Abstract
This study investigated the occurrence of left ventricular thrombus formation and subsequent embolic stroke in patients with Takotsubo syndrome. Through the case analysis of an 80-year-old Chinese elderly female patient, it was found that although Takotsubo syndrome is generally considered a self-limiting disease, it can lead to severe complications such as left ventricular thrombus and cerebral stroke. The patient experienced a cardioembolic stroke during hospitalization, and magnetic resonance imaging revealed acute cerebral vascular occlusion. This case highlights the importance of anticoagulation therapy, especially in the prevention of thromboembolic events in patients with Takotsubo syndrome complicated with intracardiac thrombosis.
Keywords
Introduction
Takotsubo syndrome (TTS), first described by Japanese scholar Sato in 1990, is characterized by transient left ventricular systolic dysfunction due to severe psychological and/or physical stress. 1 Despite its low incidence of 0.5% to 0.9% in the general population, with most angiograms showing no significant obstructive lesions, it was initially considered a benign and self-limiting condition. However, recent clinical reports have unveiled an increased incidence of severe complications in TTS patients, including systemic thrombosis, arrhythmias, cardiogenic shock, and life-threatening ventricular rupture. These complications suggest that approximately 20% of TTS patients experience severe complications during hospitalization, potentially leading to short-term and long-term adverse outcomes that are comparable to those observed in acute myocardial infarction.2–4
In this report, we describe a case of TTS in which ventricular angiography disclosed an intracardiac thrombus, subsequently precipitating a cerebrovascular embolism. The report of this study conforms to the CARE guidelines, and all patient information has been de-identified. 5
Case presentation
An 80-year-old Chinese elderly woman with a history of smoking but having quit 40 years ago was admitted to the Department of Cardiology at Hebei Yanjia Hospital on 27 March 2024, due to chest pain accompanied by nausea and vomiting. Upon consultation, 1 month prior to the visit, there was noticeable pain due to a rib fracture accompanied by anxiety. On admission, her blood pressure was 118/79 mmHg, and her pulse rate was 82 beats per minute. An electrocardiogram revealed sinus rhythm with persistent ST-segment elevation in leads V2-V5 and inverted T-waves in leads V2-V5 over 31 days (Figure 1). The laboratory results demonstrated elevated levels of cardiac troponin I and brain natriuretic peptide. An echocardiogram revealed left ventricular end-diastolic dimension of 41 mm, end-systolic dimension of 32 mm, and an ejection fraction of 40% (Simpson). The interventricular septal thickness was 13 mm, and the posterior wall thickness was 12 mm. There was significantly reduced contraction motion in the mid and apical segments of the left ventricle, with increased motion in the basal segment. The left ventricular outflow tract was narrowed with increased velocity, measuring 3.31 cm/s, and a maximum pressure gradient of 44 mmHg. The echocardiogram suggested the possibility of TTS (Figure 2(a) and (b)).
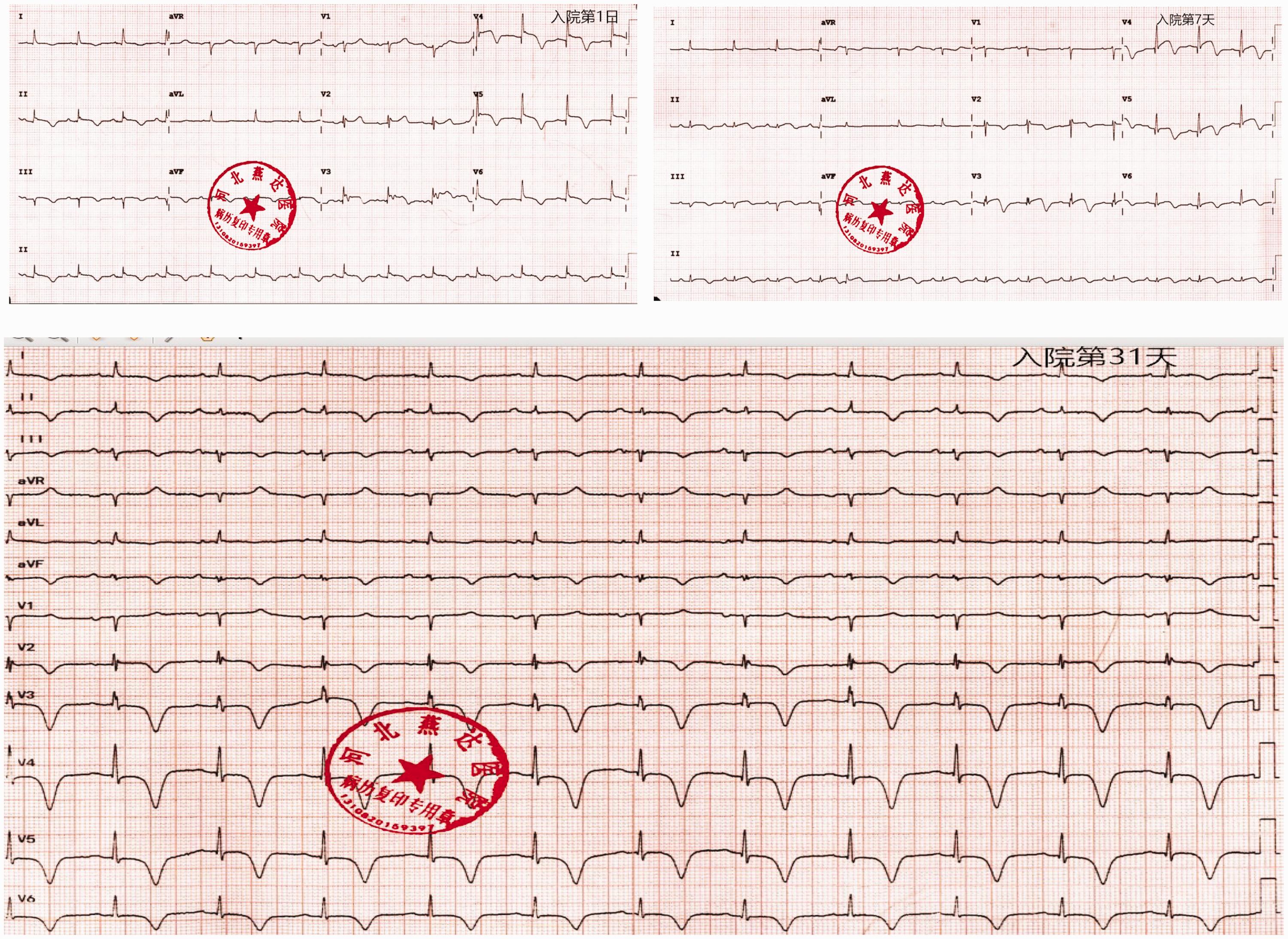
Electrocardiogram on admission showed leads V3-V5. On day 7, persistent ST-segment elevation was observed in leads V2-V5. On day 31, a deeply inverted T-wave was observed in leads V2-V6.
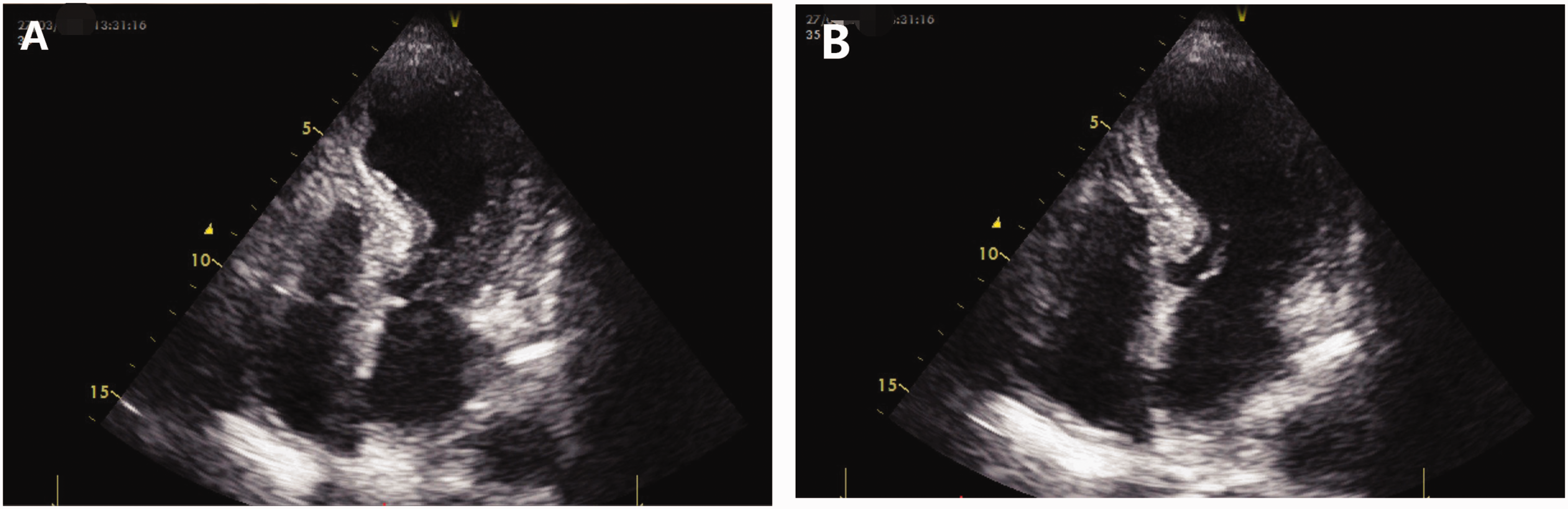
Transthoracic echocardiography (TTE) apical four-chamber view following admission. The echocardiogram during systole (a) and diastole (b) showed apical ballooning of the left ventricle and right ventricle apical free wall akinesis.
In order to differentiate TTS from acute coronary syndrome, we performed cardiac catheterization on 3 April 2024. Coronary angiography did not reveal any coronary artery stenosis, particularly the left anterior descending (Figure 3(a) and (b)), and left ventriculography demonstrated apical ballooning and basal hyperkinesis, apex of the left ventricle thrombus (Figure 3(c) and (d)).

Coronary angiography did not reveal any coronary artery stenosis, particularly the left anterior descending (a, b). Left ventriculography demonstrated apical ballooning and basal hyperkinesis consistent with TTS, with a thrombus at the apex of the left ventricle indicated by a black arrow (c, d).
Upon admission, biochemical and related tests revealed low-density lipoprotein cholesterol (LDL-C) at 2.28 mmol/L, creatine (Cr) at 43.5 µmol/L, troponin I (TnI) at 5.33 ng/mL, B-type natriuretic peptide (BNP) at 1735.31 pg/mL, D-dimer at 3518.02 ng/mL. Erythrocyte sedimentation rate was 48 mm/h, with negative results for antinuclear antibodies and antiphospholipid antibodies. Thyroid function was within normal limits.
On 9 April 2024, after admission, cardiac magnetic resonance imaging (MRI) revealed abnormal regional myocardial wall motion with basal outflow enhancement without pericardial effusion. The cardiac MRI shows uneven myocardial thickness, with localized thickening of the interventricular septum, measuring approximately 15 mm at end-diastole and 20 mm at end-systole. There is also a narrowing of the outflow tract. The apex exhibits myocardial thinning and reduced motion. In the short-axis view, the left ventricular free wall thickness is approximately 7.6 mm at end-diastole and 12.3 mm at end-systole, with a growth rate of approximately 61.8%. T2-weighted imaging shows edema in the region near the endocardium of the anterior wall and apex of the left ventricle. The left ventricular apex thrombus is marked with an arrow (Figure 4(a) to (e)).

Cardiac magnetic resonance imaging (CMR) revealed abnormal regional myocardial wall motion, with basal outflow enhancement, without pericardial effusion. The left ventricular apex thrombus is marked with an arrow (a–d). T2-weighted imaging shows edema in the region near the endocardium of the anterior wall and apex of the left ventricle (e).
After the patient’s admission on 12 April 2024, they experienced sudden difficulty in moving their left limbs and speaking. Upon examination, their left muscle strength was rated at three out of five, and pathologic signs were positive. The cranial brain MRI shows hyperintense signals in the right pontine area on T2 fluid attenuated inversion recovery (FLAIR) sequence and high signal intensity in the right pontine-brainstem junction on diffusion-weighted imaging, suggesting acute cerebrovascular occlusion (Figure 5(a) to (c)). The ejection fraction (EF) value on the day before the embolism returned to 57%.
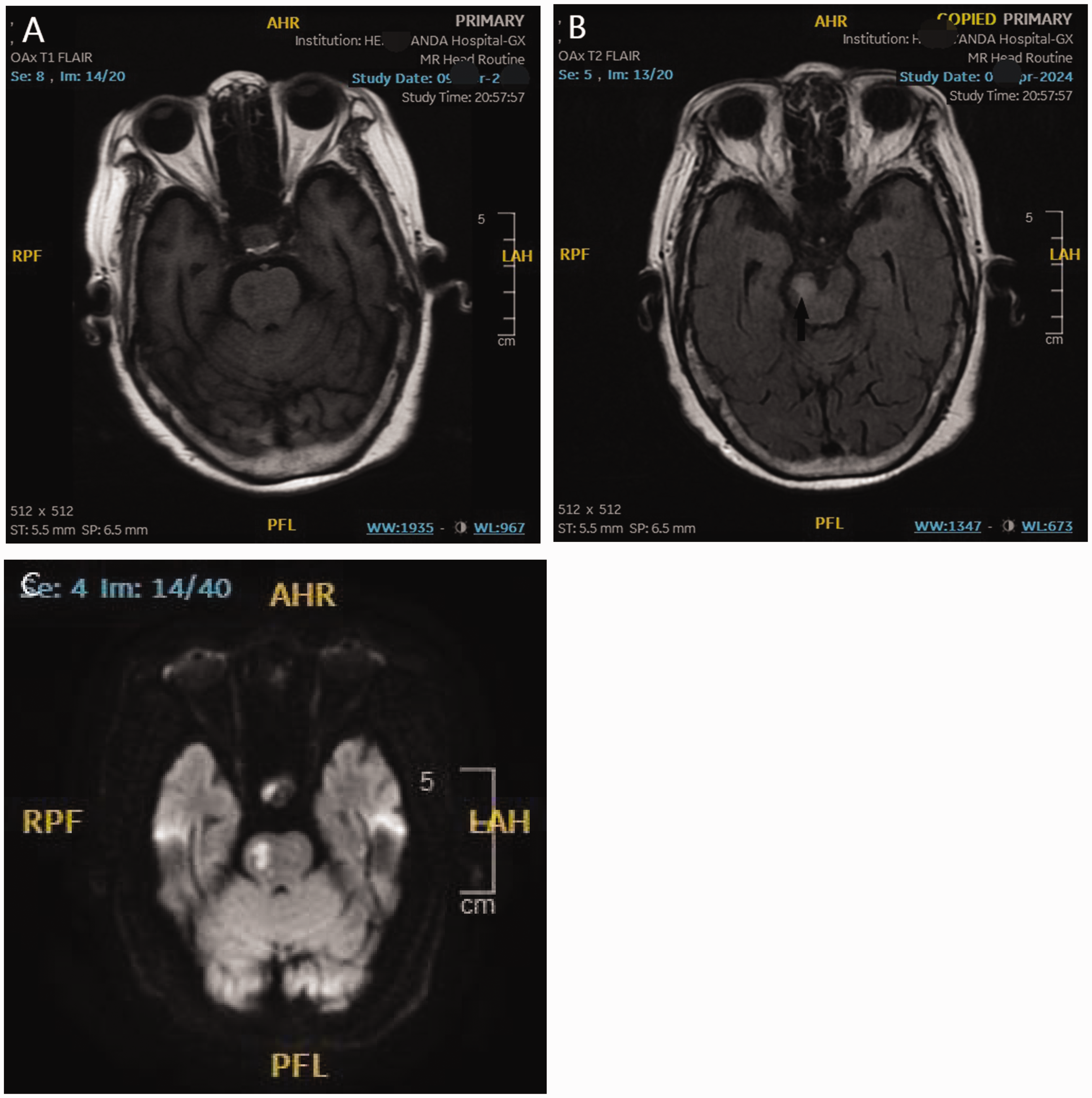
Magnetic resonance imaging (MRI) shows hyperintense signals in the right pontine area on the T1 and T2 fluid attenuated inversion recovery (FLAIR) sequence (a, b) and high signal intensity in the right pontine-brainstem junction on diffusion-weighted imaging (c), suggesting acute cerebrovascular occlusion (indicated by a black arrow in (b)).
Following admission, the patient’s total white blood cell count, neutrophil percentage, and C-reactive protein response showed a significant and prolonged increase (Figure 6).

White blood cell count, neutrophil percentage, and C-reactive protein response following admission.
Initially, antiplatelet therapy was initiated due to the suspicion of myocardial infarction. Subsequently, upon the detection of ventricular thrombi, the treatment regimen was adjusted to rivaroxaban 15 mg orally once daily for anticoagulation therapy. Concurrently, lipid control was maintained, and metoprolol 12.5 mg orally twice daily, along with dapagliflozin 10 mg orally once daily, were administered to improve stress response and remodeling. The above treatment is planned for 3 months, with adjustments to be made based on follow-up results.
A follow-up echocardiogram 1 month later showed normal contraction movements of the mid-segments, basal portions, and apices of the left ventricle (Figure 7(a) and (b)). The patient has undergone a 5-month follow-up period. No cardiac-related symptoms have been reported. Echocardiography reveals that both cardiac function and ventricular wall motion have normalized. Furthermore, no recurrence of cerebral embolism has occurred following 3 months of anticoagulant therapy.

Follow-up transthoracic echocardiography (TTE) 1 month later showed normal contraction movements during systole (a) and diastole (b).
Discussion
TTS, also commonly known as Takotsubo cardiomyopathy or apical ballooning syndrome, is characterized by transient left ventricular (LV) wall dysfunction triggered by physical or emotional factors.
In this case, assessments according to the Mayo criteria revealed abnormal apical and regional wall motion with preserved basal function, as evidenced by echocardiography, ventriculography, and cardiac MRI. Full resolution of wall motion abnormalities was noted upon subsequent follow-up. The electrocardiogram exhibited sustained ST-segment elevation for 7 days, profound inverted T-waves, and increased levels of troponin I and BNP. Coronary angiography showed no significant stenosis in the coronary arteries that could account for the electrocardiography changes, and concurrent factors, including rib fracture pain and anxiety, were identified. Contrast-enhanced cardiac MRI ruled out myocarditis and other cardiomyopathies, such as hypertrophic or amyloid types. These collective findings support a diagnosis of stress-induced cardiomyopathy.
TTS is characterized by acute left ventricular dysfunction, which can contribute to intraventricular thrombus and embolism. 6 A multicenter registry study on TTS LV thrombi (3%) and stroke (1%) among 209 patients identified Q-waves on admission and ejection fraction ≤30% as independent predictors for complications. 7 The International Takotsubo Registry’s multivariate penalized maximum likelihood estimation analysis of TTS patients with ventricular thrombus and/or embolism showed that the model type at the apical region, left ventricular EF ≤30%, a history of vascular disease, and a white blood cell count >10 × 10³ cells/μL at admission were independent predictors of thrombus formation or embolism. 8 In addition to this case being a typical example of apical ballooning, the patient also had an elevated white blood cell count.
The left ventricular outflow tract (LVOTO) gradient of 44 mmHg, in this case, results from basal hypercontractility in the small LV cavity with symmetric hypertrophy of the interventricular septum. It is associated with systolic anterior motion of the mitral valve (SAM). Its prevalence ranges from 12.8% to 25%. The degree of LVOTO is reversible, depending on myocardial function recovery. Patients with a high left ventricular outflow tract (LVOTO) gradient have a higher tendency for left ventricular thrombus formation (5% vs. 1%; p = 0.057). 9
There are other factors that contribute to intracardiac thrombosis and embolization. The C-reactive protein in prediction of LV thrombus formation in patients with TTS was 100% in the study by Haghi et al. 10 D-dimer was found to be significantly higher in patients with LV thrombus compared with the patients without thrombosis; a troponin level of more than 10 ng/mL was the only predictor for LV thrombus formation. 11
Upon admission, this patient presented with a thrombus detected via cardiac catheterization on the eighth day. Considering the patient’s electrocardiogram showed sinus rhythm, there was no objective evidence of atrial fibrillation in the past, no valvular vegetations associated with infective endocarditis, and no history of dilated cardiomyopathy or heart failure; the echocardiogram ruled out atrial septal defect, patent foramen ovale, and other abnormal shunts. The possibility of cardiogenic embolism and paradoxical embolism due to the aforementioned causes can be excluded. The sudden cerebral vascular accident is mainly due to embolic events triggered by intracardiac thrombus. The analysis indicated that abnormal LV wall motion, vortices within the cardiac chambers resulting from these changes, elevated myocardial injury markers such as TnI, increased early inflammatory response (elevated serum C-reactive protein), and platelet activation induced by stress and catecholamines were key factors contributing to ventricular thrombus formation in this patient.
A short course of limited anticoagulation therapy may be needed to prevent stroke, particularly if there is sizeable apical/midventricular akinesis/dyskinesis with apical ballooning, which predisposes to thrombus formation when coupled with the sympathetic overdrive, which induces hypercoagulability. 12 Increased troponin levels (TnI level >10 ng/mL) and apical ballooning were strongly associated with the occurrence of LV thrombi in a recent study by Santoro et al., 13 and anticoagulation should be considered as a prophylactic treatment in such cases. 14 Anticoagulation can be delayed up to 4–14 days due to an increased risk of hemorrhagic transformation in patients with cardioembolic stroke, but no treatment for TTS complical intraventricular thrombus has been examined in a randomized trial. 15 The findings of the BROKEN-SWEDEHEART study may yield insights into strategies for preventing thromboembolism in the left ventricle of the heart in TTS. 16 About anticoagulant drug selection, recent years have seen growing interest in rivaroxaban for treating LV thrombosis. Clinical data and meta-analyses suggest that rivaroxaban is comparable to warfarin in terms of thrombus regression, reduction in thrombus size, bleeding risk, and rehospitalization rate.17,18 Rivaroxaban also offers advantages such as ease of use, rapid onset, and no need for routine monitoring. However, due to the limited number of randomized controlled trials, the 2014 American Heart Association Stroke Guidelines classify direct oral anticoagulants (DOACs) as a reasonable alternative to warfarin for LV thrombosis (Class IIb, level of evidence: C). The 2022 American Heart Association Scientific Statement also supports the use of DOACs as a viable alternative.
Finally, despite the known thromboembolic complications, there are no anticoagulant treatment guidelines in TTS, and clinical attention should be paid to the objective evaluation of the rational initiation of anticoagulation to prevent the occurrence of cerebral embolism.
Conclusion
TTS is a potentially life-threatening form of acute heart failure, yet it lacks evidence-based treatment protocols, especially concerning Finally, despite the known thromboembolic complications, there are no anticoagulant treatment guidelines in TTS, and clinical attention should be paid to the objective evaluation of the rational initiation of anticoagulation to prevent the occurrence of cerebral embolism, intraventricular thrombus and associated cerebral embolism. Consequently, the focus lies on medical optimization to avert subsequent episodes and complications. Preliminary evidence suggests that the prompt initiation of anticoagulation therapy may play a pivotal role in mitigating the risk of concurrent cerebral embolism in TTS patients.
Patient perspective
As a patient diagnosed with stress-induced cardiomyopathy (Takotsubo syndrome), the initial chest pain and nausea were particularly unbearable for me, causing extreme anxiety about my health. However, after the diagnosis of TTS complicated with ventricular thrombus, the adjustment to rivaroxaban anticoagulant therapy and the administration of drugs to improve cardiac function effectively controlled my symptoms and improved my cardiac function.
I am extremely grateful for these timely and effective interventions, which may have prevented further complications. Regular follow-up care has ensured that my condition is closely monitored and the treatment plan is adjusted in a timely manner when necessary. The clear communication from the medical team about my condition and treatment plan has greatly relieved my anxiety. I am fortunate to have received such advanced medical care, which has significantly improved my health status, with no recurrence of cardiac symptoms or cerebral embolism during the follow-up period. This experience has demonstrated the importance of timely and appropriate medical interventions in managing complex diseases such as stress-induced cardiomyopathy.
Footnotes
Acknowledgements
We want to thank Kimi. We acknowledge artificial intelligence for providing English language polishing.
Author contributions
Jingshu Lei contributed in writing original draft manuscript; Ningning Yin and Lina Wang made contributions to the data acquisition and analyse; Zhiqi Zhang is the corresponding author and guided the revisions. All authors have participated sufficiently in the work and agreed to be accountable for all aspects of the work.
Data availability statement
All data included in this study are available upon request by contact with the corresponding author.
Declaration of conflicting interests
None.
Funding
No funding.
Human ethics
Consent was obtained or waived by all participants in this study. The hospital ethics committee approved the publication of this article.
