Abstract
Objective
We assessed the efficacy of fasudil hydrochloride, a Rho-kinase inhibitor, as adjunct therapy for enhancing cardiac function, managing blood sugar, and improving quality of life in patients with diabetes who have coronary heart disease (CHD) and who underwent percutaneous coronary intervention (PCI).
Methods
We conducted a randomized controlled trial including 100 patients with diabetes and CHD who underwent PCI. Participants were randomly assigned to an experimental group receiving fasudil hydrochloride plus standard therapy or a control group receiving standard therapy alone. Treatment outcomes were evaluated over 3 months, focusing on cardiac function, blood sugar levels, and quality of life across physical, social, activities of daily living, and psychological domains.
Results
The experimental group showed significant improvement in cardiac function and blood sugar control, compared with the control group. Additionally, quality of life scores were markedly higher for the experimental group in all evaluated domains.
Conclusions
Fasudil hydrochloride effectively targets endothelial dysfunction and atherosclerosis, contributing to better cardiac performance and metabolic regulation. These effects translate into improved post-PCI quality of life for patients with diabetes and CHD. As an adjunct to standard PCI therapy, fasudil hydrochloride treatment offers a promising strategy for enhancing clinical outcomes in this high-risk patient population.
Keywords
Introduction
Coronary heart disease (CHD), clinically known as coronary artery atherosclerotic heart disease, is primarily caused by pathological changes in the coronary arteries, such as the formation of atherosclerotic plaques leading to their narrowing or blockage. 1 This condition substantially reduces blood flow, resulting in myocardial ischemia and potentially myocardial infarction, where parts of the heart muscle may undergo necrosis owing to inadequate blood supply. Consequently, patients often experience chest pain, cardiac dysfunction, and, in severe cases, heart failure or sudden death. The management of CHD is further complicated when the patient also has diabetes, a chronic condition characterized by abnormal insulin secretion or action, resulting in hyperglycemia.2,3
Long-term hyperglycemia in patients with diabetes damages vascular endothelial cells, promoting atherosclerosis, which increases the risk of cardiovascular diseases, including CHD. This creates a vicious cycle where diabetes exacerbates CHD, and vice versa, complicating treatment and prognosis.4–8 Consequently, patients with both CHD and diabetes are more prone to postoperative complications and have poorer prognoses, impacting their quality of life and increasing the burden on families and society.
Percutaneous coronary intervention (PCI) has emerged as a crucial treatment for CHD. 9 This interventional procedure involves the insertion and inflation of a balloon catheter, and often a stent placement, to expand narrowed or blocked coronary arteries, thus restoring blood flow. 10 Despite its efficacy in alleviating myocardial ischemia and improving clinical symptoms, patients with concurrent diabetes who undergo PCI face higher risks of restenosis and thrombosis post-surgery, necessitating meticulous postoperative management.11–13 Fasudil hydrochloride, a selective Rho-kinase inhibitor, has shown promise in addressing these challenges. This drug inhibits platelet aggregation and reduces thrombus formation, which is particularly beneficial for patients with diabetes who exhibit increased platelet activity and coagulation tendencies.14,15 To better understand the potential benefits of fasudil hydrochloride, we explored its impact on cardiac function and quality of life in patients with CHD complicated by diabetes who underwent PCI. By comparing postoperative outcomes between patients receiving standard treatment and those receiving additional fasudil hydrochloride therapy, we aimed to provide valuable insight into optimizing postoperative care for this high-risk patient group.
Methods
Study design and patients
This study included patients diagnosed with CHD complicated by diabetes who were treated at our hospital, North China Petroleum Administration General Hospital in China, between May 2021 and August 2022. This study received approval from the Ethics Committee of North China Petroleum Administration General Hospital (approval no. ZYY-LL-2019-17; 24/06/2019). This research was conducted in accordance with the principles of the Helsinki Declaration of 1975, as revised in 2013, ensuring the protection of participants’ rights and welfare. Prior to participation, all patients were thoroughly informed about the nature and purpose of the study and provided their written informed consent. The reporting of this study conforms to the CONSORT statement. 16
Each patient provided written informed consent for the publication of their clinical information and/or clinical images. A copy of the consent form is available for review.
Eligibility criteria
Participants were selected based on well-defined inclusion and exclusion criteria to ensure a clinically relevant and homogeneous patient population, minimizing confounding factors and enhancing the study’s validity and reliability.
The inclusion criteria were eligible participants aged between 18 and 75 years, diagnosed with CHD complicated by type 2 diabetes mellitus, and having undergone PCI within the past 6 months. Clinical stability was required, with no acute cardiac events in the 3 months preceding enrollment. Additionally, participants must have provided their informed consent and demonstrated willingness to comply with all study procedures.
The exclusion criteria were the presence of severe organ dysfunction such as hepatic or renal impairment (e.g., serum creatinine exceeding 1.5 times the upper limit of normal), uncontrolled hypertension defined as systolic blood pressure above 180 mmHg or diastolic blood pressure above 110 mmHg, and significant comorbid conditions including malignancies, autoimmune diseases, or severe psychiatric disorders. Pregnancy or lactation, as well as concurrent participation in another clinical trial or any interventional study within the past 30 days, also led to exclusion. These criteria were designed to ensure the selection of patients who could benefit most from the study intervention while minimizing risks and extraneous variables that could influence the study outcomes.
Study protocol and treatment methodology
Both groups of patients with CHD complicated by diabetes received conventional treatment following PCI surgery. The conventional treatment regimen included oral administration of clopidogrel sulfate (Sanofi Hangzhou Pharmaceutical Co., Ltd., Shanghai, China; SFDA # H20130296) at a dosage of 75 mg per tablet once daily. Additionally, patients were prescribed oral rosuvastatin (AstraZeneca Pharmaceuticals, Shanghai, China; SFDA # J20170008), which was taken at a dosage of 10 mg per tablet once daily. Another component of the treatment regimen was oral metoprolol tartrate (AstraZeneca Pharmaceuticals; SFDA # H32025391), administered at a dosage of 30 mg per dose twice daily. Furthermore, the treatment included oral captopril (Shanghai Sine Promod Pharmaceutical Co., Ltd., China; SFDA # H31022986), taken at a dosage of 12.5 mg per pill twice daily. Lastly, patients were given oral aspirin (Bayer HealthCare Pharmaceuticals, Beijing, China; SFDA #J20130078), taken at a dosage of 300 mg per tablet once daily. In addition to the aforementioned conventional treatments, the experimental group received fasudil hydrochloride treatment postoperatively. Fasudil hydrochloride (30 mg/2 mL; Tianjin Hongri Pharmaceutical Co., Ltd., China; SFDA #H20040356) was administered intravenously. Each dose comprised 30 mg of fasudil hydrochloride, which was diluted in a 100 mL sodium chloride injection solution (100 mL: 0.9 g) and administered once daily. Both groups continued their respective treatment protocols for a duration of 3 months.
Evaluation of clinical efficacy
In assessing clinical efficacy of the treatments administered, we established criteria based on changes in the patients’ clinical symptoms, physical signs, electrocardiograms, and New York Heart Association (NYHA) cardiac function classification. The treatment was deemed ineffective if there was no significant change in these parameters. An effective treatment was characterized by an improvement in clinical symptoms and physical signs, a significant normalization of the electrocardiogram, and improvement of I to II levels in the NYHA classification. A highly effective treatment was indicated by a substantial disappearance of clinical symptoms and physical signs, a return to normal electrocardiogram readings, and improvement of more than II levels in the NYHA classification. The total effectiveness rate was calculated as the sum of the rates of effectiveness and high effectiveness, providing a comprehensive measure of the treatment’s overall efficacy. This method allowed us to quantitatively assess the impact of fasudil hydrochloride treatment compared with conventional therapy alone in patients with CHD complicated by diabetes post-PCI.
Blood sugar level measurement
Blood sugar levels were monitored for both patient groups before and after treatment. This monitoring included measuring fasting plasma glucose (FPG), 2-hour postprandial blood glucose (2hPG), and glycated hemoglobin (HbA1c). These measurements were crucial in evaluating the effectiveness of the treatment methods in managing diabetes. FPG and 2hPG levels are particularly useful for assessing short-term fluctuations in blood glucose whereas HbA1c levels provide a more comprehensive assessment of long-term blood sugar control. This thorough approach ensured a detailed comparison of blood sugar indicators before and after treatment, thereby offering insight into the efficacy of the intervention.
Cardiac function monitoring
Cardiac function was monitored with color Doppler echocardiography, which was performed both the day before treatment initiation and 3 months post-treatment. Measurements were acquired using the modified Simpson’s method to assess several key indices: the left ventricular end-diastolic volume index (LVEDVI), left ventricular end-systolic volume index (LVESVI), and left ventricular mass index (LVMI), which is calculated as the left ventricular mass divided by body surface area. Additionally, the left ventricular ejection fraction was evaluated. All these parameters were measured over three cardiac cycles while the patient was in sinus rhythm. In patients who presented with atrial fibrillation, the values were averaged over four cardiac cycles to ensure accuracy.
Quality of life assessment
To assess quality of life post-treatment in both groups of patients, we evaluated various domains including physical function, social function, activities of daily living, and psychological function. Each domain was scored out of a maximum of 100 points, with higher scores denoting better quality of life in the respective area. This comprehensive evaluation aimed to capture the overall impact of the treatment on patients’ well-being and everyday functioning. By comparing these scores, we aimed to determine the effectiveness of the treatments in enhancing patients’ life quality across multiple facets of daily life and psychological health.
Sample size calculation
The sample size for this study was calculated to ensure adequate power to detect clinically meaningful differences in cardiac function improvement and quality of life scores between the experimental and control groups. Using a significance level (α) of 0.05 and a test power (1−β) of 0.70, the required sample size was determined based on an expected effect size of approximately 20% to 25%. The formula used for sample size calculation was:
Statistical analysis
The statistical analysis in our study was conducted using IBM SPSS version 23.0 (IBM Corp., Armonk, NY, USA). Quantitative data are reported as mean ± standard deviation (SD) and were analyzed using the t-test to determine significant differences. For categorical data, percentages were calculated and the chi-square (χ2) test was used for comparisons. Statistical significance was set at a P-value <0.05. This rigorous statistical approach ensured the reliability and validity of our findings, facilitating a comprehensive and robust analysis of the data collected throughout the study.
Results
This study included a total of 100 patients. Patients were randomly divided into two groups, each comprising 50 individuals, using a digital random allocation method. The control group received standard treatment protocols following PCI. The experimental group received fasudil hydrochloride in addition to the standard treatment. The control group comprised 26 men and 24 women with an average age of 56.36 years (SD ± 3.26), an average body mass index (BMI) of 22.23 kg/m2 (SD ± 3.56), and an average diabetes duration of 6.58 years (SD ± 3.75). The experimental group included 25 men and 25 women with an average age of 57.23 years (SD ± 3.56), an average BMI of 23.06 kg/m2 (SD ± 2.68), and an average diabetes duration of 5.88 years (SD ± 3.79).
Fasudil hydrochloride significantly improved clinical efficacy in patients post-PCI patients
In the control group, clinical efficacy was evaluated as described, with 23 patients (46%) exhibiting significant improvement, 16 patients (32%) showing moderate improvement, and 11 patients (22%) showing no improvement. This yielded a total effectiveness rate of 78%. Conversely, the experimental group receiving fasudil hydrochloride in addition to standard treatment demonstrated superior outcomes. In this group, 26 patients (52%) showed significant improvement, 21 patients (42%) experienced moderate improvement, and only three patients (6%) had no improvement. This resulted in a total effectiveness rate of 94%. Statistical analysis confirmed that the difference in treatment effectiveness between the two groups was significant (P < 0.05), indicating that the addition of fasudil hydrochloride substantially enhanced clinical outcomes (Table 1).
Clinical efficacy outcomes in the control and experimental groups following PCI and treatment with fasudil hydrochloride.
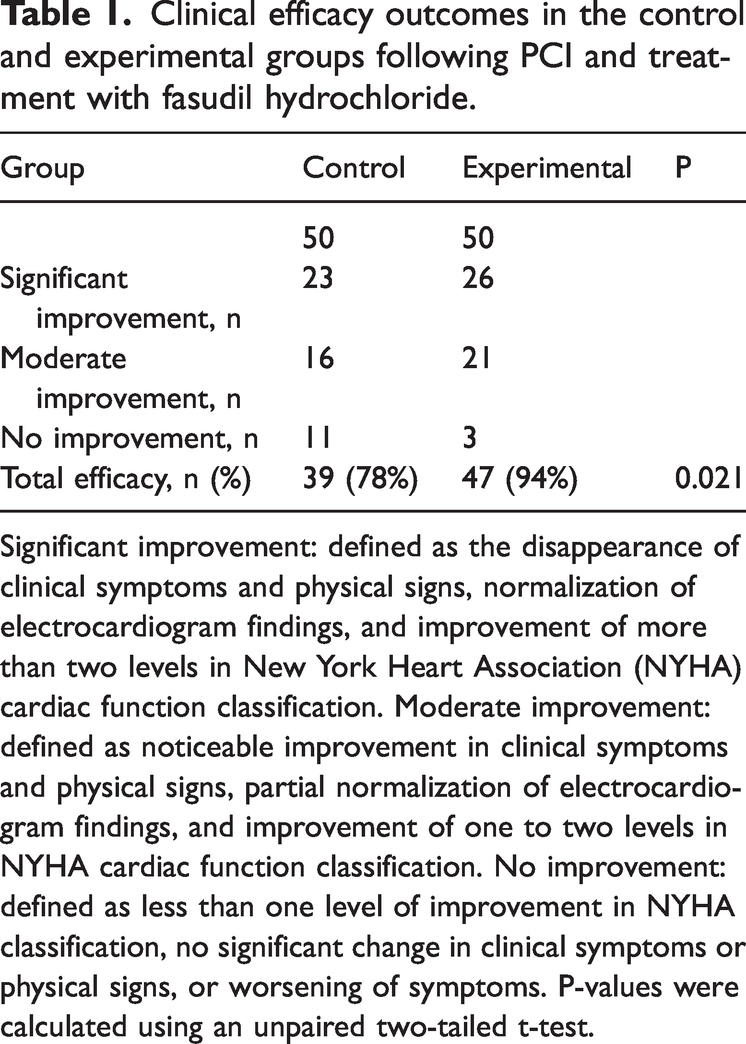
Significant improvement: defined as the disappearance of clinical symptoms and physical signs, normalization of electrocardiogram findings, and improvement of more than two levels in New York Heart Association (NYHA) cardiac function classification. Moderate improvement: defined as noticeable improvement in clinical symptoms and physical signs, partial normalization of electrocardiogram findings, and improvement of one to two levels in NYHA cardiac function classification. No improvement: defined as less than one level of improvement in NYHA classification, no significant change in clinical symptoms or physical signs, or worsening of symptoms. P-values were calculated using an unpaired two-tailed t-test.
Fasudil hydrochloride treatment improved blood sugar control after PCI
Before treatment, the blood sugar levels of patients in both groups (including FPG, 2hPG, and HbA1c) were similar; these differences were not statistically significant. After treatment, the blood sugar levels of patients in both groups were decreased, but the decrease in the experimental group was more pronounced. These post-treatment differences were statistically significant (P < 0.05), indicating that the experimental group had better blood sugar control after treatment (Table 2).
Blood glucose levels before and after treatment in the control and experimental groups.

Values are mean ± standard deviation. P-values were calculated using unpaired two-tailed t-tests.
FPG, fasting plasma glucose; 2hPG, 2-hour postprandial plasma glucose; HbA1c, glycated hemoglobin.
Fasudil hydrochloride enhanced cardiac function following PCI
Prior to treatment, both the control group and the experimental group exhibited similar cardiac function indicators, including the LVEDVI and LVESVI. Statistical analysis confirmed that these differences were not significant, indicating comparable baseline cardiac function between the groups, as shown in Table 3.
Changes in cardiac function indicators before and after treatment in the control and experimental groups (mL/m2).

Values are mean ± standard deviation. P-values were calculated using unpaired two-tailed t-tests.
LVEDVI, left ventricular end-diastolic volume index; LVESVI, left ventricular end-systolic volume index.
Post-treatment, there was a marked improvement in cardiac function indicators in both groups. However, the improvement was significantly more pronounced in the experimental group. Statistical analysis revealed that the differences between the groups post-treatment were significant (P < 0.05), suggesting that the treatment regimen followed in the experimental group had a more substantial impact on enhancing cardiac function.
Improved quality of life in patients treated with fasudil hydrochloride post-PCI
Quality of life scores across the four domains were significantly higher in the experimental group than those in the control group, as shown in Table 4. In particular, the experimental group exhibited a significantly higher mean score in physical function compared with the control group (P < 0.001), indicating better physical health and capability. The experimental group also scored significantly higher in social function (P < 0.001), reflecting better social interactions and relationships. There was a significant improvement in scores for activities of daily living for the experimental group (P < 0.001), suggesting an enhanced ability to perform daily activities. Finally, psychological function scores were notably higher in the experimental group (P < 0.001), indicating better mental and emotional well-being. The observed differences in scores were statistically significant (P < 0.001), suggesting that the administration of fasudil hydrochloride in conjunction with standard treatment significantly enhanced the quality of life in patients post-PCI.
Quality of life scores across different domains in the control and experimental groups post-PCI treatment.
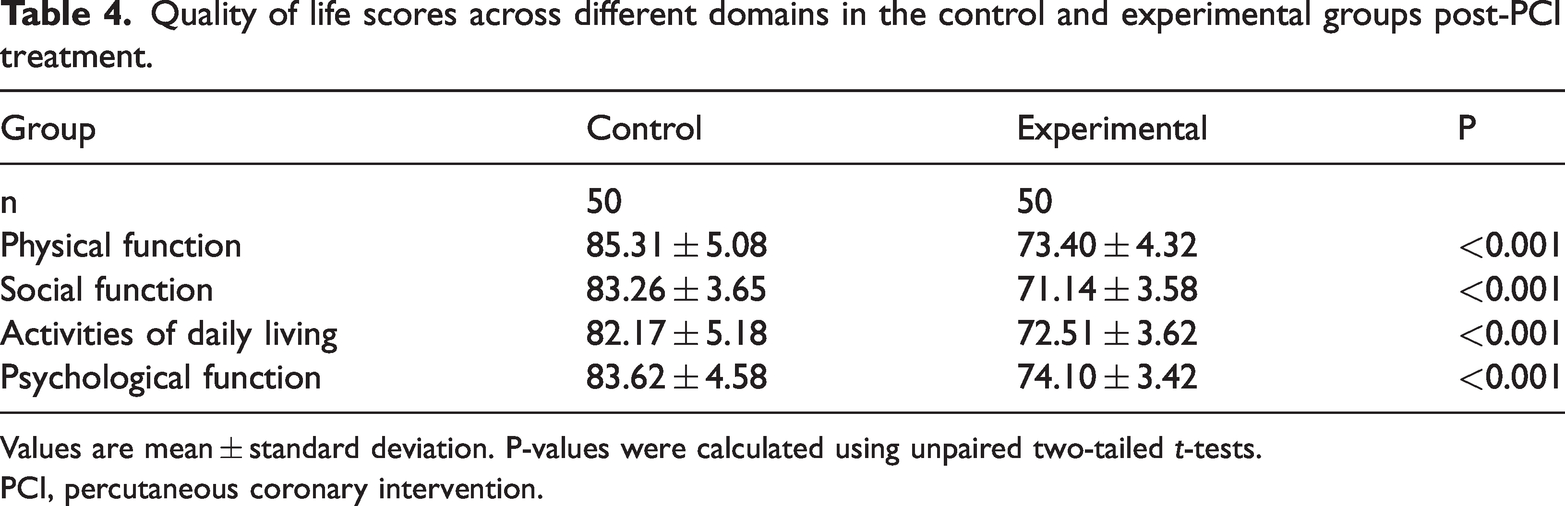
Values are mean ± standard deviation. P-values were calculated using unpaired two-tailed t-tests.
PCI, percutaneous coronary intervention.
Adverse events and safety profile
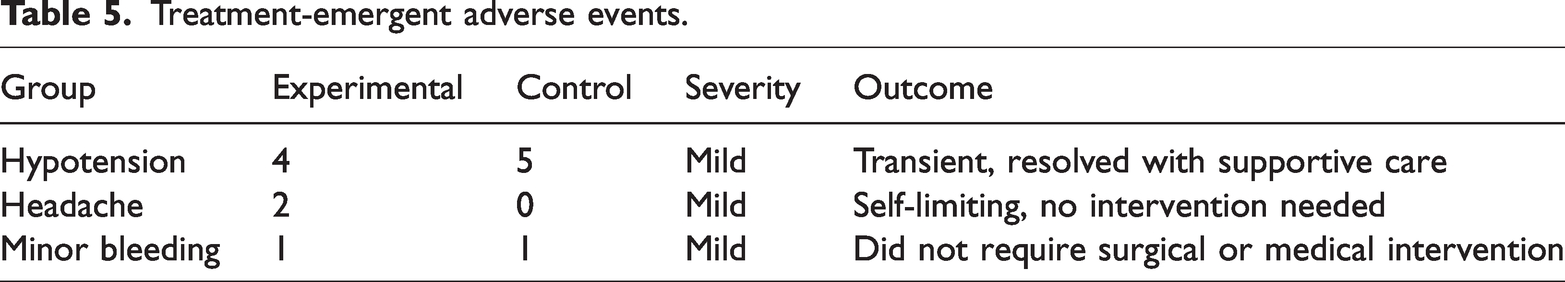
Throughout the clinical trial, adverse events were systematically monitored and recorded to ensure a comprehensive evaluation of the treatment’s safety profile, as illustrated in Table 5. Minor episodes of hypotension were observed, occurring in four patients from the experimental group and five from the control group. These events were transient and resolved promptly with supportive care, posing no long-term health risks. Two cases of headache were reported in the experimental group, both of which were associated with the administration of fasudil hydrochloride. These headaches were self-limiting and required no medical intervention. Additionally, minor bleeding complications were documented in one patient from each group, neither of which necessitated surgical intervention or further medical management. Notably, four patients in the experimental group reported chest pain, and one underwent additional PCI; however, these events were not categorized as treatment-emergent adverse events under the study protocol, which focused on events directly related to the investigational treatment. Given the high baseline cardiovascular risk of the study population and the procedural nature of PCI, these events were assessed as likely reflective of the underlying disease process or procedural outcomes rather than the treatment itself. No instances of anaphylaxis or other severe allergic reactions were reported in either group, underscoring the overall safety of the treatments. In summary, the adverse events recorded were mild and manageable, with no serious or life-threatening complications. This safety profile supports the tolerability of the intervention and highlights the importance of ongoing monitoring in clinical practice.
Treatment-emergent adverse events.
Discussion
In recent years, the incidence of diabetes has increased owing to lifestyle changes, leading to a growing population of patients with this condition. Poor diabetes control can lead to complications like CHD,17,18 often coexisting with endocrine disorders like hyperglycemia. Abnormalities in insulin and coagulation functions can cause arterial hardening or blockage of the coronary arteries. CHD combined with diabetes presents an important clinical challenge, complicating diagnosis and treatment and increasing the risk of cardiac complications. 19 PCI is a crucial treatment for CHD via the expansion of narrowed coronary arteries with a balloon catheter and stent. However, patients with CHD and diabetes face increased risk of restenosis and thrombosis post-PCI, necessitating postoperative management.11–13 Fasudil hydrochloride, a Rho-kinase inhibitor, improves blood flow and prevents thrombosis in patients with CHD. The drug works synergistically with conventional medications to enhance endothelial function and myocardial remodeling, reducing cardiovascular complications and enhancing patients’ quality of life.20–22 In this study, we investigated the effects of fasudil hydrochloride on cardiac function and quality of life in patients with CHD and diabetes undergoing PCI.
We found that the use of fasudil hydrochloride in conjunction with conventional treatment significantly improved post-PCI treatment in patients with CHD combined with diabetes. The total effectiveness rate of the experimental group was higher than that of the control group, with better cardiac function indicators, improved myocardial blood supply, enhanced patient cardiac function recovery, increased exercise endurance, and high safety levels.
Patients in the experimental group exhibited significantly better cardiac function post-treatment compared with those in the control group. This was evident from improved indicators such as the LVEDVI and LVESVI, which showed marked improvement. These results are consistent with previous studies demonstrating the effectiveness of fasudil hydrochloride in improving myocardial function and reducing cardiac remodeling in patients with cardiovascular diseases.20–22
Patients treated with fasudil hydrochloride also showed superior performance in blood sugar control, with significant reductions in FPG, 2hPG, and HbA1c levels. This aligns with existing reports highlighting the role of fasudil hydrochloride in enhancing insulin sensitivity and ameliorating glucose metabolism, which can be particularly beneficial for patients with diabetes.23,24
Finally, quality of life, assessed using various parameters such as physical function, social function, daily living, and psychological function, was deemed to be significantly better in the experimental group than that in the control group. Previous research supports these findings, indicating that fasudil hydrochloride can significantly improve patients’ quality of life by reducing cardiovascular complications and promoting better health outcomes.25,26
Despite these promising findings, our study has several limitations that warrant consideration. First, the relatively short follow-up period of 3 months may not fully capture the long-term effects and sustainability of fasudil hydrochloride on cardiac function and quality of life. Second, although sufficient power was provided for the primary outcomes, the sample size of 100 patients may limit the generalizability of the results to broader populations, including those with varying severities of CHD and diabetes. Third, this study was conducted in a single clinical center, which could introduce site-specific biases related to patient management and health care practices. Lastly, although the study used randomization, potential unmeasured confounding variables, such as adherence to prescribed therapy and lifestyle modifications, could have influenced the outcomes.
Future research should aim to address these limitations by including larger, more diverse populations and extending the follow-up period to better understand the long-term implications of fasudil hydrochloride treatment in this patient cohort. Additionally, although fasudil hydrochloride is typically administered intravenously, the availability of an oral formulation could enhance patient convenience and compliance. This aspect holds potential for future exploration, offering further clinical benefits and broadening the applicability of the treatment in everyday clinical practice.
In conclusion, fasudil hydrochloride treatment in patients with CHD combined with diabetes could more effectively control cardiac function levels, assisting in the regulation and control of diabetes blood sugar indicators, and simultaneously improving the postoperative quality of life in patients. The results of this study underscore the potential of fasudil hydrochloride as a valuable adjunctive therapy in the management of patients with these coexisting conditions, offering a promising approach to improving clinical outcomes and enhancing the quality of life for this patient population.
Footnotes
Acknowledgments
The authors would like to express their gratitude to the participants as well as the research team and colleagues for their support throughout the course of the study. We appreciate the feedback from reviewers and the editorial team.
Author contributions
The study was conceived and designed by Xuejia Yang. Data collection and analysis were conducted by Xuejia Yang, Ran Chen, Chenghui Sun, and Licha Kong. The original manuscript was drafted by Qian Li and Xuejia Yang. The final manuscript was revised by Yaning Liu and Qiuli Dong. All authors reviewed and approved the final manuscript.
Data availability statement
All relevant data from this study are not publicly available in the article. These data are part of the authors’ institutional research dataset and are subject to specific ethical and privacy constraints. We are, however, willing to provide the necessary data to qualified researchers for the verification of study results upon reasonable request and in accordance with all applicable laws and regulations. Interested researchers can request the data by contacting the corresponding author.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research was funded by the Health and Family Planning Commission of Hebei Province (Project Number: 20191171).

