Abstract
Paraneoplastic chronic intestinal pseudo-obstruction (CIPO) is a rare manifestation associated with small cell lung carcinoma and other malignancies. In this report, we present a challenging case involving a 60-year-old man with small cell lung carcinoma who developed symptoms of intestinal pseudo-obstruction, including abdominal distention, pain, and obstipation. Despite receiving extensive treatment with intravenous gamma globulin and glucocorticoids, the patient’s symptoms did not improve, ultimately resulting in irreversible nervous system damage. Through a comprehensive literature review, we explore the underlying mechanisms of paraneoplastic CIPO and discuss its diagnostic and therapeutic approaches. We emphasize the significance of timely detection of related antibodies for early diagnosis and treatment of CIPO to improve patient outcomes.
Keywords
Background
Paraneoplastic neurological syndromes (PNS) are rare, affecting fewer than 1% of all patients with cancer. 1 Paraneoplastic chronic intestinal pseudo-obstruction (CIPO) is a rare PNS mainly associated with small cell lung carcinoma (SCLC); however, cases of paraneoplastic CIPO have also been noted in connection with prostate cancer, extraskeletal myxoid chondrosarcoma, and thymoma.2–5 Typically, these patients have detectable onconeural autoantibodies in their bloodstream, with the predominant type being antineuronal nuclear antibody type 1 (also known as anti-Hu antibody).6,7 The emergence of paraneoplastic CIPO is attributed to humoral mediators secreted by tumor cells or an immune response targeting tumor antigens.1,8 In this report, we present a case involving a 60-year-old man who received pulse intravenous gamma globulin and glucocorticoid therapy following the diagnosis of paraneoplastic CIPO. Unfortunately, the patient’s symptoms did not improve.
Case presentation
Six months prior to presentation at our hospital, a 60-year-old man with a 40-year history of smoking was admitted to the respiratory medicine department of a local hospital because of a cough and bitter taste in the mouth. Physical examination and blood and biochemical tests showed no significant abnormalities. However, a chest computed tomography (CT) scan revealed a space-occupying lesion in the bronchus of the right middle lung lobe (Figure 1). This prompted further investigation through bronchoscopy, which revealed elevated neoplasms in the bronchi of the middle lobe. These neoplasms exhibited small nodular changes on their surface and were associated with localized bronchial stenosis (Figure 2). Biopsies were taken for pathological examination, which confirmed the diagnosis of SCLC. The pathological examination revealed positive staining for pan-cytokeratin, wild-type p53, CD56, thyroid transcription factor 1, synaptophysin, and chromogranin A as well as Ki67 positivity of 90%. Negative staining was observed for p63, p40, and leukocyte common antigen (Figure 3).
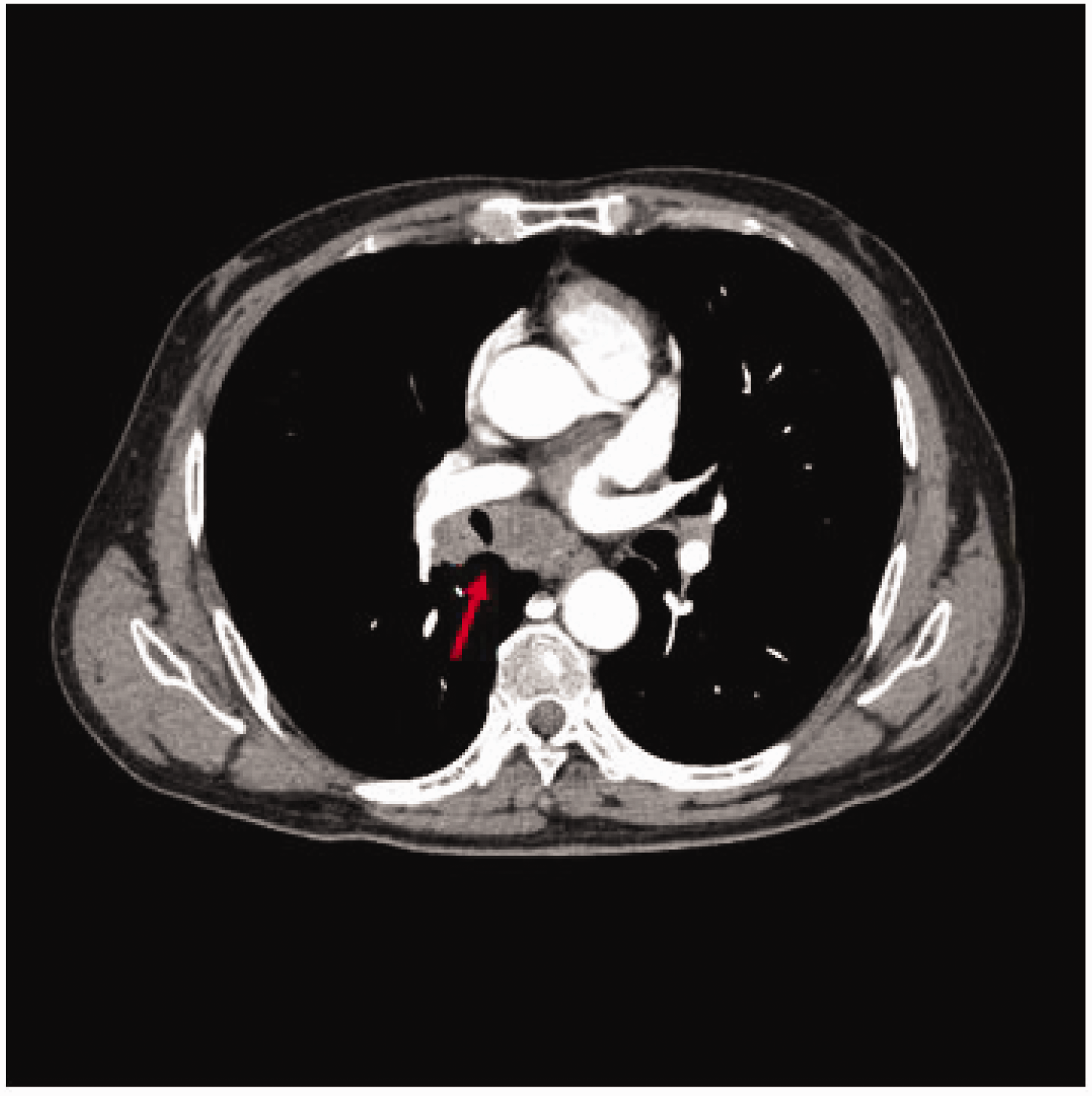
Computed tomography of the lung. An intrabronchial space-occupying lesion in the right middle lung lobe was observed.

Bronchoscopic findings. A bulging neoplasm with small nodular changes on the surface and localized bronchial stenosis was seen in the bronchus of the right middle lung lobe.

Hematoxylin–eosin staining and immunohistochemistry of bronchial tissue biopsy. The pathological examination revealed positive staining for pan-cytokeratin, wild-type p53, CD56, thyroid transcription factor 1, synaptophysin, and chromogranin A as well as Ki67 positivity of 90%. Negative staining was observed for p63, p40, and leukocyte common antigen.
Multidisciplinary consultation with medical oncology and thoracic surgery specialists led to the initiation of etoposide + carboplatin and srulizumab therapy. Following the second cycle of treatment, the patient developed abdominal distension, abdominal pain, loss of appetite, nausea, vomiting, and difficulty in defecation. Stool passage occurred only once every 7 days. CT imaging revealed conspicuous bowel dilatation with no evidence of mechanical obstruction (Figure 4). Comprehensive treatment, including enemas to promote gastrointestinal motility, was administered for 3 days in an effort to resolve the gastrointestinal dysfunction; however, the symptoms did not improve. Further investigation by electronic gastroscopy and epigastric CT revealed chronic atrophic gastritis and intra-abdominal free gas, respectively. Subsequent electronic colonoscopy suggested the presence of a very small foreign body in the colon, raising concerns about possible micro-perforation. Emergency laparoscopic exploration was performed, but no abnormalities such as gastrointestinal perforation were observed. Postoperatively, the patient continued to experience persistent abdominal distension, abdominal pain, and difficulty in defecation.

Computed tomography of the abdomen. Bowel dilatation with no evidence of mechanical obstruction was observed.
After a multidisciplinary discussion, it was concluded that the symptoms may be due to tumor metastasis. To further investigate, a positron emission tomography/CT examination was conducted, which showed no definite signs of hypermetabolic malignant lesions in the lungs. However, the right hilar lymph nodes were enlarged with increased fluorodeoxyglucose metabolism. Other parts of the body showed no significant abnormalities (Figure 5). The diagnosis and treatment of the patient’s condition remained uncertain.
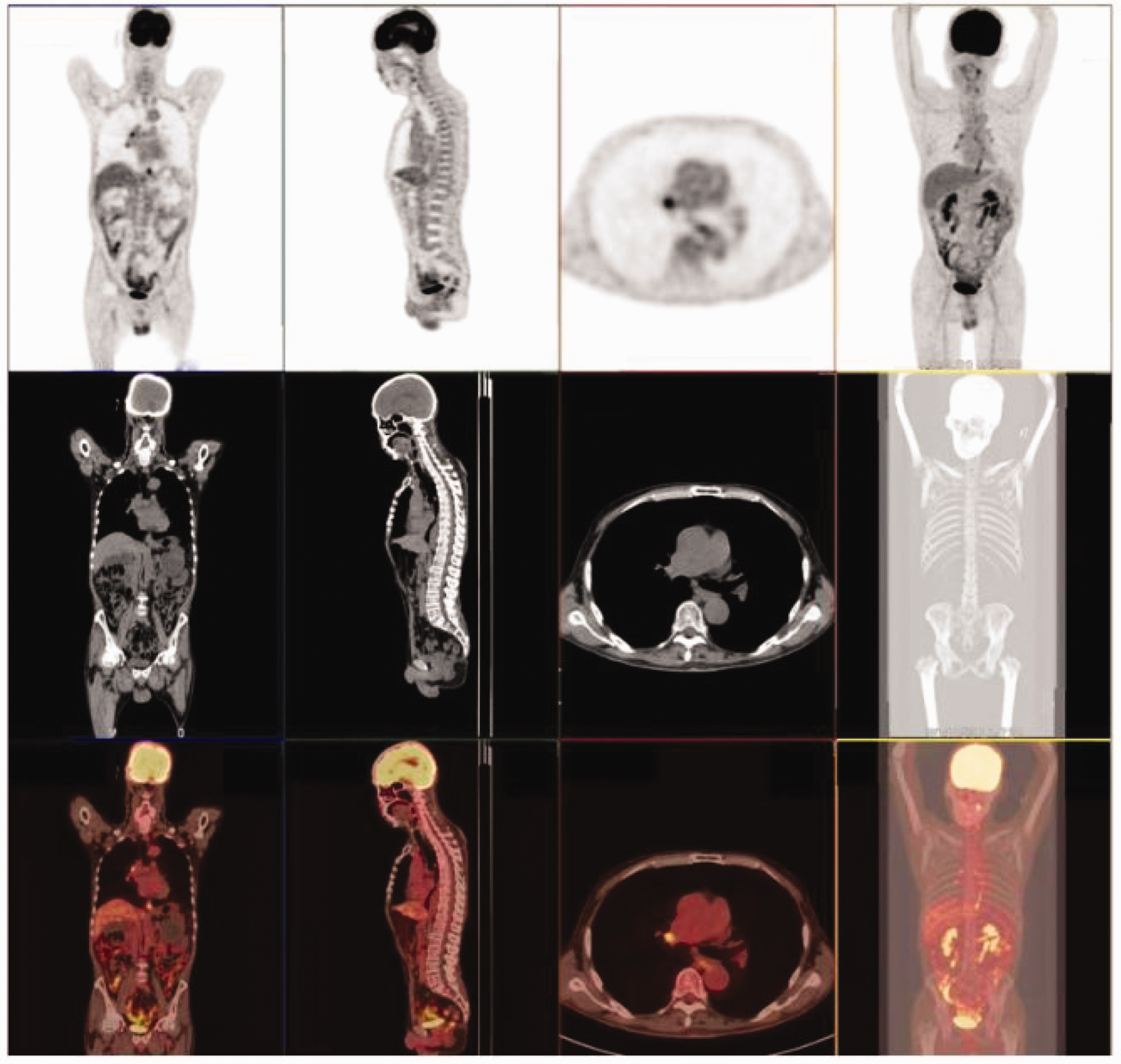
Positron emission tomography/computed tomography examination. The right hilar lymph nodes were enlarged with increased fluorodeoxyglucose metabolism. Other parts of the body showed no significant abnormalities.
The patient was referred to the gastroenterology department of our hospital for further workup. A complete gastrointestinal dynamics test revealed gastrointestinal dysfunction, prompting consultation with the neurology department to consider the possibility of PNS. Lumbar puncture was performed, and cerebrospinal fluid analysis detected positive anti-Hu IgG antibodies. Based on the patient’s medical history and cerebrospinal fluid test results, a diagnosis of PNS with intestinal pseudo-obstruction was considered.
With the consent of the patient and his family, the patient received high-dose intravenous human immunoglobulin shock treatment of 20 g once a day for 7 days. However, his symptoms of abdominal distension, abdominal pain, obstipation, and bitter taste in the mouth remained unresolved. A second-line treatment plan involving the administration of methylprednisolone 500 mg once a day for 3 days was initiated. Unfortunately, the patient’s symptoms did not improve, and considering the irreversible damage to the nervous system, the patient and his family elected discharge from the hospital. This case report follows the CARE guidelines 9 in its entirety.
Discussion
PNS is a rare condition, occurring in less than 1% of all patients with cancer. 1 The incidence of PNS is associated with the specific type of primary tumor. Studies have indicated that tumors expressing neuroendocrine proteins (e.g., SCLC and neuroblastoma), involving immunoregulatory organs (e.g., thymoma), containing neuronal components (e.g., teratoma), or affecting the production of immunoglobulins (e.g., myeloma) are highly correlated with PNS. 10 The key feature of PNS is that it is mediated by an aberrant immune response triggered by the tumor rather than being directly caused by the tumor itself. 11 Numerous studies have revealed abnormal manifestations of PNS across various organs and systems, including the brain, spinal cord, nerves, muscles, endocrine system, skin, blood, and joints. 12 Notably, however, CIPO is a relatively rare occurrence within the spectrum of PNS.13,14
CIPO represents a severe manifestation of gastrointestinal dysmotility and is frequently attributed to disturbances in innervation, smooth muscle function, or the interstitial cells of Cajal. This condition is characterized by recurrent episodes of intestinal subocclusion, closely resembling mechanical obstruction despite the absence of any demonstrable obstructive process. 15 Because of its intricate nature and diverse clinical presentations, CIPO is prone to misdiagnosis or delayed recognition, often leading to advanced disease stages. 16
A range of diseases, including paraneoplastic conditions, can lead to CIPO, 14 with paraneoplastic CIPO being closely associated with SCLC. One of the earliest documented instances of severe gastrointestinal dysmotility in patients with SCLC was reported by Lennon et al. 17 in 1991. They identified IgG antibodies reactive with neurons of the myenteric and submucosal plexuses of the jejunum and stomach in four of five patients with small cell carcinoma, all of whom had CIPO. Additionally, in 1993, Condom et al. 18 conducted an autopsy on a patient with CIPO and SCLC who exhibited high levels of anti-Hu antibodies and found significant neuronal and nerve fiber deficits in the patient’s mesenteric plexus. Similarly, in the current case report, a patient with SCLC and anti-Hu antibody positivity presented with CIPO. Upon reviewing the literature, we found an increasing number of reports of non-SCLC-associated paraneoplastic CIPO, such as cases in patients with prostate cancer, extraskeletal myxoid chondrosarcoma, and thymoma,2–5 as well as concurrent CIPO and Stauffer syndrome in patients with SCLC. 13 However, most of these cases involved a single paraneoplastic CIPO associated with SCLC.
It is currently understood that paraneoplastic CIPO is the result of a cross-reactive immune response to antigens shared by the tumor and the nervous system. 19 Specifically, anti-Hu antibodies are believed to cause disease by recognizing specific antigens that are expressed on both SCLC and neurons. Analysis of whole biopsies of intestinal tissue from patients with paraneoplastic CIPO revealed an infiltration of diffuse CD4- and CD8-positive T lymphocytes in the mesenteric plexus. 20 Additionally, in vitro experiments using SH-SY5Y neuroblasts and cultured myenteric neurons exposed to sera containing anti-HuD antibodies from patients with paraneoplastic gut dysmotility showed that anti-HuD neuronal antibodies induced caspase-3 and Apaf-1 activation as well as apoptosis. 6
The treatment of paraneoplastic CIPO is often met with limited success. In previous studies, various treatments, including prokinetic and anti-secretory agents, chemotherapy, and plasmapheresis, have not effectively alleviated clinical symptoms.21–23 In our case, despite administering high-dose gamma globulin and glucocorticoid shock therapy, the patient’s symptoms did not improve. However, there have been reports of long-lasting improvement in some cases with the use of rituximab and cyclophosphamide treatment.24,25 This improvement is thought to be due to the inhibition of antibody production by B-lymphocyte activation. Importantly, patients who exhibit sustained improvement through inhibition of B-lymphocyte activation and antibody production typically have a shorter disease course. Conversely, if a significant number of specific antibodies have already been generated and irreversible neuronal damage has occurred, inhibiting antibody production may not yield substantial symptom improvement. Therefore, the efficacy of this approach may be comparable to complete antibody removal through plasma exchange. Further research is warranted to explore alternative treatment strategies for paraneoplastic CIPO.
In clinical practice, it is crucial to consider the possibility of CIPO when encountering patients with clinical features resembling mechanical intestinal obstruction but without a clear underlying cause. Timely detection of related antibodies and early diagnosis and treatment of CIPO are of great clinical significance. Unfortunately, several factors contribute to the delayed diagnosis and treatment of CIPO. One factor is that many physicians lack experience in recognizing the condition, which hinders early identification of patients with CIPO. Another factor is that the nonspecific symptoms exhibited by patients with CIPO can be mistakenly attributed to other functional gastrointestinal disorders, further complicating accurate diagnosis. This diagnostic limitation increases the risk of subjecting patients to inadequate management strategies, including ineffective and potentially harmful surgical interventions. Therefore, efforts should be made to enhance physicians’ awareness and improve diagnostic methods for CIPO to ensure timely and appropriate management of patients.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241245868 - Supplemental material for Paraneoplastic syndrome with intestinal pseudo-obstruction in a patient with small cell lung carcinoma: a case report and literature review
Supplemental material, sj-pdf-1-imr-10.1177_03000605241245868 for Paraneoplastic syndrome with intestinal pseudo-obstruction in a patient with small cell lung carcinoma: a case report and literature review by Xiao-Jun Ren, Zhao-Hong Shi, Ying-Xia Cheng and Yun Luo in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605241245868 - Supplemental material for Paraneoplastic syndrome with intestinal pseudo-obstruction in a patient with small cell lung carcinoma: a case report and literature review
Supplemental material, sj-pdf-2-imr-10.1177_03000605241245868 for Paraneoplastic syndrome with intestinal pseudo-obstruction in a patient with small cell lung carcinoma: a case report and literature review by Xiao-Jun Ren, Zhao-Hong Shi, Ying-Xia Cheng and Yun Luo in Journal of International Medical Research
Footnotes
Acknowledgements
We would like to extend our sincere gratitude to our esteemed colleague, Mr. Kun Jiang, for his invaluable guidance in determining the precise location of the lesion on the high-definition CT scans. Additionally, we would like to express our deep appreciation to Mr. Peng-Cheng Liao for his assistance in collecting the pertinent endoscopic and pathological images of the patient.
Authors’ contributions
Data availability
The original data supporting the findings in this study are included in the article; further inquiries can be directed to the corresponding author.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethics approval and consent
This study was reviewed and approved by the Ethics Committee of Dazhou Integrated TCM and Western Medicine Hospital, Dazhou City, Sichuan Province, China. Written informed consent was obtained from the patient and the patient’s family. The patient has agreed to share their case details to contribute to the understanding and advancement of knowledge regarding paraneoplastic chronic intestinal pseudo-obstruction.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
