Abstract
Recombinant human type II tumour necrosis factor receptor-antibody fusion protein (rh TNFR:Fc) is an immunosuppressant approved for treating rheumatoid arthritis (RA). This case report describes a case of hepatitis B reactivation in a patient with drug-induced acute-on-chronic liver failure. A 58-year-old woman with a history of RA was treated with rh TNFR:Fc; and then subsequently received 25 mg rh TNFR:Fc, twice a week, as maintenance therapy. No anti-hepatitis B virus (HBV) preventive treatment was administered. Six months later, she was hospitalized with acute jaundice. HBV reactivation was observed, leading to acute-on-chronic liver failure. After active treatment, the patient’s condition improved and she recovered well. Following careful diagnosis and treatment protocols are essential when treating RA with rh TNFR:Fc, especially in anti-hepatitis B core antigen antibody-positive patients, even when the HBV surface antigen and the HBV DNA are negative. In the case of HBV reactivation, liver function parameters, HBV surface antigen and HBV DNA should be closely monitored during treatment, and antiviral drugs should be used prophylactically when necessary, as fatal hepatitis B reactivation may occur in rare cases. A comprehensive evaluation and medication should be administered in a timely manner after evaluating the patient’s physical condition and closely monitoring the patient.
Introduction
Hepatitis B virus (HBV) causes chronic infection and is a major health problem worldwide. 1 Approximately 250 million people are currently infected with chronic HBV, a significant proportion of which are located in Asia and Africa. 2 Approximately 15–40% of infected patients develop cirrhosis, liver failure, or cancer; 3 therefore, eradicating HBV infection with existing treatments alone remains challenging. Although efficacious drugs are now available to help control this condition, HBV reactivation (HBVr) has become a challenging problem with the widespread use of immunosuppressive drugs for the treatment of various diseases. Patients with HBVr may be asymptomatic, but its presence may cause serious harm in combination with the use of immunosuppressive agents. 4 Previous reports have associated HBVr with chemotherapy in malignant tumours.5–7 Rheumatoid arthritis (RA) is a chronic autoimmune disease that mainly affects joints, leading to the infiltration of inflammatory cells in active joints and the destruction of articular cartilage. 8 Tumour necrosis factor (TNF)-α is highly expressed in the synovium of RA joints, causing synovial cells to secrete a variety of inflammatory cytokines, causing an inflammatory response, promoting the proliferation of synovial cells and causing tissue damage. 9 Therefore, inhibition of the pro-inflammatory effect of TNF-α is important for RA treatment. Recombinant human type II TNF receptor-antibody fusion protein (rh TNFR:Fc) has become a widely used drug for the treatment of RA. 10 HBVr associated with rh TNFR:Fc treatment is rare; and HBVr mostly occurs either when the recommended HBV testing is performed without adhering to the recommended monitoring guidelines (such as monitoring anti-hepatitis B surface antigen antibody [anti-HBs] levels or HBV DNA) or when no testing is performed at all.11,12 Herein, this current report describes a case of acute-on-chronic liver failure induced by rh TNFR:Fc therapy-induced occult HBV reactivation and provides a medical reference for subsequent use of drugs in patients with similar diseases.
Case report
In January 2020, a 58-year-old woman was hospitalized in the Department of Rheumatology and Immunology, Affiliated Hospital of Guizhou Medical University, Guiyang, Guizhou Province, China for RA treatment. She had a long history of diabetes mellitus (>10 years) and was using insulin Aspart and Glargine injections to keep it under control. The patient had no previous history of treatment in the Department of Infectious Diseases, Affiliated Hospital of Guizhou Medical University, Guiyang, Guizhou Province, China. However, during this current hospitalization she tested positive for anti-HBs, HBeAg antigen (anti-HBe) and hepatitis B core antigen (anti-HBc) antibodies, while testing negative for HBsAg, HBeAg and HBV DNA. The patient received rh TNFR:Fc (YISAIPU: Sansheng Guojian Pharmaceutical, Shanghai, China) for RA treatment at a dose of 25 mg administered via subcutaneous injection twice a week for 6 months. In June 2020, the patient developed severe jaundice and yellowing of the mucous membrane accompanied by upper abdominal tenderness. The patient was hospitalized at Tianzhu County People’s Hospital, Qiandongnan Miao and Dong Autonomous Prefecture, Guizhou Province, China. The medical examination revealed deranged liver function. She tested positive for HBsAg and anti-HBc antibodies and HBV DNA was 5.36 × 106 IU/ml. A coagulation function test showed the following: international normalized ratio (INR), 1.61; prothrombin time (PT), 19.53 s; alanine transaminase (ALT), 222 U/l; aspartate transaminase (AST) 312 U/l; total bilirubin (TBiL), 186.7 μmol/l; direct bilirubin (DBiL), 136.6 μmol/l; and indirect bilirubin (IBiL), 50.1 μmol/l. After symptomatic treatment with entecavir (0.5 mg/day, orally, once daily for 3 months) and ursodeoxycholic acid (500 mg/day, orally, once daily for 3 months), the patient’s bilirubin levels remained elevated as follows: TBiL, 207.1 μmol/l; DBiL, 154.0 μmol/l; and IBiL, 53.0 μmol/l.
On 13 July 2020, the patient was transferred to the Department of Infectious Diseases, Affiliated Hospital of Guizhou Medical University, Guiyang, Guizhou Province, China for further evaluation and treatment. Her blood biochemical parameters were analysed and she was screened for hepatitis B (Table 1). An abdominal computed tomography examination showed oesophagogastric varices and splenomegaly. Bacterial culture of the tip of the artificial liver catheter revealed the presence of haemolytic Staphylococcus, which was sensitive to linezolid and resistant to many other antibiotics.
Hepatitis B virus screening in a 58-year-old woman before and after treatment with recombinant human type II tumour necrosis factor receptor-antibody fusion protein (rh TNFR:Fc).

HBsAg, hepatitis B surface antigen; HBs, hepatitis B surface; HBeAg, hepatitis B e antigen; HBe, hepatitis B e; HBc, hepatitis B core; HBV, hepatitis B virus.
The patient was treated with entecavir, antiviral medications, hepatocyte growth-promoting hormone for liver protection, and drugs for regulating blood sugar and intestinal flora, as follows: entecavir capsules, 0.5 mg/day, orally, once daily for 3 months; glutathione tablets, dose total to 1200 mg per day (400 mg × 3), orally for 3 months; silymarin capsules, 70 mg, orally three times a day for 3 months; ursodeoxycholic acid, 500 mg/day, orally, once daily for 3 months; clostridium butyricum Enterococcus triple live bacterial tablets, two tablets orally, three times a day for 3 months; mendong insulin injection, subcutaneous injections of 10 units before breakfast, 14 units 30 min before lunch, and 12 units 30 min before dinner; recombinant glargine insulin injection, subcutaneous injection of 18 units before bedtime. The patient was scheduled for follow-up after 3 months. Complementary interventions, such as artificial liver plasma exchange, nutritional support and symptomatic and supportive treatment, were also administered (Figure 1). Following treatment, the patient showed improvement of symptoms and normalization of the blood biochemical parameters. No occurrence of hepatic encephalopathy was observed (Table 2). The urine output in 24 h was 1400 ml. No evident skin discoloration, with only mild icterus of the sclera, was observed. The abdomen was soft and non-tender, with no rebound tenderness. Her final diagnosis was subacute-on-chronic liver failure (ACLF), decompensated stage of hepatitis B cirrhosis and RA.
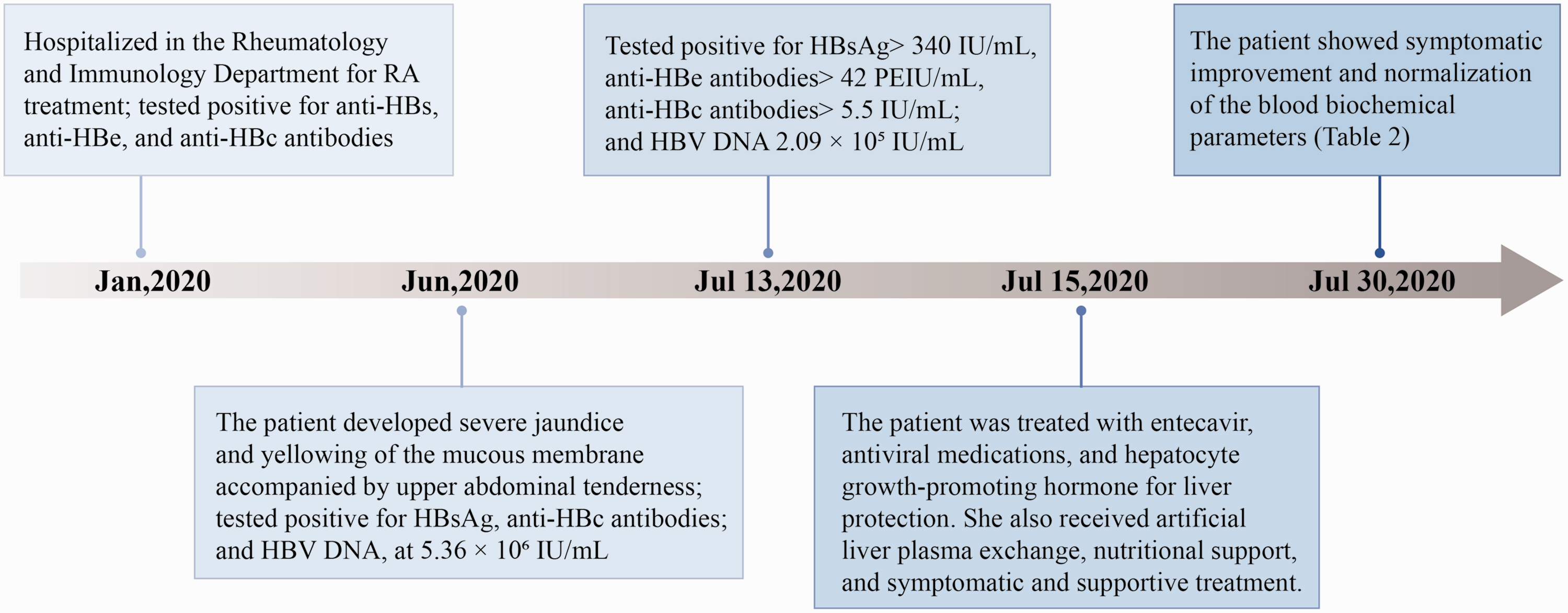
The timeline for the treatment and examination of a 58-year-old woman who was hospitalized for the treatment of rheumatoid arthritis (RA). HBs, hepatitis B surface; HBe, hepatitis B e; HBc, hepatitis B core; HBsAg, hepatitis B surface antigen; HBV, hepatitis B virus.
Changes in routine blood and biochemical indices in a 58-year-old woman before and after treatment with recombinant human type II tumour necrosis factor receptor-antibody fusion protein (rh TNFR:Fc).

RBC, red blood cell; Hb, haemoglobin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; TBiL, total bilirubin; DBiL, direct bilirubin; IBiL, indirect bilirubin; TP, total protein; ALB, albumin; TBA, total bile acid; AMM, blood ammonia.
The patient was discharged and continued to receive antiviral drugs orally: entecavir capsules 0.5 mg/day, orally, once daily for 3 months; and ursodeoxycholic acid, 500 mg/day, orally, once daily for 3 months. The patient also received diabetic medications as follows: mendong insulin injection, subcutaneous injections of 10 units before breakfast, 14 units 30 min before lunch, and 12 units 30 min before dinner; recombinant glargine insulin injection, subcutaneous injection of 18 units before bedtime. The patient was scheduled for follow-up after 3 months. The earlier administered maintenance dose of rh TNFR:Fc for RA was discontinued due to damaged liver function and the patient was advised to have a follow-up consultation in the Rheumatoid Immunology clinic after 2 weeks. The patient was closely followed up after discharge and liver function was found to be normal. HBV marker levels returned to their pre-hospitalization state. She was followed up in the Department of Rheumatology and Immunology, Affiliated Hospital of Guizhou Medical University, Guiyang for RA and her condition was successfully managed. All patient details have been removed to ensure patient privacy and written informed consent was obtained from the patient prior to treatment. In addition, the patient signed a data publishing consent form. The reporting of this study conforms to the CARE guidelines. 13
Discussion
Hepatitis B virus reactivation refers to the reappearance of HBV particles in patients with malignant tumours or other diseases combined with inactive HBV infection. 14 Glucocorticoids can bind to the HBV genome and enhance replication; immunosuppressive agents act by reducing the immune control of HBV.15,16 Subsequently, the serum HBV DNA load increases sharply, resulting in damage to liver function and liver failure in some patients. When HBVr is suspected, earlier treatments with chemotherapy, immunosuppressive or biological therapy, if any, need to be evaluated. 17 In this current case, the patient was treated with rh TNFR:Fc, which would account for the altered liver function. In rheumatic diseases, the use of immunosuppressive agents for conventional treatment induces the re-replication of HBV in the body, resulting in aggravation of hepatitis and acute liver failure. 18 HBVr occurs not only in HBsAg-positive patients, but also in HBsAg-negative patients with anti-HBc antibodies. 19 Treatment with cytotoxic or immunosuppressive drugs weakens the immune surveillance mechanisms, increasing the level of serum HBV DNA polymerase and subsequent HBV replication, thereby leading to rapid infection of normal hepatocytes. 20 ACLF is a rapidly developing decompensated disease of the liver, secondary to chronic liver disease. 21 The pathological features of ACLF are acute necrosis and apoptosis of hepatocytes accompanied by liver failure. 22 ACLF is common in chronic hepatitis B and accounts for high mortality and morbidity in the Chinese population. 23
Tumour necrosis factor-α is a naturally occurring cytokine that plays a vital role in inflammatory and immune responses. TNF-α has a direct cytotoxic effect on human hepatocytes, resulting in elevated serum transaminase and TNF-α levels in patients with fulminant liver failure. 24 The role of TNF-α in the development of RA and the resulting joint lesions has been previously reported. 25 Even though there have been few reports on the induction of HBVr by TNF-α inhibitors, its incidence is relatively high. 26 The soluble dimer rh TNFR:Fc (p75TNF receptor protein) binds to TNF-α, inhibiting the binding of TNF-α and cell surface TNFR, thereby inactivating TNF-α. Research has shown that rh TNFR:Fc has biological effects on several animal models of inflammation, including collagen-induced arthritis in mice. 27 The excessive immune cascade mediated by TNF-α may lead to hepatocyte apoptosis and damage, leading to acute liver failure. 28 The exact mechanisms of rh TNFR:Fc and HBVr are unknown; therefore, care should be taken when rh TNFR:Fc is administered to patients who have been confirmed as HBV carriers. A history of HBV infection, occult or otherwise, must be ruled out in patients requiring anti-TNF-α therapy before starting treatment.
Hepatitis B virus reactivation can be a fatal complication after systemic chemotherapy or immunosuppressive therapy. All cases of hepatitis B should be screened for HBV-related serological markers before initiating immunosuppressive therapy. Reactivation occurs not only in HBsAg-positive patients but also in anti-HBc and anti-HBs-positive (occult infections) HBsAg-negative patients. A high HBV DNA viral load and combination therapy with drugs such as immunosuppressants increase the risk of HBVr. For HBsAg-positive patients, antiviral therapy should be given for at least 6 months after completion of chemotherapy. 29 Patients with occult HBV infection (HBsAg-negative and anti-HBc-positive, but undetectable HBV DNA) should be monitored for levels of HBV DNA, ALT and AST for at least 1 year during and after chemotherapy, and antiviral therapy should be initiated after detection of HBV DNA. Even though no guidelines exist on the prophylactic use of antiviral drugs, lamivudine, entecavir or tenofovir disoproxil could be used prophylactically before receiving rh TNFR:Fc therapy. Duration of medication remains inconclusive and international guidelines for hepatitis B screening and prophylactic antiviral drugs before chemotherapy in patients with malignancy are inconsistent. In general, for serological screening, Asia-Pacific and European guidelines recommend universal screening, while US guidelines recommend a risk-stratified approach with testing in high-risk patients only.30,31
This current case report describes a patient with a history of RA treated with immunosuppressive rh TNFR:Fc, who screened positive for anti-HBc antibody, and negative for surface antigen and HBV DNA replication. Identifying HBVr requires close monitoring of the liver function, and screening for surface antigen and HBV DNA replication. Antiviral drugs should be used prophylactically when necessary. This current patient was not subjected to routine monitoring of serum HBV DNA while using rh TNFR:Fc. Consequently, the patient’s condition worsened after discharge and later presented with ACLF. This serves as a cautionary message to all clinical doctors, especially for those who use immunosuppressive drugs, that HBsAg-negative anti-HBc-positive patients need to be monitored more carefully. Important measures to prevent HBVr in high-risk groups should be taken. In this current case report, the patient was first treated in the Department of Rheumatology and Immunology for RA. The patient, who was HBsAg-negative and HBcAb-positive, developed HBVr after rh TNFR:Fc treatment, which led to liver failure. Fortunately, this was detected early and a timely diagnosis was made after the patient received treatment. Antiviral drugs are not used for routine prevention in the early stage of the disease, which reminded us that even if antiviral drugs are not used for prevention, HBsAg and HBV DNA should be assessed regularly and timely, so that disease progression can be detected at the earliest stage and for the physician to be made aware of the condition.
Research has shown that monitoring of serum HBV DNA levels can help to effectively prevent HBVr in patients with B-cell non-Hodgkin lymphoma who have previously been infected with HBV. 32 Previous studies reported that 7.3% of patients treated with anti-TNF-α therapy for inflammatory bowel disease developed liver failure due to HBVr, among which the proportion of patients that did not undergo treatment with preventive antiviral drugs was significantly higher; entecavir was shown to prevent HBVr.33,34 Another study reported that HBsAg-negative HBcAb-positive patients treated with anti- TNF-α therapy had a very low incidence of HBVr. 35 The safety of using anti-TNF-α in the treatment of RA has been confirmed, but it has also received increasing attention for HBVr. 34 HBV and hepatitis C virus infections are listed as relative contraindications in psoriasis biotherapy guidelines. 36 There have been many reports on HBVr in HBsAg-negative patients that underwent biological therapy, so HBsAg-negative patients must be carefully monitored by clinicians.37,38 Additionally, the American Society of Clinical Oncology stated in its ‘Interim Clinical Opinions’ in 2020 that for HBsAg-negative anti-HBc-positive patients, serum HBV DNA and ALT levels should be monitored regularly, and antiviral therapy should be administered immediately if HBVr occurs. 39 The Chinese guidelines for the prevention and treatment of hepatitis B also require all patients receiving radiotherapy, chemotherapy and immunosuppressive therapy for other diseases to be routinely screened for the presence of anti-HBs, anti-HBe and anti-HBc antibodies, HBsAg, HBeAg and HBV DNA before treatment. 40
This current case report emphasizes the need for a comprehensive evaluation of the case scenario before treatment initiation in patients requiring a multidisciplinary approach and a diligent follow-up of a possible HBVr following immunosuppressive medication. Although different guidelines have different guidance for diseases, anti-HBc-positive patients should be closely monitored despite HBsAg status, and antiviral drugs can be used preventively if necessary.
Footnotes
Author contributions
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This study was funded by grants from the National Natural Science Foundation of China (no. 82060123) and the Guizhou Provincial Health Commission Science and Technology Fund Project (no. gzwjk2019-1-082).
