Abstract
Nephrotic syndrome can occur secondary to malignancy, and its resolution is sometimes observed following tumor resection. We herein report the case of a 67-year-old Asian man who presented with membranous nephropathy associated with carcinoma of the ampulla of Vater. The patient initially presented with edema, hypoalbuminemia, and significant proteinuria with positive PLA2R antibody. Following surgical removal of the tumor, his urinary protein levels normalized, his serum albumin returned to normal, and the nephrotic syndrome achieved complete remission. This case underscores the importance of screening for secondary causes, such as malignancies, in patients with nephrotic syndrome. For those with malignancy-associated nephrotic syndrome, prompt and targeted treatment of the underlying tumor is critical to avoid missing the window for surgical intervention. This report aims to provide insights into the diagnosis and management of tumor-associated nephrotic syndrome, contributing to a better understanding of this rare clinical entity.
Keywords
Introduction
Nephrotic syndrome is a condition characterized by massive proteinuria, edema, hyperlipidemia, and hypoalbuminemia. It is classified as either primary or secondary, with malignancies being a potential secondary cause. Both hematologic and solid tumors can induce tumor-associated nephrotic syndrome, with the most common hematologic malignancies being leukemia and multiple myeloma, while lung, prostate, and gastrointestinal cancers are the most frequently associated solid tumors. 1 In this report, we describe a patient who initially presented with edema, proteinuria, and hypoproteinemia and was diagnosed with membranous nephropathy via renal biopsy. Further examination revealed carcinoma of the ampulla of Vater. Following surgical resection of the tumor, the patient’s nephrotic syndrome significantly improved.
This case report was approved by the Ethics Committee of The Central Hospital of Wuhan (Approval Number: WHZXKYL2022-112-01). All patient details have been removed to ensure privacy, and written informed consent was obtained from the patient before treatment. The reporting of this case conforms to the CARE guidelines. 2
Case report
A 67-year-old Asian man presented with bilateral lower extremity edema, foamy urine, and low back pain that had persisted for 4 months. In March 2022, the patient had sought care for these symptoms at a local hospital. Laboratory tests revealed an anti-phospholipase A2 receptor antibody (PLA2R) level of 36.58 RU/mL (+), serum albumin of 26.3 g/L, and urinalysis confirming proteinuria. He was diagnosed with nephrotic syndrome at that institution but remained untreated. He subsequently presented to our hospital on 18 July 2022.
The patient’s medical history included hypertension, upper gastrointestinal bleeding, and hyperlipidemia. His medication regimen consisted of irbesartan tablets (0.3 g in the morning and 0.15 g at night) and atorvastatin calcium (20 mg each evening). He did not smoke or consume alcohol.
On physical examination, mild bilateral lower extremity edema was noted, with no other abnormalities observed.
At the time of admission, the patient’s blood biochemical tests revealed the following: serum creatinine (Scr) of 1.4 mg/dL, serum albumin (Alb) of 20.5 g/L, total bilirubin of 20.9 μmol/L, direct bilirubin of 14.6 μmol/L, and PLA2R of 117.67 RU/mL (+). Tumor markers showed a carcinoembryonic antigen level of 4.06 ng/mL, alpha-fetoprotein level of 2.52 ng/mL, and cancer antigen 19-9 level of 9.46 U/mL. Immunological tests revealed an antinuclear antibody profile (IgG) of 7.2 g/L, with antinuclear antibody, antineutrophil cytoplasmic antibody, and serum protein electrophoresis results above the upper limit of the reference range. Urinalysis indicated a microalbumin level of 943.4 mg/L, a urine microalbumin/urine creatinine ratio of 56.5 mg/g, and 24-hour proteinuria of 4.9 g.
An upper abdominal computed tomography scan demonstrated mild dilatation of the intrahepatic and extrahepatic bile ducts, along with blurred margins of the pancreatic body. The patient was treated with antihypertensive, lipid-regulating, and symptomatic supportive therapy.
The pathologic findings of the renal biopsy were as follows. Under light microscopy (Figure 1(a)), the glomerular capillary walls appeared crimped, with mild stiffness and dilation. Mild segmental hyperplasia of the mesangial cells and matrix was observed. Eosinophilic protein deposits were minimally present beneath the epithelium, accompanied by granular and vacuolar degeneration of renal tubular epithelial cells. Additionally, minor proteinaceous casts and small foci of atrophy were noted, along with areas of lymphomonocytic and plasma cell infiltration in the renal interstitium. Under immunofluorescence microscopy, deposits of PLA2R (2–3+) (Figure 1(b)), IgG (2+), IgG1 (2+), IgG2 (2–3+), IgG3 (3+), IgG4 (4+), IgM (2+), IgA (2+), C1q (2+), C3 (2+), and C4 (2+) were observed, with diffuse capillary wall deposits and occasional deposits in the thylakoid zones. Electron microscopy (Figure 1(c)) revealed numerous electron-dense deposits beneath the podocytes, with peg-like formations in the basement membrane. Overall, these pathologic findings were consistent with stage II membranous nephropathy.
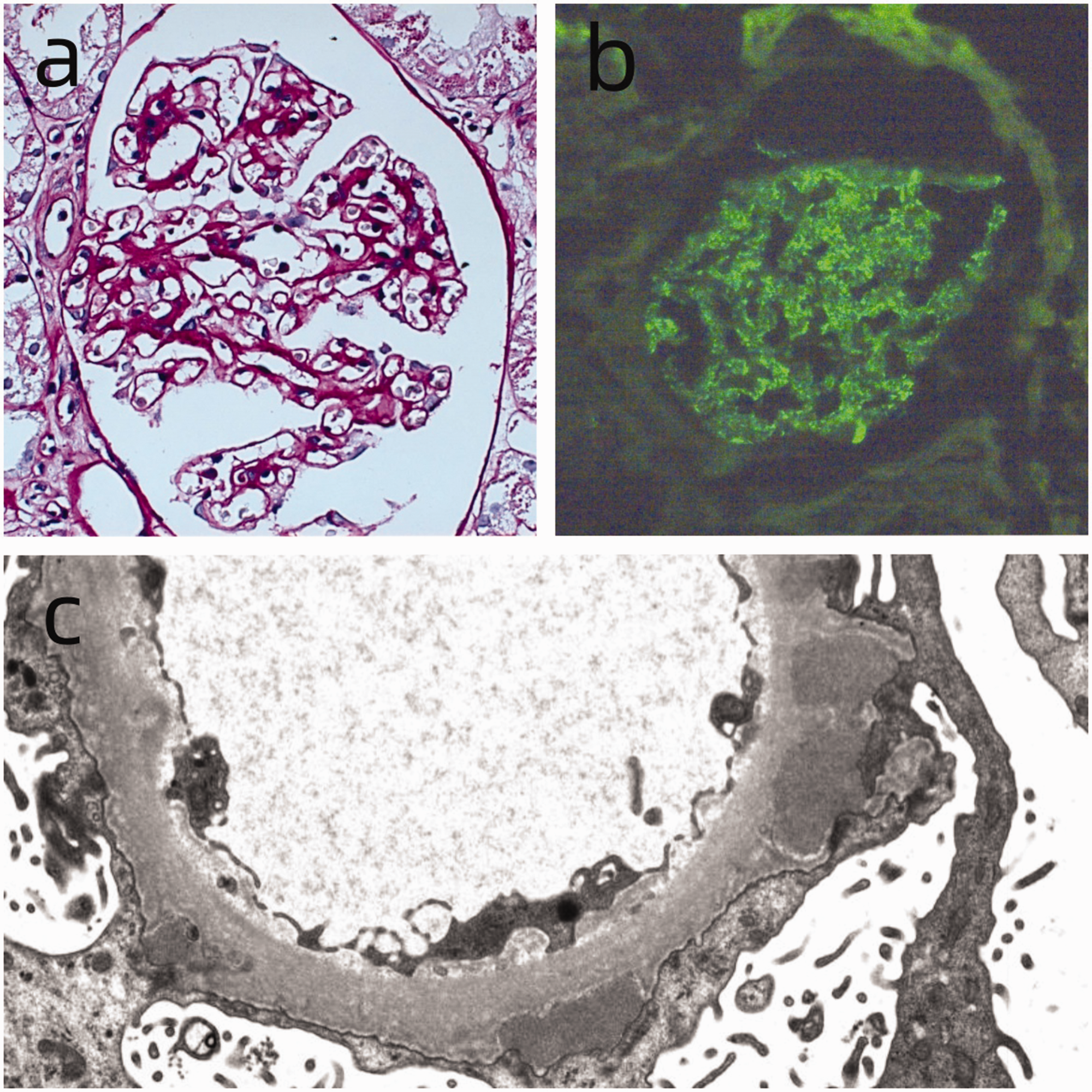
Microscopic presentation of renal biopsy. (a) Light microscopy: Periodic acid–Schiff staining shows stiffness and thickening of the basement membrane in the glomerulus (original magnification ×400). (b) Immunofluorescence microscopy: PLA2R deposition is observed along the capillary walls (original magnification ×400) and (c) Electron microscopy: Electron-dense material deposition and peg formation are visible (original magnification ×4000).
Computed tomography of the upper abdomen revealed mild dilatation of the intrahepatic and extrahepatic bile ducts, while blood biochemistry tests indicated elevated bilirubin levels. The patient was subsequently transferred to the Department of Gastroenterology for further evaluation. Endoscopic retrograde cholangiopancreatography identified a space-occupying lesion in the ampulla of Vater. A subsequent enteroscopic biopsy revealed high-grade intraepithelial neoplasia, and the possibility of adenocarcinoma could not be excluded.
On 3 August 2022, the patient underwent radical pancreaticoduodenectomy and small intestinal diverticulectomy in the Department of Hepatobiliary and Pancreatic Surgery. Pathologic examination confirmed a moderately differentiated adenocarcinoma (carcinoma of the ampulla of Vater) measuring 2.0 cm at its largest diameter, with infiltration into the muscularis propria and local involvement of the duodenal submucosal layer. There was no pancreatic parenchymal involvement; however, nerve and vascular invasion were observed. Immunohistochemical analysis revealed positive staining for PMS2, MLH1, MSH2, MSH6, CK19, CK8/18, CK20, Villin, and SATB2, with partial positivity for P53, CA19-9, CK7, CDX2, and Mucin5AC. Mucin-2, MUC-6, CD31, and D2-40 were negative. The Ki-67 labeling index in hotspot areas was approximately 60%. Postoperatively, the patient received basic treatment, including antihypertensive therapy with irbesartan.
Postoperatively, the patient’s urinary protein, Scr, and Alb levels, as detailed in Figures 2 and 3, showed gradual normalization following tumor removal. The Scr level remained stable, while urinary protein excretion progressively declined. At 4 weeks after surgery, the patient’s Alb level was 29.4 g/L. By 4 months, it had increased to 30 g/L, with the Scr level remaining within the reference range. Urinalysis showed no detectable urinary proteins, a urinary microalbumin level of 623.8 mg/L, and a urinary microalbumin/urinary creatinine ratio of 29.7 mg/g. The patient was followed up for 20 months, during which no tumor recurrence was observed. On 23 March 2024, laboratory tests revealed an Alb level of 44.1 g/L, a urinary microalbumin/urinary creatinine ratio of 26.7 mg/g, a urinary microalbumin level of 20.30 mg/L, and a negative urine protein analysis. The patient’s membranous nephropathy was in complete remission.

Patient’s urinary protein levels after admission.

Patient’s serum creatinine (Scr) and albumin (Alb) levels after hospital admission.
Discussion
Our patient was initially diagnosed with membranous nephropathy upon admission, with no abnormalities detected in tumor markers. However, because of mild bilirubin elevation and mild bile duct dilation, a multidisciplinary diagnostic approach was pursued, leading to the discovery of a digestive tract tumor. Following radical tumor resection, the patient’s membranous nephropathy was completely resolved, achieving a favorable therapeutic outcome.
The potential link between nephrotic syndrome and Hodgkin’s lymphoma was first reported by Galloway in 1922. 3 Later, in 1966, Lee et al. 4 observed that 11 of 101 patients with nephrotic syndrome also had malignancies, establishing that nephrotic syndrome can be secondary to malignancy. Among malignancy-associated nephrotic syndromes, membranous nephropathy is the most common. 5 It is linked to malignancy in 5% to 10% of adult patients, 6 primarily in association with solid tumors, with fewer cases related to hematologic malignancies. 5 Among solid tumors, lung cancer is the most frequently associated, followed by prostate and gastric cancers. 1
The pathogenesis of tumor-associated membranous nephropathy involves two primary mechanisms7,8: circulating immune complexes and in situ immune complexes. In the first mechanism, tumor antigens deposit in the glomerulus, promoting antibody deposition and complement activation. This leads to the formation of a large number of circulating immune complexes, which accumulate on the basement membrane, causing damage to epithelial cells and the glomerular basement membrane. In the second mechanism, antibodies generated against tumor antigens recognize specific antigens on podocyte foot processes, such as PLA2R and THSD7A, leading to the formation of in situ immune complexes. This process damages the foot processes, resulting in basement membrane injury. The disruption of both mechanical and electrical barriers ultimately leads to significant protein loss.
PLA2R is a specific marker for primary membranous nephropathy with diagnostic value, with approximately 70% to 80% of patients testing positive for PLA2R antibodies. 9 The level of PLA2R antibody can also reflect disease severity and treatment response. It has been suggested that PLA2R1 exerts tumor-suppressive effects by activating the downstream JAK2 signaling pathway, 10 while its transcriptional repression may facilitate tumorigenesis. 11 Patients with tumor-associated membranous nephropathy are often negative for PLA2R antibodies 12 ; however, the patient described in this report was positive for PLA2R antibodies. Recently, more reports have highlighted PLA2R antibody positivity in patients with tumor-associated membranous nephropathy,13–15 a phenomenon warranting further investigation. The relationship between PLA2R antibodies and malignancy remains unclear, and additional studies are needed to explore their potential connection. Research should also address whether PLA2R antibodies have a pathophysiological effect on tumors and examine the improvement in proteinuria following treatment of the primary tumor in patients who are PLA2R antibody positive. Furthermore, patients with membranous nephropathy who have positive PLA2R antibody tests may have comorbid malignancies, which should not be overlooked. A 2021 study identified nerve epidermal growth factor-like 1 (NELL1) as a specific antigen for tumor-associated membranous nephropathy, 16 suggesting that NELL1 could become a new marker for screening membranous nephropathy patients for potential malignancies in the future.
The prognosis of patients with malignancy-associated nephrotic syndrome is generally poor, with a 5-year mortality rate as high as 68.5%. 17 Treatment primarily focuses on addressing the underlying malignancy through surgical resection, radiotherapy, chemotherapy, or targeted therapy to control tumor progression. Concurrently, angiotensin-converting enzyme inhibitors or angiotensin II receptor blockers are administered to reduce intraglomerular pressure and decrease urinary protein excretion. In some cases, a combination of traditional Chinese and Western medicines may help protect renal function and control nephropathy progression. Studies have shown significant relief of nephrotic syndrome in patients with malignant tumors following effective antitumor therapy.18–22 The likelihood of remission in malignancy-associated nephrotic syndrome depends on the tumor’s developmental stage and the thoroughness of treatment. If detected early and without metastasis, nephrotic syndrome may be completely resolved through surgical intervention. However, if the tumor is unresectable or metastatic, the therapeutic effect is less favorable, although timely and effective radiotherapy or chemotherapy can still alleviate symptoms. Glucocorticoids and immunosuppressive agents should be used with caution because their immunosuppressive properties may increase the risk of tumor development and introduce medical risks. This case highlights the importance of thoroughly excluding secondary causes in patients with nephrotic syndrome, as treatment of the underlying condition is key to achieving effective outcomes. Inappropriate treatment, such as the indiscriminate use of hormones or immunosuppressants, not only results in poor efficacy but may also lead to complications, delay treatment of the primary cause, and ultimately worsen the patient’s prognosis and survival.
Conclusions
In this report, we have described a case of nephrotic syndrome associated with carcinoma of the ampulla of Vater in a patient who underwent radical surgery for the carcinoma. Following tumor resection, the patient’s urinary protein turned negative, and the nephrotic syndrome was significantly relieved. It is crucial to exclude primary factors, including malignancies, in patients presenting with nephrotic syndrome, regardless of the PLA2R status in membranous nephropathy. If nephrotic syndrome is determined to be secondary to an underlying tumor, aggressive treatment of the primary malignancy is essential to prevent disease progression, and diligent postoperative surveillance is necessary to monitor for tumor recurrence.
Footnotes
Acknowledgement
The authors thank all the medical staff of the Department of Nephrology, the Central Hospital of Wuhan.
Authors’ contributions
Hua Jie and Qian Yongsheng drafted the manuscript. Chen Wenli collected the data. All authors have reviewed and approved the final manuscript.
Data availability statement
All data generated or analyzed during this case report are included in this published article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
