Abstract
Objective
Breastfeeding is associated with improved health outcomes in infancy and throughout adulthood as breast milk encompasses diverse immune-active factors that affect the ontogeny of the immune system in breastfed (BF) infants. Nevertheless, the impact of infant feeding on the immune system is poorly understood, and a comprehensive understanding of immune system development in human infants is lacking. In this observational study, we addressed the effects of different infant feeding approaches on cell populations and parameters in the peripheral blood of infants to gain insight into the innate and adaptive arms of the immune system.
Methods
Using flowcytometric analysis, we performed complete blood counts and immunoprofiling of peripheral blood collected from BF and formula-fed (FF) infants at different ages.
Results
Our results showed that the blood of BF infants had a higher frequency of leukocytes and erythrocytes in early infancy. The hemoglobin concentration was enhanced in BF infants. However, the platelet count was comparable regardless of feeding regimen.
Conclusions
We observed immunophenotypic differences between the two populations of infants, mirrored by improved frequency of innate and adaptive immune cells in BF infants.
Keywords
Introduction
Breastfeeding is linked to long-term health, including lower risks of transmissible and nontransmissible diseases, and is considered an ideal method for providing key nutrients to newborns. Newborns have an underdeveloped immune system, and breastfeeding aids immune system development in early life and beyond, achieving prototypical health outcomes, especially in low-birth-weight preterm infants.1–4 Furthermore, breastfeeding confers protection against infections and probably against allergies, primarily via secretory immunoglobulin A antibodies.5,6
Human milk is regarded as the biological norm for infant nutrition, and it has specific immune proteins for the development and functioning of the immune system. Breast milk includes various bioactive factors such as hormones, cytokines, leukocytes, inflammatory elements, immunoglobulins, lactoferrin, lysozymes, antimicrobial peptides, prebiotics and probiotics, antioxidants, stem cells, human milk oligosaccharides, microbiota, and microRNAs.7–13
Breast milk is considered a communication vehicle between the maternal immune system and that of the infant. 14 The maternal transfer of immune elements via the uterus and breast milk after birth promotes ontogeny of the immune system. Upon ingestion of breast milk, the survival of cellular components of the milk and their transfer to the infant is supported by the neonatal digestive system. Infants gain natural passive immunity from milk lymphocytes from the maternal gut and upper respiratory mucosa. 15
Infants receiving human milk have milder infections than formula-fed (FF) infants 16 owing to bioactive components with a wide spectrum of biologic functions, including antimicrobial effects and immunostimulatory functions. 17 With a lack of or insufficient breast milk, artificial formula milk offers an alternative source of enteral nutrition to enhance the infant’s nutritional needs. 18 Substitution of human milk with formula milk can lead to immune response insufficiency and vulnerability to metabolic and immune-related pathologies in the newborn 19 and promotes substantial alteration of a healthy gut microbiome.20,21 It is documented that artificial milk does not efficiently promote innate and adaptative immune activation. 19
Infant formula is a commercial alternative to breastfeeding, which is partially or completely substituted. Formula milk can be grouped into infant formula, follow-up formula, and toddler formula. Formula milk is considered a processed food item composed of milk proteins (e.g., whey protein isolate, skim milk powder, whey protein concentrate), carbohydrates (e.g., lactose or other sugars), fats (e.g., vegetable oils or animal milk fats), micronutrients (e.g., vitamins and minerals), and other additives.22–25
Stemming from the consensus that formula milk lacks certain nutritional and immunological components that are rich and diverse in human milk, our aim in this study was to address the impact of different nutritional modalities on peripheral blood cell subsets and parameters in healthy term infants at different postnatal ages.
Methods
Study participants
In this observational, case-control study investigating the weaning diet, we included healthy term infants. Infants were enlisted from the Maternal and Child Health Centers in Amman City, Jordan. All infants in the study were healthy, and their weight appropriately matched their age. Written informed consent was collected from all participating parents, in compliance with guidelines of the Ethical Committee of the Ministry of Health, Jordan. All participant details were de-identified. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 26
We divided infants selectively into four distinct postnatal age groups: 1 month, 6 months, 9 months, and 12 months. Within each age group, further division was made, based on two feeding categories: natural breastfeeding and formula feeding. We selected 7 to 10 children from each age group and feeding category. We considered the FF group to be the control group.
Ethical considerations
The study was conducted in accordance with the Declaration of Helsinki of 1975, as revised in 2013, and was approved by the ethics committee of the Faculty of Allied Medical Sciences, Al-Ahliyya Amman University, Jordan (IRB: AAU/21/11/2022-2023) and the Ministry of Health, Amman, Jordan (MOH/REC/2023/153; 10.05.2023).
Sample collection
Blood samples (2 mL) were drawn by venipuncture and stored in ethylenediaminetetraacetic acid (EDTA) in an ice box. Samples were brought to room temperature before testing. First, each sample was tested for routine hematology, and then the sample was subjected to flow cytometry analysis.
Flow cytometry analysis of whole blood composition
Concentrations of total white blood cells (WBCs) and a WBC differential (lymphocytes, monocytes, and neutrophils) were assessed for forward and side scatter characteristics using flow cytometry. An automated hematology analyzer (Celltac Alpha MEK-6500/6510K; Nihon Kohden, Tokyo, Japan) was used for the analyses.
Phenotyping using flow cytometry
Flow cytometric analysis of whole blood samples drawn from infants was performed at the Bio Cell laboratory/flow cytometry facility in Amman, Jordan. All samples were labeled within 2 hours of collection. Packed cells from whole blood (50 µL) were washed once in phosphate-buffered saline and then incubated with primary fluorochrome-labeled monoclonal antibodies for 30 minutes at 4°C (Becton Dickinson Biosciences, Franklin Lakes, NJ, USA), followed by red blood cell (RBC) lysis. Antibodies against CD3 (defining T lymphocytes), CD19 (total B lymphocytes), CD4 (defining helper T cells), CD8 (defining cytotoxic T cells), CD16 + CD56 (defining natural killer [NK] cells in the absence of CD3 co-expression), human leukocyte antigen DR (HLA-DR; defining dendritic cells, DCs), and CD14 (defining monocytes) were used in this study. The samples were processed on a MACSQuant® Analyzer 10 flow cytometer (Miltenyi Biotec, Bergisch Gladbach, Germany).
Statistical analysis
For the baseline characteristics, statistical analysis was performed using IBM SPSS version 26.0 (IBM Corp., Armonk, NY, USA). Data are presented as number and percentage for categorical variables and mean ± standard deviation (SD) for continuous variables. Comparisons of demographic data and body weight by infant age in the breastfed (BF) and FF groups was conducted using an independent samples t-test. The chi-square test was performed for categorical variables. Statistical significance was set at a P value <0.05.
For the scatter plots, data are depicted as mean ± SD. Unpaired Student t-tests were used to detect statistically significant differences between groups. A P value <0.05 was considered statistically significant. Statistical analyses and graphical presentations were completed using Graph Pad Prism, version 8.4.2 (GraphPad Software, La Jolla, CA, USA).
Results
Baseline characteristics of the study population
The baseline characteristics of the study group are presented in Table 1. In total, 80 infants divided into two groups (40 BF [23 male] infants and 40 FF [21 male] infants) were included in this case–control study. Each group was further classified into one of four groups based on the infant’s age, with a maximum of 10 infants per age group; group I was aged 1 month, group II was aged 6 months, group III was aged 9 months, and group IV was aged 12 months. Infants’ body weight was compared among the study groups (Table 1). There was no significant difference between the mean weight of BF and FF infants (6.89 ± 2.07 kg vs. 6.50 ± 2.30 kg, respectively). However, as shown in Table 1, the difference in the mean weight of infants aged 6 months was statistically significant between the BF and FF groups (6.86 ± 0.61 kg vs. 6.11 ± 0.75 kg, respectively; P = 0.025). The mean weight of infants aged 9 months was also significantly different between the BF (8.08 ± 0.62 kg) and FF (7.29 ± 0.67 kg) groups (P = 0.013). The infants’ weight was comparable among the other age groups.
Baseline characteristics of the study population.
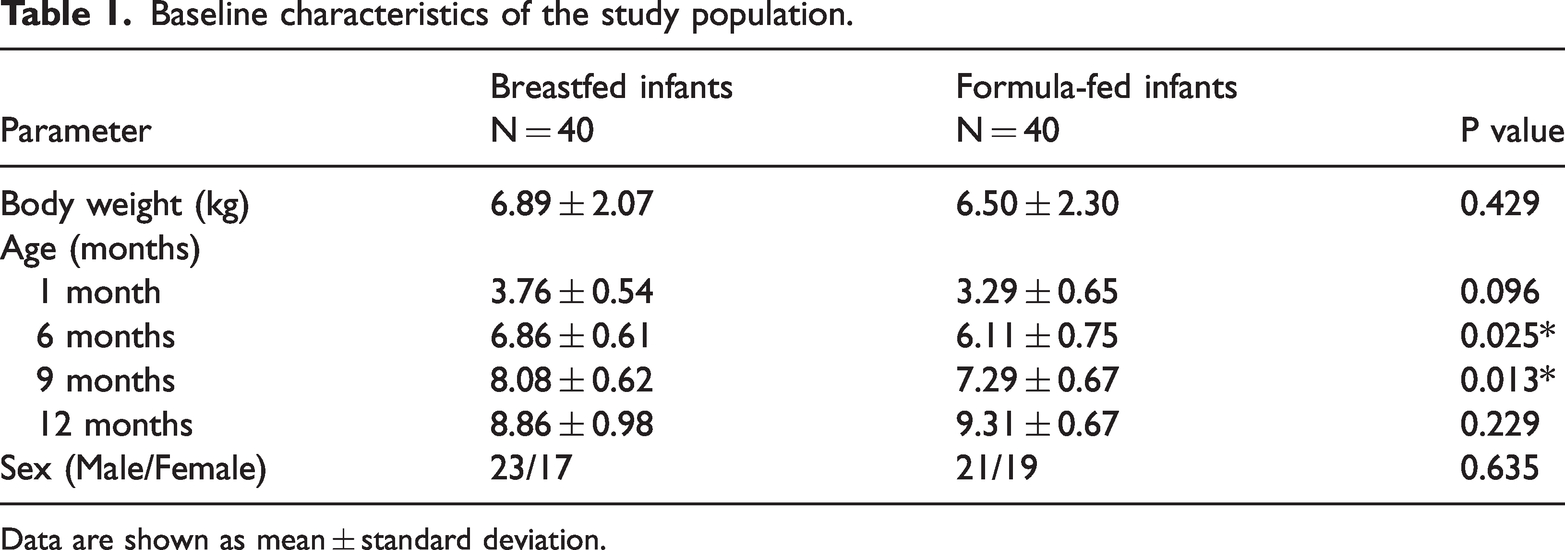
Data are shown as mean ± standard deviation.
Breastfeeding modulates complete blood count parameters
To investigate the impact of breastfeeding on the blood parameters of infants, including newborns, we carried out complete blood count analysis using blood samples from BF and FF infants at 1, 6, 9, and 12 months of age. We found that the mean concentration of hemoglobin and the packed cell volume (PCV) in BF infants was significantly higher than the hemoglobin concentration in infants receiving formula milk at age 1 month; both parameters were comparable between the two groups at age 6, 9, and 12 months (Figure 1(a), 1(b)). Consistently, the number of RBCs was higher in BF infants than in FF infants at age 1 month; this number was comparable between the two groups at age 6, 9, and 12 months (Figure 1(c)). Furthermore, the WBC count was significantly higher in BF infants than that in the FF infants at age 1 and 6 months but not at age 9 and 12 months (Figure 1(d)). Analysis of platelet counts showed that breastfeeding had no impact on the number of platelets in the BF group compared with the FF group, as demonstrated by a non-significant difference in platelets between the groups at different infant ages (Figure 1(e)).

Breastfeeding modulates infant complete blood count parameters (n = 7–10 per group). (a) Scatter plot depicting hemoglobin concentration analyzed using an automated hematology analyzer in whole blood from breastfed and formula-fed infants at different ages. (b) Packed cell volume in blood samples from the two infant groups. (c) Number of red blood cells in samples from the two infant groups. (d) Number of white blood cells in samples from the two infant groups and (e) scatter plot depicting the number of platelets in whole blood samples. Data are shown as mean ± standard deviation. Significant differences between the two groups were detected using unpaired two-tailed t-tests, as follows: ns, not significant; *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001.
Breastfeeding enhances the frequency of adaptive lymphocyte subsets
Next, to assess the effect of natural breastfeeding on cellular components of the adaptive arm of the immune system, we performed flow cytometry using whole blood collected from BF and FF infants. These blood samples were analyzed for expression of CD45 (pan-leukocyte marker), CD8, and CD4. Interestingly, we detected elevated frequencies of CD45+ cells, CD8+ T cells, and CD4+ T cells in infants who received breast milk in comparison with those who received formula milk; this significant increase was noted at age 1 and 6 months (Figure 2(a)–2(c)). Analysis of B lymphocytes (CD19 cells) revealed a higher frequency in BF infants aged 1 month but not in those aged 6 and 9 months. Interestingly, this increase in the percentage of B cells was restored at age of 12 months in BF infants (Figure 2(d)).
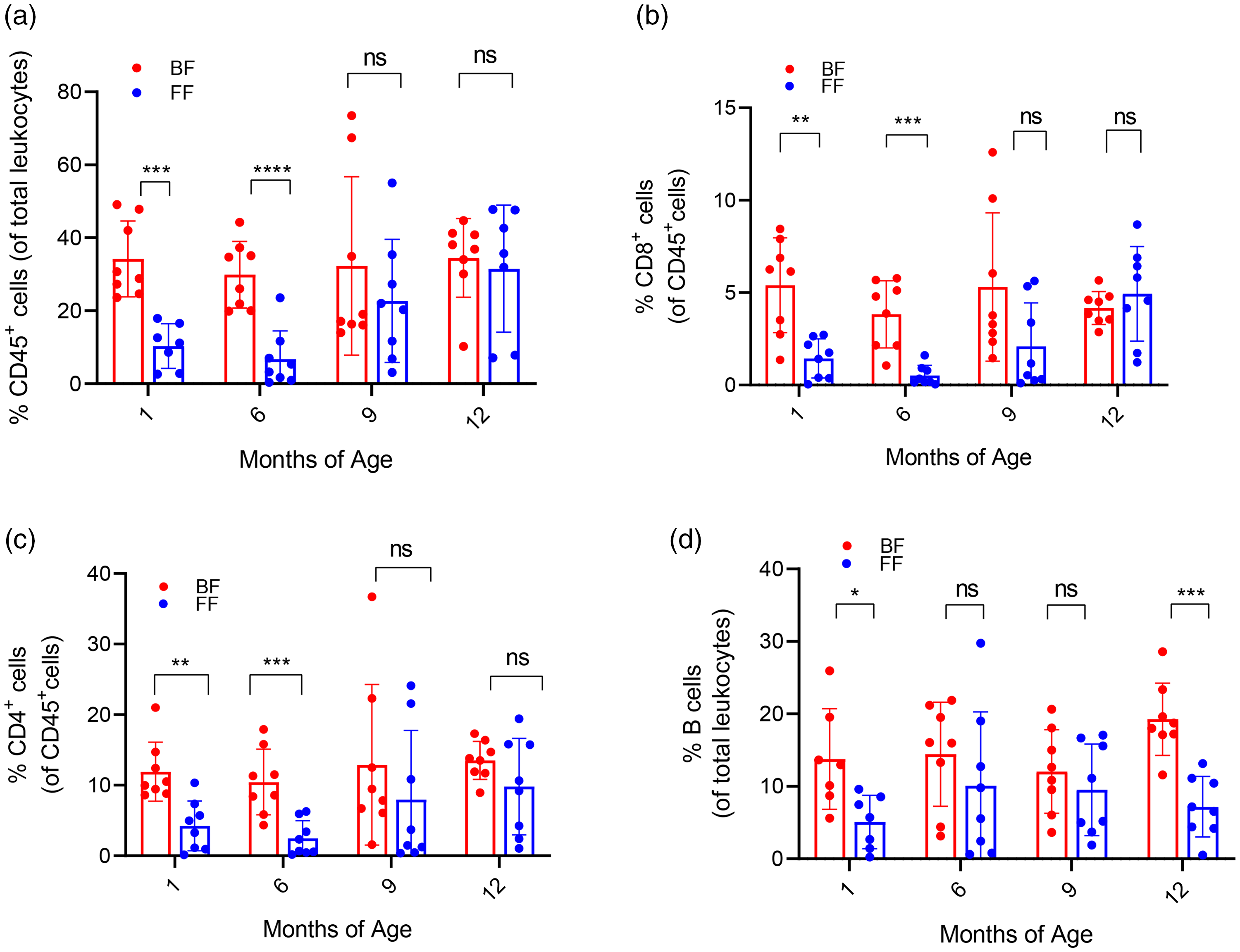
Breastfeeding enhances the frequency of adaptive lymphocyte subsets (n = 7–10 per group). (a) Scatter plot showing the frequency of cells expressing CD45 among total leukocytes after red blood cell lysis in whole blood samples from breastfed and formula-fed infants at different ages. (b) Percentage of cytotoxic T lymphocytes of total CD45 cells in blood samples from the two infant groups. (c) Percentage of helper T lymphocytes of total CD45 cells in blood samples from the two infant groups and (d) scatter plot illustrating percentage of B cells among total leukocytes in blood samples from the two infant groups. Data are shown as mean ± standard deviation. Significant differences between the groups were detected with unpaired two-tailed t-tests, as follows: ns, not significant; *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001.
Breastfeeding augments cellular components of innate immunity
To gain greater insight into the relationship between breastfeeding and selected nonspecific immune cells, we investigated the frequency of NK cells (CD 56+CD16+), monocytes (CD14+ cells), and DCs (HLA-DR+ cells) in BF infants and FF infants at different ages. We observed that the increase in the frequency of NK cells, monocytes, and DCs in BF infants was statistically significant compared with that among FF infants at age 1 month, but not at age 6 and 9 months for NK cells and monocytes. The frequency of DCs showed restored enhancement at age 12 months in BF infants (Figure 3(a)–3(c)).

Breastfeeding augments cellular components of innate immunity (n = 7–10 per group). (a) Scatter plot showing the frequency of natural killer cells among the total CD45 cells in blood samples after red blood cells lysis in whole blood samples from breastfed and formula-fed infants at different ages. (b) Shown is the percentage of monocytes among total leukocytes in blood samples from the two infant groups. (c) Scatter plot illustrating the percentage of dendritic cells among Continued.total leukocytes in blood samples from the two infant groups. Data are shown as mean ± standard deviation. Significant differences between the two groups were detected using unpaired two-tailed t-tests, as follows: ns, not significant; *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.0001.
Discussion
In the present study, we addressed the effect of breastfeeding on infants’ peripheral blood, in comparison with infants fed formula milk, at different postnatal ages. Human milk is nutritional and confers protection against diseases during and beyond the lactation period because it comprises a plethora of bioactive factors and heterogeneous populations of maternal immune cells. 27
Our results showed that breastfeeding was associated with an enhanced hemoglobin concentration and PCV as well as an increased number of RBCs at age 1 month; however, this effect was not persistent. We observed marginal increases in the aforementioned parameters at age 6 months and thereafter for infants receiving formula milk. This might be owing to low iron concentrations in human milk versus iron-fortified formula milk. However, the low concentration of iron in human milk is modestly compensated by its high bioavailability.28,29
Our findings showed an enhanced percentage of B lymphocytes in the BF group compared with the FF group. This finding is in line with a study by Carver et al. who observed a decreased B-cell count in infants who received formula milk versus those who were fed human milk. 30 Similarly, Tarcan et al found that the blood of infants fed formula exhibited reduced numbers of T cells, including CD4+ and CD8+ T cells, NK cells, and B cells, compared with infants fed human milk and fortified human milk. 1 Consistently, in vitro experiments showed that splenic cultures supplemented with colostrum or human milk enhanced B-cell proliferation and concomitant secretion of lipopolysaccharide-stimulated immunoglobulin, owing to the activation signals mediated by epidermal growth factor and interleukin 1 (IL-1) generated by macrophages. 31 In contrast, one study showed that BF and FF infants exhibited comparable numbers of B lymphocytes. 32 In another series of experiments, spontaneous and stimulated B-cell proliferation in cultured cells retrieved from newborns fed human milk and formula milk were on a par. 33 Another study showed that B lymphocyte numbers and frequencies were not affected by breastfeeding. 34
Our analysis of NK cells and T lymphocytes, including CD8 and CD4 T cells, revealed higher frequencies in the BF group than those in the FF group, indicating continuous activation of CD8 T cells owing to breast milk intake. Consistent with our study, infants who were breastfed until age 3 months had enhanced numbers of cytotoxic T cells, especially the central memory subset, in comparison with infants who were never breastfed. This change continued for 6 months of breastfeeding whereas CD4 T-cell numbers were unchanged. 35 Another study showed that memory helper T cells were not influenced by breastfeeding. 36 These observations support previous findings in which the frequencies of cytotoxic T cells are increased in BF infants.30,34,37
Breast milk is a potent source of lactoferrin and exosomes, which have immunostimulatory roles. In addition to the microbicidal function of lactoferrin, lactoferrin augments the proliferation of T cells.32,34 Further, the major histocompatibility complex content of exosomes generated in B cells and DCs stimulate T lymphocytes.38,39 Additionally, breast milk contains functional immune cells, which affect the stimulation of cytotoxic T cells.40–42
We also investigated the frequency of NK cells in the BF and FF infant groups and found that the percentage of NK cells was higher in BF infants compared with those fed formula regimens. This finding is in line with many compelling studies showing higher percentages of NK cells in BF infants.1,30,32 This enhanced activity of NK cells in infants who receive human milk is owing to lactoferrin and nucleotides in the human milk, as supported by investigations conducted in murine models.43,44 More specifically, lactoferrin was found to enhance NK cell activity. Consistently, feeding model pigs with formula milk supplemented with conjugated linoleic acid was associated with enhanced cytotoxic T- and NK-cell proliferation.45,46 This could be explained by the fact that polyunsaturated fatty acids are critically involved in the metabolic machinery of T cells. 47
Our analysis revealed that breastfeeding enhanced the percentage of DCs and monocytes in infants at age 1 month. This effect was not persistent at older ages, but we found that the enhanced percentage of DCs for BF infants was restored at age 12 months. Studies involving clinical and preclinical models have demonstrated that breast milk comprises various immune cell types typically found in the blood, such as myeloid precursor cells, DCs, and macrophages, which are transferred to the infant during breastfeeding. 48 Despite ambiguity regarding the transfer mechanism of maternal immune cells to the fetus’ blood, studies on humans have proposed that breast milk components engage with the newborn’s saliva 15 and this interaction confers protection from the acidic pH of the stomach; then, these cells infiltrate the gut mucosa and translocate to the blood circulation in the newborn. 19 The transfer of immune components from mother to infant, including monocytes, might contribute to the increased frequency of monocyte subsets in the blood of BF infants. As previously mentioned, human milk contains immune factors such as soluble CD14 (sCD14) during different times of lactation. Milk sCD14, secreted by mammary epithelial cells, includes secreted innate immunomodulators such as IL-8, which favors the proinflammatory activity of monocytes and macrophages.49,50
Our study has some limitations, including the small sample size and exclusion of the mixed feeding group from the analysis. Nevertheless, our findings suggest that babies who are fed formula milk may have different lymphocyte subset compositions and distinct complete blood cell count parameters from those fed breast milk. Our results may support the consensus that breastfeeding is a rational choice for enhancing immunity in the newborn.
Footnotes
Acknowledgements
We thank all the participants for their involvement in this study. We also thank the Stem Cell Arabia staff for their help in the flowcytometric analysis.
Author contributions
Conceptualization, T.A.H., S.A., and G.O.; methodology, T.A.H., S.A., and A.A.; software, T.A.H., S.A., and A.A.; validation, T.A.H., S.A., and K.A.A.; formal analysis, T.A.H., S.A., and K.A.A.; investigation, T.A.H., S.A., G.O., and A.A.; resources, T.A.H., S.A., and A.A.; data curation, T.A.H. and, S.A.; writing—original draft preparation, T.A.H.; writing—review and editing, T.A.H.; visualization, T.A.H.; supervision, T.A.H. and G.O.; project administration, T.A.H. and G.O. All authors have read and agreed to the published version of the manuscript.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
