Abstract
Objective
Non-alcoholic fatty liver disease is a risk factor for atrial fibrillation. We assessed whether liver fibrosis markers are associated with atrial remodeling indicators in patients with atrial fibrillation in whom fatty liver disease has not been identified.
Methods
In this prospective, observational study, 174 patients with atrial fibrillation without chronic liver disease who underwent catheter ablation were enrolled. Using blood collected from the right atrium, type IV collagen (COL4), type IV collagen 7S (COL4-7S), and tumor necrosis factor-α concentrations were measured as markers of liver fibrosis and inflammation. The left atrial volume and P-wave duration were used as atrial remodeling indicators.
Results
Left atrial volume was significantly positively correlated with COL4, COL4-7S, and tumor necrosis factor-α concentrations. COL4-7S concentrations were significantly positively correlated with tumor necrosis factor-α concentrations and the P-wave duration. To exclude the effect of alcohol consumption, a multiple regression analysis was performed for left atrial volume in patients with a <30-g daily alcohol intake (n = 124). Age, sex, and COL4-7S were significant explanatory variables (R = 0.44, adjusted R2 = 0.142, COL4-7S standardized β = 0.20).
Conclusion
Liver fibrosis may be involved in atrial remodeling via inflammation in patients with atrial fibrillation who do not have obvious fatty liver disease.
Keywords
Introduction
Atrial fibrillation (AF) is one of the most common arrhythmias and is considered the result of electrophysiological and structural remodeling of the left atrium. 1 Although the risk factors for AF are diverse, non-genetic risk factors, such as obesity, smoking, alcohol consumption, and high blood pressure, are consistently associated with the occurrence of AF, regardless of age. 2 Among these, the association between AF and nonalcoholic fatty liver disease (NAFLD), a metabolic disease, has recently garnered attention.
NAFLD indicates a series of liver diseases characterized by the excessive accumulation of liver fat, excluding chronic alcohol intake, along with inflammation and fibrosis due to fat accumulation. 3 Fatty liver disease is the main cause of chronic liver disease in many developed countries, with an estimated global prevalence of approximately 30%. 4
Several epidemiological studies have suggested an association between AF and NAFLD.5 –7 NAFLD has a wide range of pathologies, such as simple steatosis, nonalcoholic steatohepatitis, cirrhosis, and hepatocellular carcinoma. Serum concentrations of type IV collagen 7S (COL4-7S), which is a marker of liver fibrosis, are elevated during the progression of liver fibrosis associated with chronic hepatitis. 8
Therefore, in this study, we focused on liver fibrosis markers in patients with AF and hypothesized that liver fibrosis is associated with atrial remodeling even in patients with AF and no apparent fatty liver disease. To examine this hypothesis, we evaluated the association between hepatic fibrosis and atrial remodeling in patients with AF by non-invasive testing.
Materials and methods
In this prospective, single-center, observational study, we recruited 200 consecutive subjects with AF who underwent catheter ablation treatment between July 2021 and November 2023. We defined patients as having paroxysmal AF (Paf) if sinus rhythm could be confirmed by a preoperative electrocardiogram (ECG). Subjects with non-paroxysmal AF (non-Paf) included those with persistent AF (lasting > 7 days but <12 months) and those with long-standing persistent AF (lasting > 12 months) if the sinus rhythm could not be confirmed. 9 The exclusion criteria were patients with a combination of chronic liver diseases, such as alcoholic liver disease and NAFLDs 10 (viral hepatitis, cirrhosis, hepatocellular carcinoma, drug-induced liver injury, hereditary liver disease, autoimmune liver disease, and primary biliary cholangitis), those who were complicated by inflammatory disease, advanced valvular disease, hyperthyroidism, or end-stage renal disease, or those on dialysis. Nine patients refused to participate, and 17 patients who met the exclusion criteria were excluded.
This study was conducted in accordance with the standards of the Declaration of Helsinki, and written informed consent was obtained from all study participants. This study was approved by the Ethics Committee of Kanazawa Medical University Hospital (Research Ethics Review Number: I609; approval date: 13 May 2021). All data were de-identified. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 11
Clinical and laboratory data (age, sex, comorbidities, smoking, alcohol intake, oral medication, body mass index, abdominal circumference, platelet count, lactate dehydrogenase, aspartate aminotransferase [AST], alanine aminotransferase [ALT], γ-glutamyl trans peptidase, cholinesterase, creatinine, estimated glomerular filtration rate, albumin, total cholesterol, triglycerides, high-density cholesterol, fasting glucose, hemoglobin A1c, fasting insulin, ferritin, and brain natriuretic peptide) were evaluated upon admission. Type IV collagen (COL4), COL4-7S, the nonalcoholic fatty liver disease fibrosis score (NFS), the fibrosis-4 (FIB-4) score, and the NAFIC (NASH, ferritin, insulin, and COL4-7S) score were used as indicators of noninvasive liver fibrosis. Tumor necrosis factor-α (TNF-α) was used as an indicator of inflammation in the body.
COL4, COL4-7S, and TNF-α concentrations were measured using blood samples obtained from the right atrium before ablation treatment, all of which were measured using commercially available kits. COL4 concentrations were measured using a latex agglutination turbidimetric immunoassay (Panassay®; Sekisui Medical Co., Tokyo, Japan). COL4-7S concentrations were measured using a chemiluminescent enzyme immunoassay (Lumipulse Presto®; Fujirebio, Tokyo, Japan). TNF-α concentrations were measured using an enzyme-linked immunosorbent assay (Human TNF-α QuantikineTM ELISA Kit; R&D Systems, Minneapolis, MN, USA). The scoring system used to evaluate liver fibrosis was calculated using the following formula:
The FIB-4 index was calculated as follows:
The NAFIC score was calculated as follows: serum ferritin concentrations ≥ 200 ng/mL (female) or ≥300 ng/mL (male), 1 point; serum fasting insulin concentrations ≥10 µU/mL, 1 point; and serum COL4-7S concentrations ≥5.0 ng/mL, 2 points.
According to accumulating evidence, a high cutoff was 2.67 for the FIB-4 index and 0.676 for the NFS, while a low cutoff was 1.30 for the FIB-4 index and −1.455 for the NFS. 12 The cutoff value of the NAFIC score was ≥2 points. 13
The P-wave duration (PWD), left atrial diameter, and left atrial volume (LAV) were used as indicators of atrial remodeling. The PWD was measured using lead II of a 12-lead ECG recorded at the time of sinus rhythm, which was performed before ablation for Paf and after ablation for non-Paf. A postoperative ECG was obtained during hospitalization at least 24 hours after catheter ablation without oral antiarrhythmic medication. 14 The left atrial diameter was measured using transthoracic echocardiography before ablation. LAV was measured using computed tomography (CT) images obtained the day before ablation, using a 128 multidetector row CT scanner (SOMATOM Definition Flash; Siemens Healthineers, Erlangen, Germany) with ECG gating. The parameters of CT image acquisition were a tube voltage of 120 kVp, a tube current automatic exposure control system (Care Dose 4D), rotation speed of 0.28 s, pitch of 3.4, reconstruction phase of 40%, reconstruction function of I36, and slice thickness/reconstruction interval of 1.0/1.0 mm. All CT images were transferred and analyzed using a medical imaging workstation (Ziostation 2; Ziosoft, Tokyo, Japan). LAV was measured by an experienced cardiologist who was blinded to the patients’ information. The coefficient of variation of measurement reproducibility in 10 randomly extracted cases was <10%. CT values of the liver and spleen were measured to evaluate fat deposition in the liver. CT values were obtained by setting a circular region of interest in an axial section image from plain CT and using the average value within that region of interest. CT values were measured at the right and left lobes of the liver and at two sites in the spleen, and the mean values were subjected to statistical analyses. 15
Continuous variables are presented as the mean ± standard deviation (SD) unless otherwise indicated. Categorical variables are presented as absolute numbers with percentages. Regarding continuous variables, the difference within one group was evaluated using the unpaired t-test or Mann–Whitney U-test, depending on the distribution of the data (normal or non-normal). Categorical variables paired with the clinical background of each patient were compared using the chi-square test. The correlation between two variables was evaluated by calculating Spearman’s rank correlation coefficient. To examine the relationship between demographics, imaging, and laboratory variables, including COL4-7S, a univariate regression analysis was first performed, followed by a multiple regression analysis. The multiple regression analysis model used age, sex, hypertension, the estimated glomerular filtration rate, hemoglobin A1c values, and COL4-7S concentrations as the initial independent variables. To exclude the effect of alcohol, the groups were divided according to a daily alcohol intake of 30 g, and a multiple regression analysis was performed for each group. Data analyses were performed using the Statflex® software program, version 0.05 (Artech Co., Osaka, Japan). All statistical tests were bilateral, and statistical significance was set at p < 0.05.
Results
The baseline characteristics of the patients are shown in Table 1. A total of 174 patients were enrolled, of whom 100 had Paf and 74 did not have Paf. The mean (±SD) age of the patients was 68.6 ± 10.2 years old and 127 (73.0%) patients were men. The mean age and sex distributions were similar between the two groups. No patients were receiving amiodarone, which might have a potential relation with hepatic steatosis.
Baseline characteristics of the patients.
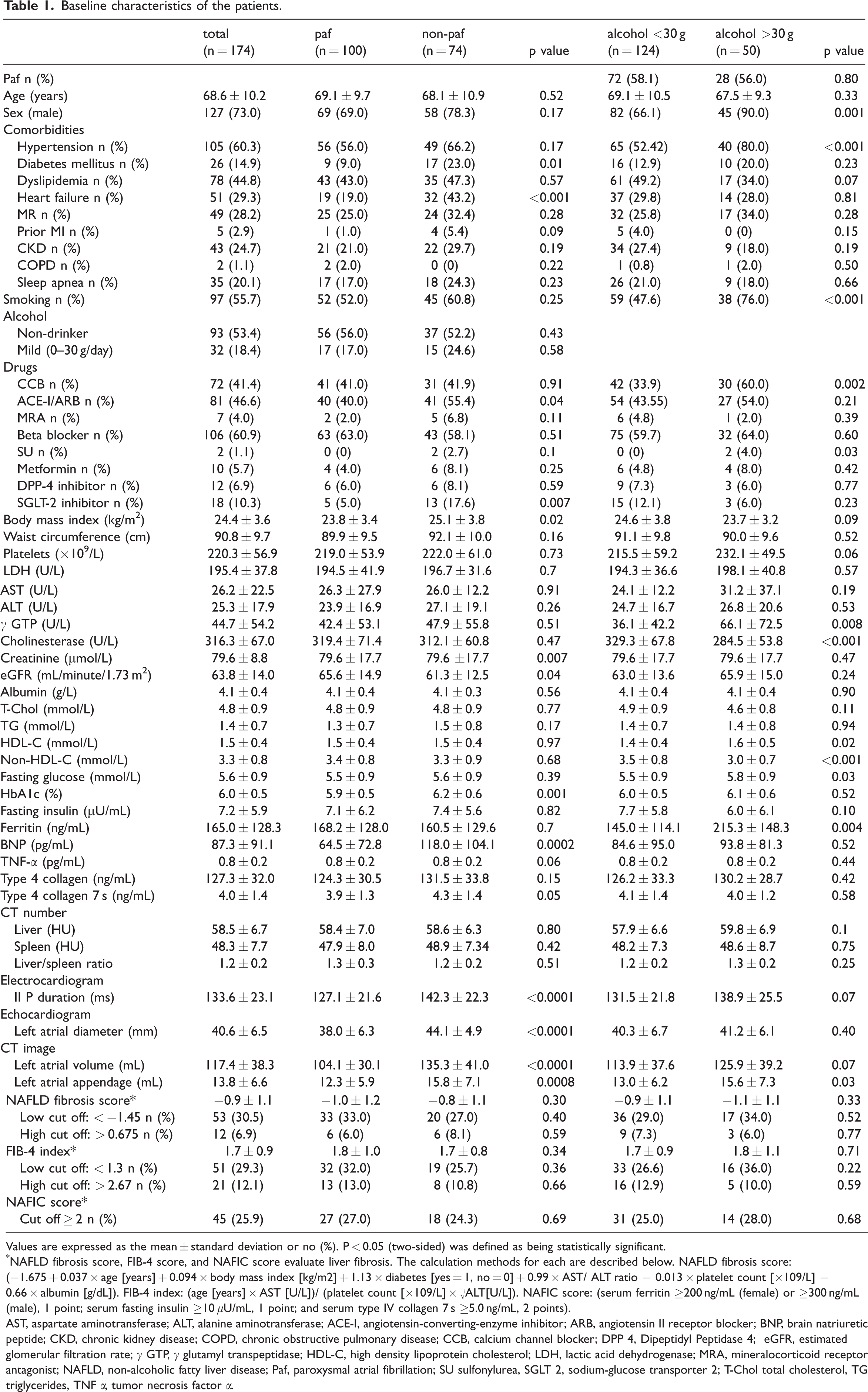
Values are expressed as the mean ± standard deviation or no (%). P < 0.05 (two-sided) was defined as being statistically significant.
NAFLD fibrosis score, FIB-4 score, and NAFIC score evaluate liver fibrosis. The calculation methods for each are described below. NAFLD fibrosis score: (−1.675 + 0.037 × age [years] + 0.094 × body mass index [kg/m2] + 1.13 × diabetes [yes = 1, no = 0] + 0.99 × AST/ ALT ratio − 0.013 × platelet count [×109/L] − 0.66 × albumin [g/dL]). FIB-4 index: (age [years] × AST [U/L])/ (platelet count [×109/L] × √ALT[U/L]). NAFIC score: (serum ferritin ≥200 ng/mL (female) or ≥300 ng/mL (male), 1 point; serum fasting insulin ≥10 μU/mL, 1 point; and serum type IV collagen 7 s ≥5.0 ng/mL, 2 points).
AST, aspartate aminotransferase; ALT, alanine aminotransferase; ACE-I, angiotensin-converting-enzyme inhibitor; ARB, angiotensin II receptor blocker; BNP, brain natriuretic peptide; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; CCB, calcium channel blocker; DPP 4, Dipeptidyl Peptidase 4; eGFR, estimated glomerular filtration rate; γ GTP, γ glutamyl transpeptidase; HDL-C, high density lipoprotein cholesterol; LDH, lactic acid dehydrogenase; MRA, mineralocorticoid receptor antagonist; NAFLD, non-alcoholic fatty liver disease; Paf, paroxysmal atrial fibrillation; SU sulfonylurea, SGLT 2, sodium-glucose transporter 2; T-Chol total cholesterol, TG triglycerides, TNF α, tumor necrosis factor α.
Initially, we divided the subjects into two groups (with or without left atrial remodeling) according to LAV measured using CT images and compared liver fibrosis-related biomarkers between the groups. Subjects with an LAV ≥ 91 mL (n = 126), which has been reported as the cutoff value for left atrial remodeling, 16 showed a significantly higher mean NFS (−0.81 ± 1.06 vs. −1.26 ± 1.27, p = 0.033), FIB-4 score (1.78 ± 0.91 vs. 1.58 ± 0.97, p = 0.025), COL4 concentration (131.11 ± 33.57 ng/mL vs. 117.4 ± 25.19 ng/mL, p = 0.004), and COL4-7S concentration (4.2 ± 1.34 ng/mL vs. 3.61 ± 1.29 ng/mL, p = 0.009) than those without remodeling with an LAV < 91 mL (n = 48). To investigate these findings further, we examined the associations between an index of left atrial remodeling (LAV) and biomarkers related to liver fibrosis. LAV was significantly positively correlated with COL4 concentrations (rS = 0.227, p = 0.003), COL4-7S concentrations (rS = 0.207, p = 0.006), the NFS (r = 0.205, p = 0.0065), and the FIB-4 score (rS = 0.159, p = 0.037) (Figure 1). In addition, LAV was significantly and positively correlated with TNF-α concentrations (rS = 0.201, p = 0.008) and the PWD (rS = 0.45, p < 0.001). A negative correlation was observed between LAV and liver CT values (r = −0.149, p = 0.05) (Figure 1).

Associations between LAV and COL4, COL4-7S, NFS, FIB-4 score, TNF-α, PWD, and liver CT values. LAV was significantly correlated with indices of liver fibrosis (a–d), inflammation (e), and electrical remodeling (f). LAV tended to be negatively correlated with liver fat deposition (g). LAV, left atrial volume; COL4, type 4 collagen; COL4-7S, type 4 collagen 7S; NFS, nonalcoholic fatty liver disease fibrosis score; PWD, P-wave duration; TNF-α, tumor necrosis factor-α.
The PWD was significantly and positively correlated with COL4-7S concentrations (rS = 0.197, p = 0.009), the NFS (r = 0.175, p = 0.021), and TNF-α concentrations (rS = 0.336, p < 0.001) (Figure 2). However, there was no significant relationships between PWD and COL4 concentrations, liver CT values, and the FIB-4 score (data not shown).
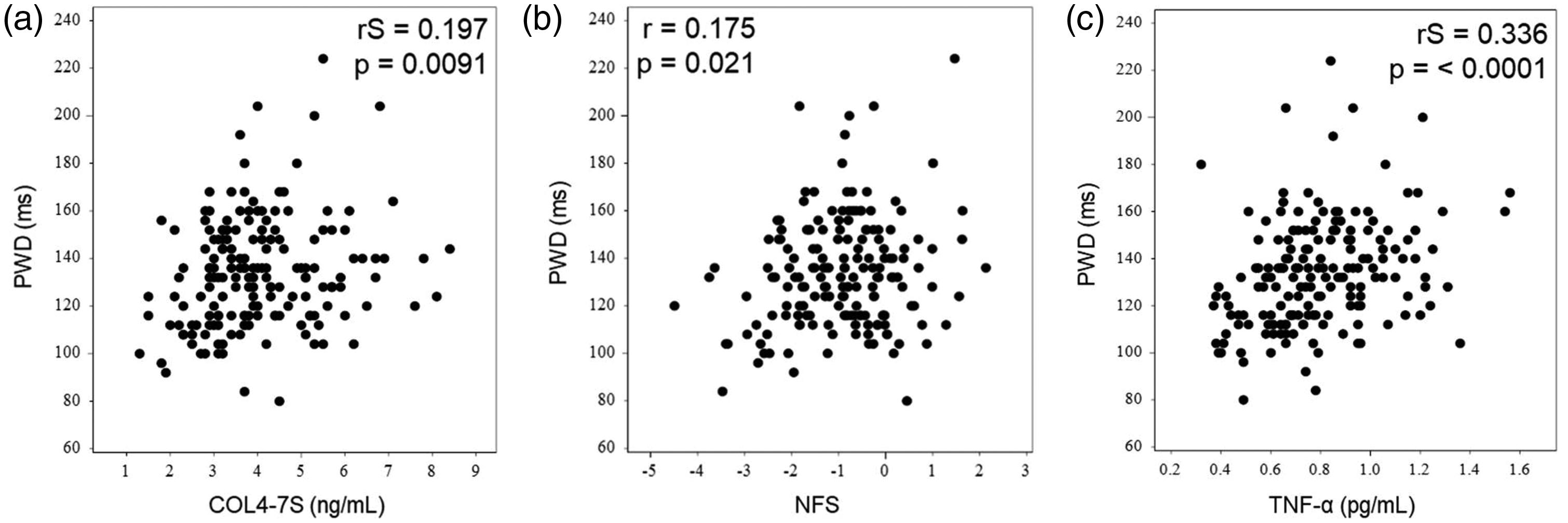
Associations between the PWD and COL4-7S, NFS, and TNF-α concentrations. The PWD was significantly correlated with indices of liver fibrosis (a, b) and inflammation (c). PWD, P-wave duration; COL4-7S, type 4 collagen 7S; NFS, nonalcoholic fatty liver disease fibrosis score; TNF-α, tumor necrosis factor-α.
COL4-7S concentrations were positively correlated with TNF-α concentrations (rS = 0.272, p < 0.001) and negatively correlated with liver CT values (rS = −0.161, p = 0.034) (Figure 3). Although COL4 concentrations were positively correlated with TNF-α concentrations (rS = 0.159, p = 0.036), the relationships between COL4 concentrations and the PWD (rS = 0.138, p = 0.069) and liver CT values were not significant (Figure 3).

Associations between liver fibrosis markers (COL4-7S and COL4) and TNF-α and liver CT values. COL4-7S concentrations were correlated with inflammation (a) and liver fat accumulation (b), and these relationships were stronger than those for COL4 (c, d). TNF-α, tumor necrosis factor-α; COL4, type 4 collagen; COL4-7S, type 4 collagen 7S.
To confirm the potential role of COL4-7S in left atrial remodeling, we divided the patients into two groups according to the median COL4-7S value (3.9 ng/mL). In subjects with higher COL4-7S concentrations (n = 84), the mean LAV was significantly larger (124.1 ± 36.58 mL vs. 111.6 ± 27.26 mL, p = 0.008) and the mean PWD was significantly wider (137.52 ± 24.02 ms vs. 124.91 ± 21.02 ms, p = 0.044) than those with lower COL4-7S concentrations.
A multiple regression analysis was performed using LAV as the objective variable. We found that age (p < 0.001) and sex (p < 0.001) were significant explanatory variables, while COL4-7S concentrations were not significant as an explanatory variable. When a similar analysis was conducted in the group with a daily alcohol intake of ≤30 g (n = 124), age, sex, and COL4-7S concentrations (all p < 0.05) were significant explanatory variables for LAV (R = 0.44, adjusted R2 = 0.142, COL4-7S Standardized β = 0.20). In the group with a daily alcohol intake of ≥30 g, body mass index was the only significant explanatory variable (p = 0.03, Table 2).
Univariate and multivariate linear regression analyses of variables related to LAV.
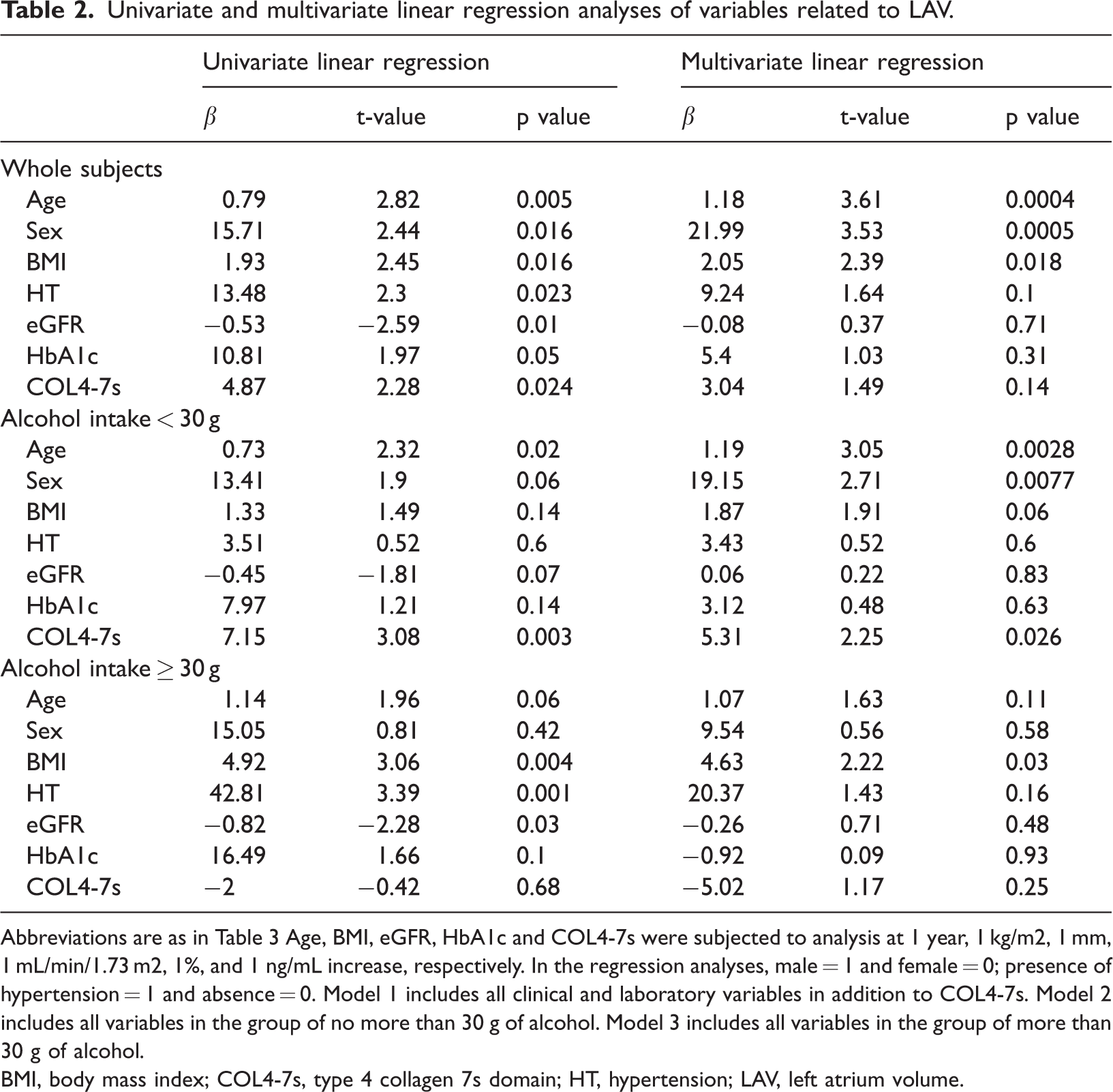
Abbreviations are as in Table 3 Age, BMI, eGFR, HbA1c and COL4-7s were subjected to analysis at 1 year, 1 kg/m2, 1 mm, 1 mL/min/1.73 m2, 1%, and 1 ng/mL increase, respectively. In the regression analyses, male = 1 and female = 0; presence of hypertension = 1 and absence = 0. Model 1 includes all clinical and laboratory variables in addition to COL4-7s. Model 2 includes all variables in the group of no more than 30 g of alcohol. Model 3 includes all variables in the group of more than 30 g of alcohol.
BMI, body mass index; COL4-7s, type 4 collagen 7s domain; HT, hypertension; LAV, left atrium volume.
Discussion
In the present study, we evaluated the relationship between hepatic fibrosis and atrial remodeling in patients with AF using non-invasive tests. TNF-α, COL4, and COL4-7S concentrations were within the reference range, with no significant liver fat deposits as shown by CT. In patients with AF, COL4 and COL4-7S concentrations were positively correlated with LAV and TNF-α concentrations. In particular, COL4-7S concentrations were positively correlated with the PWD, which is an electrical remodeling index, while they were negatively correlated with liver CT values. In the multiple regression analysis, only age and sex were significant explanatory variables overall. However, COL4-7S concentrations were a significant explanatory variable in the group with a daily alcohol intake of ≤30 g. Our finding that COL4-7S concentrations were correlated with LAV is similar to other liver fibrosis markers and scoring systems. However, our finding that COL4-7S concentrations were also correlated with liver CT values and electrical remodeling indices, such as the PWD, suggests that COL4-7S reflects left atrial remodeling. COL4-7S is also known as a marker that can sensitively detect fibrosis in NAFLD 12 and may be useful as an indicator for evaluating the relationship between AF and liver disease, even when fatty liver disease is not evident.
The relationship between AF and NAFLD has been investigated in several previous studies,5 –7 and recent meta-analyses have shown an increased risk of AF in patients with NAFLD. 7 However, the precise mechanism by which NAFLD increases the AF risk remains unclear. Although there are several possible explanations for this finding, systemic inflammation and oxidative stress may be important. 17 TNF-α is considered an endogenous mediator of inflammation and is involved in a variety of cellular processes, including activation, proliferation, inhibition of proliferation, and cell death of genes involved in inflammation and immune regulatory responses. 18 TNF-α is thought to be involved in various stages of atrial remodeling in AF 19 and has been shown to be associated with the severity of NAFLD. 20 In the present study, TNF-α concentrations were significantly and positively correlated with COL4 and COL4-7S concentrations, both markers of liver fibrosis, in addition to LAV, which is consistent with the results of previous studies.21–22
Atrial remodeling is important for the development and maintenance of AF, and electrical, contractile, and structural remodeling have been confirmed in representative models. 1 We evaluated atrial remodeling using electrical and structural indicators in the present study, and used the PWD as an index of electrical remodeling. The PWD is shortened after returning to sinus rhythm from AF until at least 24 hours have elapsed because of the recovery process of atrial electrical remodeling. 14 In our study, in the non-Paf group, postoperative ECG was obtained during hospitalization for at least 24 hours after catheter ablation. We could not detect a significant difference between the preoperative and postoperative PWD in the Paf group, which suggested that the ablation procedure itself had little effect on the PWD. Furthermore, neither the Paf nor the non-Paf group received additional linear ablation of the left atrium, which may have had a considerable effect on prolongation of the PWD. The relationship between liver fibrosis and the potential for structural and electrical reverse remodeling of the atria during long-term follow-up after AF ablation requires further investigation. In this study, COL4-7S concentrations were significantly and positively correlated with LAV and the PWD, which suggested an association between liver fibrosis and atrial remodeling. COL4 is widely distributed in the basement membrane and is nonfibrous, with COL4-7S constituting the N-terminus. Hepatocytes usually do not have a basement membrane. However, in diseased conditions, hepatocytes form a basement membrane around sinusoids. This situation increases COL4 circulation. Therefore, COL4 is used as an indicator of liver fibrosis in chronic liver diseases. 23 The accuracy of COL4-7S has been reported to be superior to that of other noninvasive markers for identifying advanced fibrosis in NAFLD. 12 In addition, COL4-7S concentrations tend to increase under conditions of heart failure or pulmonary hypertension, suggesting the involvement of hepatic congestion under these circumstances.24,25 Although an effect of hepatic congestion on COL4-7S concentrations cannot be fully excluded, inflammatory fibrosis may be more important than congestion because the patients in this study had little evidence of liver congestion.
In this study, COL4 and COL4-7S were not significant explanatory variables for LAV in the group with high alcohol intake. The reason for this finding could be due to the difference in collagen expression in alcoholic liver disease 26 along with the effect of other confounding factors such as smoking.
There are several limitations to the present study. First, a liver biopsy, which is the gold standard test for assessing liver fibrosis, was not performed. Liver biopsies are invasive and costly. Therefore, surrogate markers can be used to predict the stage of liver fibrosis. Second, some selection bias may have occurred because the study was conducted at a single center, thus potentially resulting in unrecognized bias. In particular, the lack of inclusion of obvious heart failure cases may have introduced bias in atrial volume. In contrast, the absence of cases treated for heart failure with drugs such as angiotensin receptor-neprilysin inhibitors, which can cause atrial reverse remodeling, may have facilitated the observation of the effect of liver fibrosis on atrial volume. Third, sampling bias may have existed because this study enrolled only patients scheduled for catheter ablation, and all patients with AF received one of the anticoagulants that are metabolized by several drug-metabolizing enzymes, including the cytochrome P450 mechanism. The potential effect of drugs in causing hepatic steatosis cannot be completely excluded. Fourth, no clear standard has been established for CT evaluations of the liver. In this study, we applied the methods used in several previous studies. 27 Further studies are required to evaluate CT assessments. Finally, because this was a cross-sectional study, we were unable to establish a causal relationship between atrial remodeling and liver fibrosis. Therefore, a multi-institutional, prospective study including follow-up surveys needs to be performed.
Conclusions
Despite the above-mentioned limitations, to the best of our knowledge, this study is the first to show that serum concentrations of COL4 and COL4-7S in the right atrium are positively correlated with atrial volume and inflammatory markers in patients with AF. These findings suggest that liver fibrosis is involved in atrial remodeling through inflammation. Notably, COL4-7S concentrations are also correlated with electrical remodeling indices, suggesting its superiority as a biomarker linking liver fibrosis and AF. In this study, central venous blood from the right atrium was used. Therefore, future studies should determine the clinical significance of peripheral blood measurements.
Footnotes
Acknowledgements
A part of this study was presented at the 87th Annual Scientific Meeting of the Japanese Circulation Society (JCS2023). We would like to express our appreciation to all of the study participants and those who provided helpful suggestions and discussions throughout the study.
Authors’ contributions
Dr Kuzume: Principal investigator (conceptualization, data curation, formal data analysis/interpretation, and manuscript draft writing).
Dr Fujibayashi: Director (study supervision, conceptualization, data curation, and formal data analysis/interpretation).
Dr Tanaka, Dr Sawaguchi, Dr Ueno, Dr Fujioka, and Dr Kawai: Staff cardiologists (data curation and preliminary data analysis and interpretation).
Dr Kajinami: Supervisor (overall study supervision and manuscript editing/finalization).
Data availability statement
The dataset supporting the conclusions of this study is available upon request from the corresponding author.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported in part by a research grant for Assist KAKEN from Kanazawa Medical University (K2022-11) to KF.


