. 9 demonstrated a high prevalence of osteoporosis in more than one-third of patients with primary biliary cirrhosis. They hypothesized that inflammation of the bile duct epithelium triggers the overexpression of sclerostin, a crucial regulator of bone metabolism that negatively influence the Wnt/B-catenin signaling pathway, leading to bone loss. 9 The same authors also observed an increase in the RANKL/OPG ratio in this population, a condition closely linked to osteoblast-induced osteoclastogenesis and bone resorption. 10 These pathophysiological mechanisms appear to play a role in various chronic liver diseases, particularly in the early stages. 11
The pathogenesis of bone involvement may vary depending on the type of liver disease. In cases of viral hepatitis, the release of proinflammatory cytokines increases osteoclastic activity, resulting in bone resorption.11,12 In contrast, cholestatic liver diseases are associated with reduced bone formation. 10 Specifically, levels of an isoform of fibronectin, known as oncofetal fibronectin, are elevated in patients with cholestatic liver disease. 11 Oncofetal fibronectin, secreted by matrix-producing stellate cells in the liver, inhibits osteoblast activity. Furthermore, elevated bilirubin levels in patients with cholestatic liver diseases have been shown to suppress osteoblast proliferation. 13
We hypothesize that in our clinical case, similar to patterns observed in other bone marrow edema syndromes (BMESs), such as complex regional pain syndrome (CRPS) type 1, 14 hepatic impairment (elevated serum ALT levels) in a patient exhibiting maladaptive plasticity could have potentially initiated the development of TOH as no other causative factors were identified.
From a histopathological perspective, bone marrow edema is characterized by increased vascularization, proliferation of synovial tissue, and inflammatory lymphocytic infiltrates within the bone marrow. 15 Ischemic processes resulting from venous drainage disturbances are believed to contribute to its development. Angiographic and scintigraphy studies have confirmed the presence of dilated blood vessels in the femoral head, leading to increased perfusion in the affected area. 16
The bone resorption observed in TOH is attributed to excessive osteoclast activity. 17 Bisphosphonates, potent anti-resorptive drugs, are commonly prescribed to treat various MBDs characterized by elevated bone turnover, including BMES. Furthermore, these drugs may exert additional anti-inflammatory and analgesic effects, making them a frontline option for managing CRPS type 1.1,18,19
Varenna et al.20,21 investigated the efficacy of neridronate for treating CRPS type 1, demonstrating that this drug is effective in managing pain and edema at cumulative dose of 400 mg, by intravenous (i.e. in 10 days) 20 or intramuscular (i.e. in 16 days) administration routes. 21
Based on the available evidence, we hypothesized that neridronate could be a viable option also for the treatment of TOH by targeting both the MBD and the associated bone pain observed in this condition. 22
Specifically, we proposed the same therapeutic schedule, administered via the intramuscular route, could be effective for TOH management.21–23 Although the anti-resorptive mechanism of neridronate is well established, its pain-relieving effects appear to be linked to multiple mechanisms. First, neridronate inhibits the local production of inflammatory mediators and modulates nociceptors. 24 Additionally, by reducing osteoclast activity, it alleviates pain by mitigating local acidosis. 25
In addition to pharmacological treatment, we integrated calcium and vitamin D supplementation as administered in previous studies and physical modalities into the management of TOH. 26 This approach leveraged the common use of PEMFs for patients with bone edema, given their ability to enhance osteoclast apoptosis and increase osteoblast viability, thereby modulating bone turnover. 27 Studies have demonstrated that PEMF therapy increases ALP activity and mineralization in vitro and animal models, while reducing both extracellular matrix degrading enzymes, such as metalloproteinases-11, and sclerostin.28–30 Furthermore, PEMFs regulate bone metabolism influencing the RANKL/OPG ratio and Wnt/β-Catenin signaling pathways. 31 In our clinical case, this combined approach led to significant improvements in both clinical symptoms and instrumental findings.
However, this does not allow to establish a cause–effect relationship due to the absence of histopathological evidence confirming the proposed pathophysiological mechanisms. Additionally, the report is retrospective in nature, meaning that despite the documentation of numerous clinical and instrumental findings, some medical records may not include all relevant data. Lastly, the findings cannot be generalized, as the case report is not representative of the broader population.
Conclusion
TOH poses a diagnostic and therapeutic challenge for healthcare providers. The condition can be triggered by a range of underlying factors, requiring comprehensive investigation to identify potential causes. This case report underscores the possible role of liver disorders, specifically NAFLD in the development of TOH. Given the established connection between liver diseases and MBDs, we propose potential pathophysiological mechanisms linking NAFLD to TOH. Considering the likely contribution of osteoclast overactivity to TOH pathogenesis, we administered neridronate in combination with physical therapy. This strategy was guided by the known effects of these interventions on bone metabolism and pain management, alongside existing evidence supporting neridronate efficacy in treating BMES. Although these findings are promising, further research is required to better understand the underlying mechanisms of TOH and to establish optimal management strategies for BMES in patients with liver disorders.
Footnotes
Acknowledgements
None.
Authors’ contributions
MP, AM, and GI designed the report; MP and SL collected the patient’s clinical data; and AM, MP, and FG analyzed the clinical data and drafted the paper. All authors read and approved the final manuscript.
Consent for publication
Written informed consent was obtained from the patient for publication of this case report and any accompanying images.
Data availability statement
The data supporting this case report are available from the corresponding author on reasonable request, in compliance with the Ethics Committee’s guidelines to ensure patient confidentiality.
Declaration of conflicting interests
The authors have no conflicts of interest to disclose.
Funding
None.
