Abstract
Objective
We investigated the role of serum 25(OH)D (25-hydroxyvitamin D) in COVID-related health outcomes.
Methods
We conducted a retrospective cross-sectional study using data of the National Center of Disease Control and Public Health, Georgia. We extracted patient data including length of hospital stay, transfer to the intensive care unit, requirement for oxygen therapy, treatment with glucocorticoids, and symptoms. After obtaining written informed consent, 384 individuals were enrolled. We divided participants into three groups according to 25(OH)D levels: group 1 = 25(OH)D <12 ng/mL (n = 83), group 2 = 25(OH)D 12–20 ng/mL (n = 141), and group 3 = 25(OH)D >20 ng/mL (n = 160).
Results
The odds ratio (OR) for hospitalization in group 1 versus group 2 was 8.7 (95% confidence interval [CI] 3.6–21.3) and 5.6 for group 1 versus 3 (95% CI 2.7–11.9). Regarding oxygen therapy, OR = 28.41 for group 1 versus 2 (95% CI 3.7–220.5) and OR = 5.2 for group 1 versus 3 (95% CI 1.9–14.1). Regarding treatment with glucocorticoids, OR = 3.7 for group 1 versus 2 (95% CI 1.1–12.5) and OR = 8.4 for group 1 versus 3 (95% CI 1.8–40.7).
Conclusion
COVID-19-related morbidity was associated with decreased serum 25(OH)D levels. Future studies should investigate the potential role of vitamin D sufficiency in preventing SARS-CoV-2 infection and mortality.
Introduction
At the end of 2019, severe acute respiratory disease coronavirus-2 (SARS-CoV-2) spread rapidly across all continents and was declared a global pandemic by the World Health Organization (WHO) on March 11, 2020. 1 Unlike previous epidemics, SARS-CoV-2 has a higher rate of transmission and therefore poses more challenges in terms of prevention and treatment. 2 Older individuals are most susceptible to the adverse effects of COVID-19, the disease caused by SARS-CoV-2, including mortality and complications. 3 The risk is also increased owing to the presence of comorbidities such as cardiovascular diseases, diabetes, respiratory diseases, cancer, and obesity.4–6 Older patients who survive COVID-19 infection may take longer to achieve full recovery compared with other age groups. The outcome of a long illness course includes rapid loss of muscle mass after hospital discharge owing to immobilization, which can increase the risk of frailty, falls, fractures, and mortality. 7 In addition, this susceptible population is likely to be deficient in vitamin D owing with a reduced ability of the skin to synthesize vitamin D, limited sun exposure, and malabsorption. Obesity has also been associated with vitamin D deficiency owing to low vitamin D intake, poor dietary habits, and alteration of enzymes responsible for vitamin D uptake. 8 Several studies have concluded that there is a possible association between vitamin D deficiency (25(OH)D level <20 ng/mL) and susceptibility to COVID-19 infection. 9 Although the impact of vitamin D on bone metabolism has been proven, extra-skeletal effects have also been described. 10 In particular, vitamin D plays a role in inflammation processes and the modulation of innate and adaptive immunity, and it may reduce the risk of infections via several mechanisms. 11 Vitamin D supplementation reduces the risk of acute respiratory infections, including influenza, by 12% overall. Daily or weekly doses of vitamin D reduce the risk of infection by 19%. 12 Some researchers have recently confirmed these results; vitamin D supplementation reduces the risk of respiratory infections by 11%, and doses of 400 to 1000 IU/day for at least 12 months are most protective. 13
Several studies14–18 have investigated the impact of vitamin D on the risk of infection or disease outcomes of COVID-19, with controversial results. Several systematic reviews19–21 have also evaluated the relationship of COVID-19 with vitamin D levels as well as the clinical outcomes. These authors have stated that the risk of COVID-19-related hospitalization (odds ratio [OR] 1.43–1.81) and mortality (OR 1.82; 95% CI, 1.6–2.58) is increased in patients with vitamin D deficiency. On the contrary, Ferrari et al. 22 and Brandao et al. 23 rejected the evidence regarding an association between vitamin D levels and susceptibility to SARS-CoV-2 infection. Therefore, more evidence based on multicenter randomized controlled trials and large population-based studies is needed to identify the risk of COVID-19 infection in vitamin D-deficient populations.
Based on these data, the aim of our study was to investigate the causal role of serum 25(OH)D levels in COVID-related health outcomes (hospitalization, transfer to the intensive care unit [ICU], requirement for oxygen therapy, treatment with glucocorticoids) as well as the clinical characteristics (symptoms) of patients with SARS-COV-2 infection.
Methods
This study is reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 24 All procedures performed in the study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The study protocol and a draft consent agreement for participation in the study were approved by the Council on Ethical Issues in Biomedical Research of University of Georgia (#11-11477; May 15, 2023). Informed consent was obtained from all individual participants included in the study, and all participant details were de-identified.
Study design and participants
We conducted this retrospective cross-sectional study from May 2023 to February 2024 using data of the National Center of Disease Control and Public Health (NCDC) in Georgia. We randomly selected records of patients with data for serum 25(OH)D levels. The researchers conducted visits with these patients and after obtaining their written informed consent, NCDC data of the selected individuals were included in this study. The inclusion criteria were: age ≥18 years, available hospitalization data in the NCDC database, willingness to participate in the survey, and written informed consent. Exclusion criteria were: age <18 years, missing hospitalization data in the NCDC database, and refusal to participate in the survey.
Study parameters
Patient data for hospitalization, the duration of hospitalization, transfer to the ICU, a requirement for oxygen therapy, treatment with glucocorticoids, and symptoms were extracted from the NCDC database. Patients were surveyed using structured questionnaires to collect sociodemographic data, the time patients spent in the sun per day (0 or unknown, 1 hour, 1–3 hours, 3+ hours) before being diagnosed with SARS-CoV-2 infection, whether patients were taking vitamin D supplements (no, unknown, yes), and the number doses of COVID-19 vaccine received before SARS-COV-2 infection was confirmed (none, one dose, two doses, three doses).
Study groups
Study participants were divided into three groups according to serum 25(OH)D levels: 25(OH)D <12 ng/mL (group 1, vitamin D deficiency, n = 83); 25(OH)D = 12–20 ng/mL (group 2, vitamin D insufficiency, n = 141), and 25(OH)D ≥20 ng/mL (group 3, normal vitamin D levels, n = 160).
Statistical analysis
The study results were statistically analyzed using IBM SPSS 22.0 software (IBM Corp., Armonk, NY, USA). Continuous variables are expressed as mean ± standard deviation, and differences were assessed using analysis of variance. Categorical variables were compared using Pearson’s chi-square test or Fisher’s exact test. The Kolmogorov–Smirnov Z-test was used to check for the normal distribution of the obtained study data. ORs, 95% confidence intervals (CIs), and the F-test were estimated in the present study. All ORs were corrected for age, sex, and body mass index (BMI). We considered p values <0.05 to indicate statistical significance.
Results
A total of 384 individuals in the NCDC database were included in this study. Age and BMI data, as well as the distribution by sex and body weight of patients in the study, are given in Table 1. According to the distribution, there was no significant difference according to age or sex among groups. Mean BMI values also did not differ significantly among groups. However, the distribution of vitamin D levels and excess body mass showed significant differences by group. There were significantly more patients with excess body weight and obesity in the group with vitamin D deficiency (group 1) than the groups with vitamin D insufficiency (group 2) and normal values (group 3); χ = 15.459, df (degrees of freedom) = 4, p = 0.004.
Age, BMI, and distribution of patients by sex and body weight in the study groups.

SD, standard deviation; BMI, body mass index.
Participants' responses regarding the daily number of hours spent in the sun per day (by group) are given in Table 2. The groups did not differ significantly with responses of no time (or unknown time) spent in the sun per day. The odds of spending 1 hour in the sun per day for group 1 versus group 2 were OR = 4.90, 95% CI 2.72–8.85, p < 0.001; for group 1 compared with group 3, these were OR = 4.79, 95% CI 2.71–8.48, p < 0.001. The odds of spending 1 to 3 hours/day in the sun were higher for group 2 versus group 1 (OR = 3.96, 95% CI 2.20–7.12, p < 0.001) and group 3 (OR = 3.11, 95% CI 1.75–5.51, p < 0.001). Finally, the odds of spending 3+ hours/day in the sun were higher for patients in group 3 than for those in group 1 (OR = 6.80, 95% CI 1.56–29.60, p = 0.011).
Distribution of reported hours spent in the sun per day.

p1–2, p-value between groups 1 and 2; p1–3, p-value between groups 1 and 3; p2–3, p-value between groups 2 and 3.
Participants' responses regarding vitamin D supplementation before being diagnosed with SARS-COV-2 infection are given in Table 3. The distribution among groups for a response of “unknown” did not differ significantly. The odds of taking no vitamin D supplements for group 1 compared with group 2 were OR = 4.12, 95% CI 2.31–7.34, p < 0.001; for group 1 compared with group 3, these were OR = 5.78, 95% CI 3.24–10.32, p < 0.001. The odds of taking vitamin D supplements were higher for group 2 compared with group 1 (OR = 4.09, 95% CI 2.29–7.32, p < 0.001) and for group 3 versus group 1 (OR = 4.77, 95% CI 2.69–8.46, p < 0.001).
Distribution of reported vitamin D supplementation before diagnosis of SARS-CoV-2 infection.

p1–2, p-value between groups 1 and 2; p1–3, p-value between groups 1 and 3; p2–3, p-value between groups 2 and 3.
Responses regarding the number doses of COVID-19 vaccine participants received before SARS-COV-2 infection was confirmed are given in Table 4. The odds of receiving no vaccination were higher for group 1 versus group 2 (OR = 2.17, 95% CI 1.21–3.93, p < 0.001); for group 1 compared with group 3, these were OR = 2.91, 95% CI 1.64–5.19, p < 0.001.
Distribution of reported doses of COVID-19 vaccine received before diagnosis of SARS-CoV-2 infection.
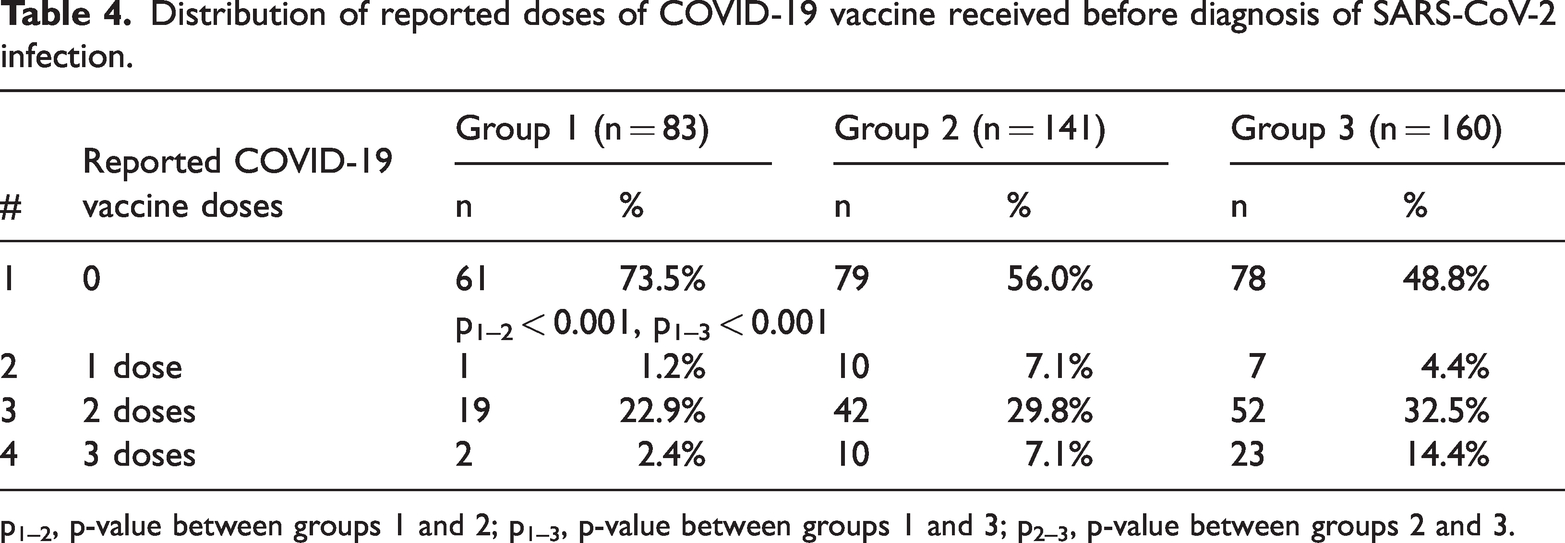
p1–2, p-value between groups 1 and 2; p1–3, p-value between groups 1 and 3; p2–3, p-value between groups 2 and 3.
Data for patients' rates of hospitalization, transfer to the ICU, the requirement for oxygen therapy, treatment with glucocorticoids, and SARS-COV-2 infection symptoms were extracted from the NCDC database (Table 5). Hospitalization was more likely in group 1 compared with group 2 (OR = 8.73, 95% CI 3.58–21.27, p < 0.001) and group 3 (OR = 5.62, 95% CI 2.66–11.90, p < 0.001). The requirement for oxygen therapy was more likely among patients in group 1 compared with those in group 2 (OR = 28.41, 95% CI 3.66–220.47, p = 0.001) and in group 3 (OR = 5.21, 95% CI 1.92–14.12, p = 0.001). Treatment with glucocorticoids was also more likely in group 1 compared with group 2 (OR = 3.65, 95% CI 1.06–12.53, p = 0.039) and group 3 (OR = 8.43, 95% CI 1.75–40.65, p = 0.008).
Distribution of patients by hospitalization rate, transfer to ICU, requirement for oxygen therapy, treatment with glucocorticoids, and symptoms of SARS-CoV-2 infection.
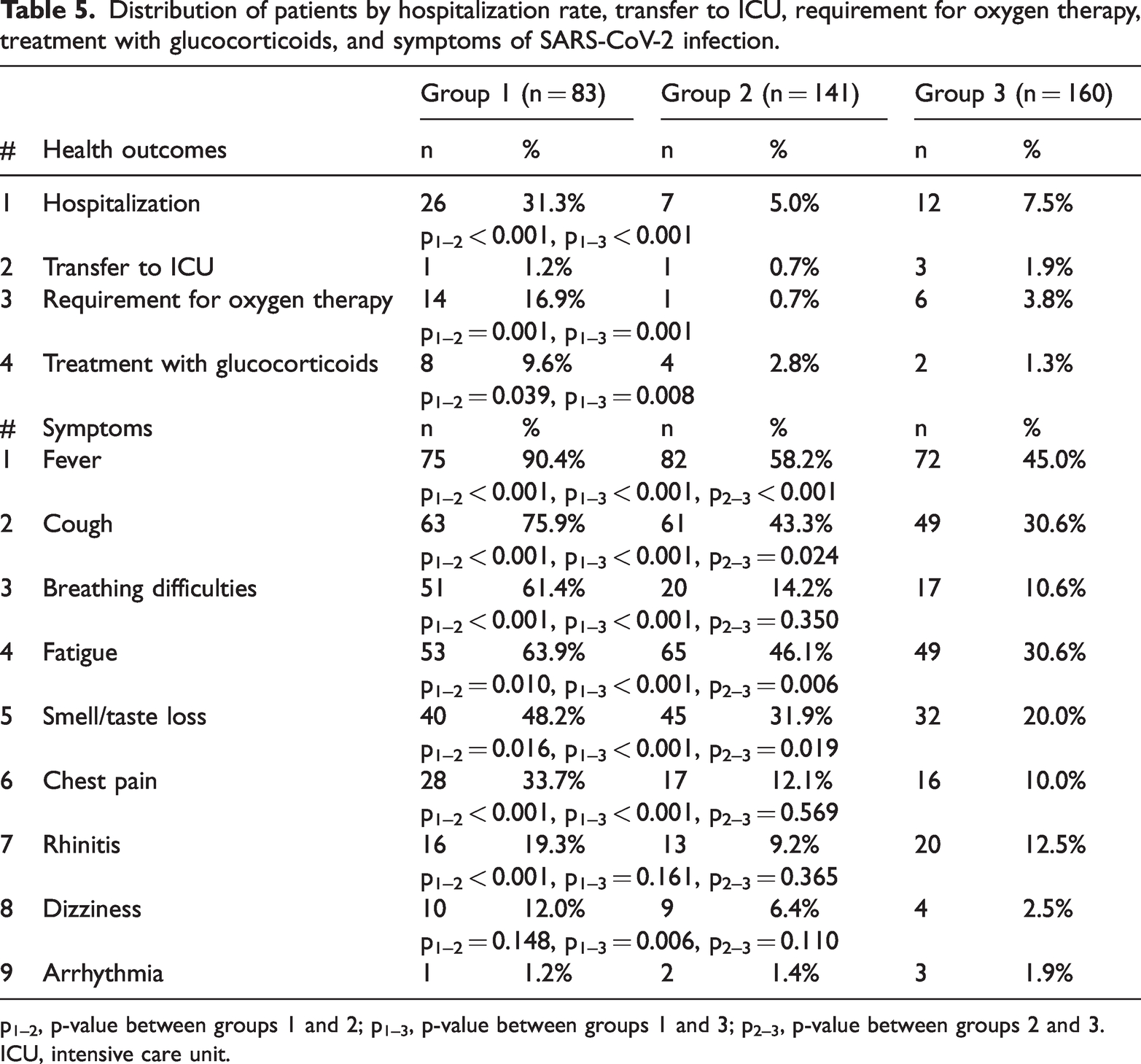
p1–2, p-value between groups 1 and 2; p1–3, p-value between groups 1 and 3; p2–3, p-value between groups 2 and 3.
ICU, intensive care unit.
The analysis of symptoms showed that the odds of fever was higher in group 1 compared with group 2 (OR = 6.75, 95% CI 3.02–15.04, p < 0.001) and group 3 (OR = 11.46, 95% CI 5.19–25.32, p < 0.001); fever was also more likely in group 2 compared with group 3 (OR = 1.73, 95% CI 1.09–2.73, p < 0.001). Cough showed higher odds in group 1 patients compared with those in group 2 (OR = 4.13, 95% CI 2.26–7.55, p < 0.001) and in group 3 (OR = 7.14, 95% CI 3.90–13.07, p < 0.001). Cough was also more likely in group 2 versus group 3 (OR = 1.73, 95% CI 1.08–2.77, p = 0.024).
Breathing difficulties were more likely in group 1 compared with group 2 (OR = 9.64, 95% CI 5.05–7.42, p < 0.001) and group 3 (OR = 13.41, 95% CI 6.86–26.19, p < 0.001). The odds of breathing difficulties were also greater in group 2 compared with group 3 (OR = 1.39, 95% CI 0.70–2.77), but this was not significant. The likelihood of having fatigue was greater in group 1 compared with group 2 (OR = 2.07, 95% CI 1.18–3.61, p = 0.010) and group 3 (OR = 4.00, 95% CI 2.29–7.01, p < 0.001) as well as greater in group 2 compared with group 3 (OR = 1.94, 95% CI 1.21–3.11, p = 0.006). The odds of losing the sense of smell or taste were higher in group 1 compared with group 2 (OR = 1.98, 95% CI 1.14–3.47, p = 0.016) and group 3 (OR = 3.72, 95% CI 2.09–6.64, p < 0.001) as well as higher in group 2 compared with group 3 (OR = 1.88, 95% CI 1.11–3.17, p = 0.019).
Chest pain was more likely among patients in group 1 compared with those in group 2 (OR = 3.71, 95% CI 1.88–7.34, p < 0.001) and group 3 (OR = 4.58, 95% CI 2.30–9.12, p < 0.001). The odds of chest pain were also higher in group 2 than in group 3 (OR = 1.23, 95% CI 0.60–2.54), but this was not significant. Rhinitis was more likely to occur in group 1 compared with group 2 (OR = 3.71, 95% CI 1.88–7.34, p < 0.001) and group 3 (OR = 1.67, 95% CI 0.81–3.43), which was not significant. However, rhinitis was more likely in group 3 compared with group 2 (OR = 1.42, 95% CI 0.67–2.94), which was not significant. Dizziness was more likely in group 1 compared with group 2 (OR = 2.01, 95% CI 0.78–5.17), without significance, as well as compared with group 3 (OR = 5.34, 95% CI 1.62–17.60, p = 0.006). The likelihood of dizziness was also higher in group 2 compared with group 3 (OR = 2.66, 95% CI 0.80–8.83), but this was not significant.
Discussion
There has been much discussion regarding the impact of vitamin D on SARS-COV-2 infection. Vitamin D may alter manifestations of the disease depending upon its influence on macrophage function and innate immunity. Vitamin D supplementation has become relevant in the absence of highly effective prevention and treatment strategies during the COVID-19 pandemic. Taking into account the availability and cost of drugs to treat COVID-19, especially in developing countries like those in Group A and B of the Research4Life program, 25 vitamin D supplementation could be an important option for those populations at risk.
Previous systematic reviews have clearly shown an inverse association between 25(OH)D levels and acute respiratory tract infections.19–21 Similar to our findings, Panagiotou et al. 26 found that low serum 25(OH)D levels in 134 hospitalized patients with COVID-19 were associated with a more severe disease course. Contrarily, in a study using data from 348,598 patients in UK Biobank, 449 (0.13%) had a diagnosis of COVID-19 infection, confirmed with a positive lab test for SARS-CoV-2; no association was found between 25(OH)D and the risk of COVID-19 infection. 27
The mean 25(OH)D levels of 154 patients in India admitted to a hospital with COVID-19 were in the insufficient range (<30 ng/mL). Patients admitted to the ICU and those who died owing to COVID-19 had lower serum levels of vitamin D than survivors. 28 Patients in Belgium with a more severe course of COVID-19-infection showed greater rates of deficient 25(OH)D levels. 29 Swiss patients with COVID-19 demonstrated significantly lower 25(OH)D levels compared with non-infected individuals. 30 The data of 7807 individuals in Israel showed that 25(OH)D levels among those who were positive for COVID-19 infection were significantly lower than those who were COVID-19 negative. 31 A Chinese study using multivariable logistic regression found that vitamin D deficiency was a significant risk factor for COVID-19 severity. 32
Vitamin D deficiency is recognized as a risk factor for COVID-19 infection, which has affected more than 1 billion people worldwide and caused approximately 3 million deaths. Vitamin D deficiency is a global problem, particularly in northern countries above the 35th parallel where people have less exposure to the sun. Our results are in agreement with those of Jain et al., 28 who reported a mean 25(OH)D level in asymptomatic patients with COVID-19 of 27.9 ± 6.2 ng/mL whereas that in patients with COVID-19 requiring ICU admission was 14.4 ± 5.8 ng/mL. In a study by Merzon et al., 31 the adjusted OR of COVID-19-related hospitalization for individuals with vitamin D deficiency versus those with normal 25(OH)D levels was 1.95 (95% CI 0.99–4.78).
To understand and improve COVID-19 outcomes, various risk scores have been developed. Among them, QCOVID 33 and OURMAPCN 34 are used to assess the risk of hospital admission and mortality owing to COVID-19. These risk scores comprise demographic data and biochemical parameters, as well as information about comorbid conditions. However, vitamin D status is not included in these algorithms. Therefore, we propose the inclusion of serum 25(OH)D levels in COVID-19 risk score algorithms.
Study limitations
We acknowledge certain limitations in our study, which are inherent to the retrospective design of the data collection and lack of availability of other confounding factors, such as comorbid illnesses, which might have increased the hospitalization rates in patients with COVID-19. Because we obtained the data of COVID-19 cases from hospital admissions registers in the NCDC database, we were unable to assess the risk of asymptomatic COVID-19 infection among individuals with vitamin D deficiency in the community. We were also unable to exclude other potential confounding factors (such as obesity) that are associated with vitamin D deficiency and greater morbidity owing to COVID-19. 35 We have no anthropometric data for patients with COVID-19 infection. The seasonal variations of vitamin D deficiency and timeline of the spread of various SARS-COV-2 strains should also be noted.
The timing of vitamin D assessment is a matter of serious debate. Because this study was retrospective, we relied on records from the NCDC in Georgia. In our study participants, vitamin D levels were determined within 2 to 3 days of confirmation of the acute phase of COVID-19. Therefore, in some cases, low levels of vitamin D may have been caused by a proinflammatory state. 36 Patients' records not including information regarding vitamin D supplementation is another limitation of the study. However, regardless of whether the phase of COVID-19 infection was acute and whether patients were taking vitamin D supplements, our findings suggest the association of low vitamin D levels with hospitalization, transfer to the ICU, and the requirement for oxygen therapy in patients with COVID-19 infection. Thus, despite the above limitations, our observational study may have important public health implications for planning and policymaking to prevent and treat COVID-19.
Conclusion
Our study findings showed that vitamin D deficiency was associated with 8.7-fold greater risk of hospitalization owing with SARS-CoV-2 infection, and vitamin D insufficiency was associated with 5.6-fold greater risk of hospitalization owing to SARS-CoV-2 infection. Therefore, COVID-19-related morbidity was associated with decreased serum 25(OH)D levels. Future studies should investigate the potential role of vitamin D levels in the prevention of SARS-CoV-2 infection and mortality. The relationship between vitamin D sufficiency and infection with various strains of SARS-CoV-2 merits special attention.
Footnotes
Acknowledgements
We appreciate the patients’ participation and the contributions of the medical staff. We thank Prof. Giorgi Kamkamidze for his critical review of the manuscript.
Author contributions
IM: conceptualization, methodology, investigation, formal analysis, writing the original manuscript draft; NB: conceptualization, investigation, writing the original manuscript draft, review and editing of the manuscript; TG: conceptualization, formal analysis, writing and editing the original manuscript draft; DT: conceptualization, formal analysis, writing and editing the original manuscript draft. All authors read and approved the submitted version of the manuscript.
Data availability statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
