Abstract
Lipoprotein(a) (Lp(a)) is an atherogenic low-density lipoprotein (LDL)-like particle that is currently regarded as a non-modifiable risk factor for atherosclerotic cardiovascular disease. The number of patients detected with elevated Lp(a) concentrations has been increasing in recent years, although the implication of this finding is unclear for patients and physicians. We screened our lipid clinic database for patients aged >65 years with very high Lp(a) concentrations, which were defined as >230 nmol/L, and cardiovascular outcomes were assessed. The patients’ (n = 16) mean (±standard deviation) age was 72.2 ± 7.1 years and the mean Lp(a) concentration was 313 ± 68 nmol/L. After a cumulative 129.0 patient-year follow-up (mean: 8.1 ± 4.2 years), the mean age was 80.3 ± 7.0 years. We observed a low baseline prevalence of cardiovascular events, with only two patients having a history of cardiovascular events. Furthermore, zero incident adverse cardiovascular events were recorded over the follow-up. Therefore, very high Lp(a) concentrations and disease-free old age are not mutually exclusive. Our aggregated clinical experience is that there is only a modest association between elevated Lp(a) concentrations and adverse outcomes. Nonetheless, we still advise treating modifiable risk factors in these patients.
Keywords
Introduction
Lipoprotein(a) (Lp(a)) is a low-density lipoprotein (LDL)-like particle in which apolipoprotein (apo) B is covalently bound to apo(a), which is a plasminogen-like molecule.1–3 Elevated plasma Lp(a) concentrations are an independent risk factor for atherosclerotic cardiovascular disease (ASCVD) 3 and aortic valve calcification. 4 Lp(a) concentrations are mainly determined by the LPA gene locus on chromosome 6q25. 5 The 2021 Canadian Cardiovascular Society (CCS) adult dyslipidemia guidelines recommend that Lp(a) concentrations should be measured once in a patient’s lifetime during initial lipid screening. 6
In primary prevention, the CCS lipid guidelines advise that Lp(a) concentrations >100 nmol/L 6 (i.e., approximately the 80th percentile for the population) is a “high risk” state that should prompt discussion of risk factor modification. 4 However, there is no universally accepted clinical threshold for an “elevated” Lp(a) concentration; the ASCVD risk gradient is curvilinear. 4 Among European individuals, the 80th, 90th, 95th, and 99th percentile thresholds for Lp(a) are >100, >200, >280, and >400 nmol/L, respectively, with corresponding odd ratios for myocardial infarction of approximately 1.3, 1.7, 2.1, and 2.8, respectively.4,7
There is growing awareness of Lp(a) owing to 2021 CCS lipid guideline recommendations and unrestricted laboratory testing in Canada. 6 Health care providers are increasingly encountering elevated Lp(a) concentrations on routine screening for primary prevention of cardiovascular risk in asymptomatic individuals. In our institution, we have noticed increasing anxiety among certain referred patients and their health care providers when an increased plasma Lp(a) concentration is discovered. How much risk high Lp(a) concentrations actually pose if other risk factors are adequately controlled is unknown. In addition, whether elevated Lp(a) concentrations are a dependable portent of increased long-term ASCVD risk and mortality is unclear. To address these issues, we performed an observational study to determine the long-term clinical course of lipid clinic patients >65 years of age with very elevated Lp(a) concentrations (>230 nmol/L). We found that elevated Lp(a) concentrations are usually not associated with adverse outcomes in older patients whose global cardiovascular risk is being managed.
Patients and methods
Since its inception >25 years previously, the Lipid Genetics Clinic, London Health Sciences Centre (London, ON, Canada) clinic has been routinely measuring Lp(a) concentrations in all patients on their first visit. The threshold for which the 2021 CCS lipid guidelines recommend intensive lifestyle modification and management of ASCVD risk factors is Lp(a) concentrations >100 nmol/L, which is approximately the 80th percentile and corresponds to an odds ratio of approximately 1.3 for ASCVD. However, to focus on cardiovascular outcomes, we focused on a subset of patients with much higher Lp(a) concentrations (i.e., >230 nmol/L), which is the 92nd percentile for the population and corresponds to an odds ratio of approximately 1.8 for ASCVD.
All data were collected from the patients’ charts. Outcome variables included cardiovascular events (i.e., myocardial infarction, acute coronary syndrome, coronary bypass surgery, or percutaneous intervention), cerebrovascular events (i.e., stroke or transient ischemic attack), and aortic valve replacement. Event history was assessed at baseline and over the follow-up.
Baseline clinical and demographic variables included age, sex, body mass index (BMI), the presence of diabetes or hypertension, the smoking status, a positive family history of early cardiovascular disease or dyslipidemia (defined as <55 and <65 years in male and female relatives, respectively), a familial history of hyperlipidemia, and medications. Biochemical data included fasting plasma concentrations of total cholesterol, triglycerides, LDL cholesterol, high-density lipoprotein (HDL) cholesterol, non-HDL cholesterol, apo B, apo A-I, and Lp(a). Lp(a) concentrations were determined nephelometrically as previously described.8,9 Statistical analyses were conducted using GraphPad Prism 10 (GraphPad Software, La Jolla, CA, USA). The reporting of this study conforms to the CARE guidelines. 10 The study was approved by Western University’s ethics review board (reference number: REB 379). All patients provided informed written consent.
Results
Patients’ demographic and clinical characteristics
To focus on ASCVD endpoints, we restricted our analysis to patients >65 years of age. Complete data were available on 16 individuals (6 men and 10 women) with Lp(a) concentrations >230 nmol/L (Table 1). The mean (±standard deviation) age was 72.2 ± 7.1 years (range: 61–86 years) and the mean BMI was 29.6 ± 7.2 kg/m2. All patients were of European ancestry. Mean baseline plasma lipid concentrations are shown in Table 1. The mean plasma Lp(a) concentration was 313 ± 68 nmol/L, which is the 98th percentile, and corresponds to an odds ratio of approximately 2.3 for ASCVD from epidemiological studies.4,7
Clinical and demographic features of the patients.

Age, duration of follow-up, and lipid values are presented as the mean ± standard deviation. Smoking status, diabetes, hypertension, and the presence of a cardiovascular or cerebrovascular event are presented as number (%.).
CABG, coronary artery bypass graft surgery; Lp(a), lipoprotein (a); LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol.
No patient had renal, liver, thyroid, or autoimmune disease. Two individuals had type 2 diabetes and were treated with metformin. Five patients had essential hypertension, which was well-controlled with monotherapy in each case. Two individuals were current smokers, three were ex-smokers for at least 1 year, and 11 were lifelong non-smokers. Eleven individuals had a positive history of cardiovascular disease in a first-degree relative without age restrictions, while six had a positive history for early cardiovascular disease in a first-degree relative (<55 and <65 years in male and female relatives, respectively). Five individuals had a positive family history of cerebrovascular disease in a first-degree relative without age restrictions. Eight individuals reported a family history of dyslipidemia and one had a known family history of elevated Lp(a) concentrations. Six individuals met the diagnostic criteria for possible or probable familial hypercholesterolemia.
At baseline, two individuals had a history of coronary artery bypass grafting secondary to unstable angina at 55 and 71 years of age. Both of these patients had minimal risk factors (i.e., no diabetes, no smoking, and well-controlled hypertension and dyslipidemia). Two other individuals had mild non-occlusive carotid plaques on ultrasound. No patients had aortic valve disease.
Follow-up
The patients were observed yearly in the clinic as part of routine care. The total cumulative follow-up was 129.0 patient-years. The individual follow-up duration was up to 18 years, with a mean follow-up of 8.1 ± 4.2 years, and the mean age at follow-up was 80.3 ± 7.0 years. Standard counselling for all patients included the following: 1) informing patients that Lp(a) is a risk factor that is not amenable to treatment; and 2) focusing on management of modifiable risk factors, including LDL cholesterol. All patients were advised to start or intensify statin therapy. Ten individuals were able to adhere to their maximally tolerated statin doses while six experienced some degree of statin intolerance. Of the six who were statin intolerant, one took ezetimibe only, one took ezetimibe plus icosapent ethyl, one took ezetimibe plus over-the-counter niacin, one took evolocumab as monotherapy, and two managed their lipids with diet and lifestyle alone. At the final follow-up appointment, the mean LDL cholesterol concentration was 3.43 ± 1.68 mmol/L, which represented a 10% decrease from baseline. Patients with hypertension or diabetes were on appropriate pharmacological management. Over the 129.0 patient-years of follow-up, there were no incident cardiovascular or cerebrovascular events. One patient died of small cell lung cancer during the follow-up.
Discussion
We studied 16 elderly patients with a mean Lp(a) concentration of 313 ± 68 nmol/L who were followed cumulatively for 129.0 patient-years (approximately 8 years) to a final age of 80.3 years. Our principal findings were as follows: 1) an older age and elevated Lp(a) concentrations were not mutually exclusive; 2) there was a low baseline prevalence of cardiovascular and cerebrovascular events, including 14 patients with primary prevention and 2 with secondary prevention, despite the mean age being in the eighth decade; and 3) there were no incident adverse cardiovascular events, particularly no coronary artery disease, cerebrovascular disease, or aortic disease, over the follow-up. All patients received goal-directed medical therapy for lipid control and risk factor modification. All patients attended appointments and followed medical advice, although several could not tolerate statin treatment. The mean reduction in LDL cholesterol concentrations at the end of follow-up was 10%. Our anecdotal findings suggest that very elevated Lp(a) concentrations can occur at an older age, and that incident cardiovascular events are not a major concern in most older patients with extremely elevated Lp(a) concentrations.
These observations reflect our overall clinical experience with measuring Lp(a) concentrations over the past 25 years. Two common clinical scenarios were found in our study. One clinical scenario is that elevated Lp(a) concentrations are occasionally observed in a subset of patients with secondary prevention, often without traditional risk factors. The other clinical scenario is that elevated Lp(a) concentrations are an incidental finding in patients with primary prevention. The 14/16 patients in our case series with very elevated plasma Lp(a) concentrations had already survived to a mean age of 72 years without any symptoms. Furthermore, after 129.0 patient-years of subsequent follow-up, there were no cardiovascular events at a mean age >80 years. Interestingly, there is generally a modest association between elevated Lp(a) concentrations and adverse clinical outcomes in the real world. When elevated Lp(a) concentrations are detected in primary or secondary prevention, our practice has been and continues to be 1) to not overreact, 2) to reassure the patient, and 3) to focus on managing modifiable risk factors such as LDL cholesterol.
These observations may appear to be counterintuitive because of the recent excitement regarding Lp(a) as a “new” risk factor.1–5 However, we believe that the following evidence supporting Lp(a) as a causal factor for vascular disease is weaker than that for LDL cholesterol. First, after >40 years of research, the exact pro-atherogenic mechanism of Lp(a) is still not definitively established.1–5 Purported ASCVD mechanisms include lipid oxidation and pro-thrombotic and pro-inflammatory effects, in addition to the effect of cholesterol carried within Lp(a) particles, but none of these have been validated beyond doubt. 5 The uncertainty of the basic science of Lp(a) is in stark contrast to the well-established mechanistic basis linking LDL with ASCVD. 11
Second, close inspection of recent genetic epidemiology and Mendelian randomization studies shows relatively marginal associations of Lp(a) concentrations with the risk of cardiovascular disease and longevity. In Danes, Lp(a) concentrations greater than the 95th percentile had a hazard ratio for cardiovascular disease of 1.8. 12 In contrast, Danish patients with LDL cholesterol concentrations in the 95th percentile had an odds ratio of cardiovascular disease of approximately 3, which rose to approximately 6 for premature cardiovascular disease compared with those with average LDL cholesterol concentrations. 13 Similar risk ratios have been found in case–control studies in which LDL cholesterol concentrations >4.9 mmol/L (i.e., 95th percentile) were associated with a relative risk ratio of approximately 6 for premature myocardial infarction compared with those with LDL-C concentrations <3.0 mmol/L. 14 This risk ratio rose further to approximately 22 in patients with a discrete molecular genetic basis such as familial hypercholesterolemia. 14 Therefore, the increment in risk for LDL cholesterol is at least two or three times larger than that for Lp(a) at comparable percentile plasma levels. In addition, a recent genome-wide meta-analysis aimed to indirectly investigate the association between Lp(a) concentrations and human longevity using LPA genetic variation as a surrogate. 15 This meta-analysis showed that the single nucleotide polymorphism rs55730499 in LPA was associated with increased Lp(a) concentrations and a decrease in lifespan by 0.7 years. Another Mendelian randomization analysis showed that the mortality risk of those with Lp(a) concentrations ≥ 95th percentile was equivalent to being approximately 1.5 years older in chronological age, but not decades older. 16 Such modest effects do not appear to justify overly aggressive intervention or counselling.
Third, genetic modelling using UK Biobank data predicts a relatively modest effect associated with a marked reduction in Lp(a) concentrations compared with a reduction in LDL cholesterol concentrations. An example of this effect is that in patients with Lp(a) concentrations >100 nmol/L, a theoretical drug that reduces Lp(a) concentrations by 80% to <20 nmol/L would have an equivalent effect on ASCVD risk reduction to that of an approximately 0.5-mmol/L reduction in LDL cholesterol concentrations (i.e., approximately half of the effect of ezetimibe). 4 In patients similar to those in the current study (i.e., Lp(a) concentrations >230 nmol/L), a theoretical drug that reduces Lp(a) concentrations by 90% to <20 nmol/L would have an equivalent effect on ASCVD risk reduction to that of an approximately 1-mmol/L reduction in LDL cholesterol concentrations (i.e., the effect of ezetimibe or a low-intensity statin). 4 Therefore, a profound reduction in Lp(a) concentrations is predicted to yield an event rate reduction of similar magnitude as that observed with a modest reduction in LDL cholesterol concentrations.
Fourth, while Lp(a) has been suggested to be similar or more pro-atherogenic than LDL, 17 the plasma concentration of Lp(a) is <1% to 10% that of LDL cholesterol concentrations in most people. 18 This finding indicates that the net contribution of all Lp(a) particles to the ASCVD process is minor compared with the huge integrated effect of total circulating LDL particles.
Fifth, studies from the 1990s have suggested that high plasma Lp(a) concentrations are compatible with longevity.19–22 In healthy French and Italian centenarians, mean Lp(a) concentrations were 30% higher than those who were >20 years younger.19–22
Sixth, the HERITAGE screening study showed that the majority of individuals with cardiovascular disease did not have elevated Lp(a) concentrations. In addition, only 25% of patients with cardiovascular disease had Lp(a) concentrations >100 nmol/L compared with 20% in controls. 23 There are high reported screening failure rates in cardiovascular outcomes in patients with secondary prevention, possibly because of the paucity of individuals with elevated Lp(a) concentrations.
Finally, the current attention on Lp(a) is similar to the situation in a generation previously when interest in Lp(a) led to a marked increase in published research, which then receded once the association between Lp(a) and atherosclerosis could not be replicated and basic science work stalled (Figure 1). Currently, there is a second wave of an increasing number of publications on Lp(a) (Figure 1). Hopefully, randomized, clinical trials of cardiovascular outcomes testing new medicines that specifically target Lp(a) and considerably reduce Lp(a) concentrations should soon definitively demonstrate whether interest in Lp(a) will continue to increase or perhaps decline again as it did 20 years previously.
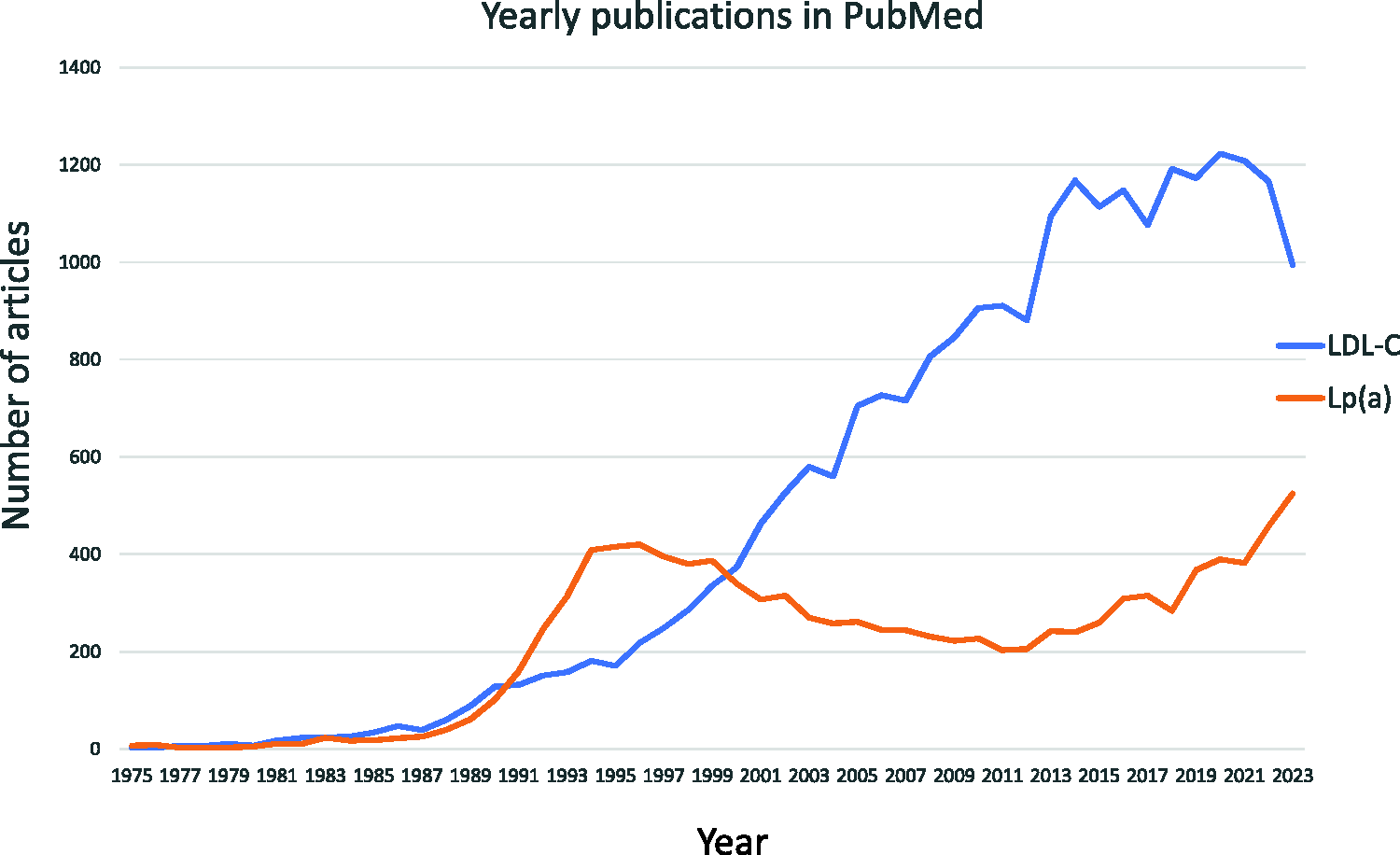
Yearly publications from 1975 to 2023 in PubMed from a search using the terms “LDL cholesterol” or “Lipoprotein(a)” and “human cardiovascular risk”. LDL-C, low-density lipoprotein cholesterol; Lp(a), lipoprotein (a).
Currently, there is no drug to specifically reduce Lp(a) concentrations. Furthermore, there is no evidence to suggest that lowering Lp(a) concentrations decreases the risk of ASCVD. Recommendations for Lp(a) concentrations >100 nmol/L in primary prevention include intensified management of modifiable risk factors. In patients with secondary prevention with elevated Lp(a) concentrations, LDL-lowering therapy should be intensified, which might also include a proprotein convertase subtilisin/kexin type 9 inhibitor. These agents lower Lp(a) concentrations by 15% to 30% in patients with baseline Lp(a) concentrations >100 nmol/L. 24 Furthermore, agents directly targeting mRNA from the LPA gene (i.e., olpasiran and pelacarsen) are being tested in randomized, controlled trials, which may yield definitive evidence of the risk associated with Lp(a) and benefits of treatment.25,26 An oral inhibitor of Lp(a) called muvalapin is also under development. 27
Limitations of our study include its observational nature, small sample size, lack of matched controls, and potential ascertainment bias for individuals who survived despite their very elevated Lp(a) concentrations. We also did not systematically use non-invasive imaging to screen patients for aortic valvular disease. Furthermore, because the patients were European, we cannot extrapolate our findings to individuals of African, Asian, or Hispanic ethnicity, which is relevant because of the known ethnic differences in Lp(a) distributions and risk thresholds (e.g., a risk threshold of 70 nmol/L has been suggested in those of African descent compared with >100 nmol/L in those of White and Hispanic descent). 28 Another technical limitation is that while Lp(a) concentrations are purported to remain stable throughout life, we recently showed intra-individual variation of up to 60% over 10 years among those with high Lp(a) concentrations. 8
Conclusion
Awareness of Lp(a) by health care providers and patients is increasing since publication of the 2021 CCS Dyslipidemia Guidelines. 6 We have noted increased anxiety associated with incidental discovery of elevated Lp(a) concentrations in some patients and clinicians. This small observational study suggests that very elevated Lp(a) concentrations are compatible with an older age and should not be a source of undue concern in an otherwise healthy patient who is diligently controlling modifiable risk factors. Even in patients with very high Lp(a) concentrations, the risk of ASCVD and aortic stenosis is relatively modest. Larger prospective, controlled studies are required to further investigate the concepts raised in this study. Until results of cardiovascular outcomes trials are available, we suggest that patients and providers who are concerned about elevated Lp(a) concentrations should redirect their energy towards intervening in proven modifiable risk factors such as LDL cholesterol.
Footnotes
Acknowledgements
We gratefully acknowledge the involvement of our clinic patients.
Author contributions
Both authors contributed to the investigation, development, and writing of this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author (R.A.H.) upon reasonable request.
Declaration of conflicting interest
R.A.H. reports consulting fees from Acasti, Aegerion, Akcea/Ionis, Amgen, Arrowhead, HLS Therapeutics, Pfizer, Novartis, Regeneron, Sanofi, and UltraGenyx. S.H. declares that there is no conflict of interest.
Funding
R.A.H. is supported by the Jacob J. Wolfe Distinguished Medical Research Chair, the Edith Schulich Vinet Research Chair in Human Genetics, and the Martha G. Blackburn Chair in Cardiovascular Research. R.A.H. has also received operating grants from the Canadian Institutes of Health Research (Foundation award) and the Heart and Stroke Foundation of Ontario.
