Abstract
Idiopathic gingival fibromatosis (IGF), a rare fibroproliferative disease of unknown etiology, affects gingival tissue and has substantial adverse effects on patients. Therefore, the pathogenesis of IGF requires more extensive and in-depth research. In this case, a patient with confirmed IGF underwent initial nonsurgical periodontal therapy and gingivectomy, and the prognosis was good. The patient had no loss of periodontal attachment but had a history of swelling and bleeding of the gingiva prior to fibrous enlargement, which prompted further investigation. We explored the patient’s subgingival microbiome and found a high abundance of periodontal pathogens. Gingival tissue biopsy revealed abundant fibrous tissue containing multiple inflammatory cell infiltrates. These results suggest that gingival inflammation secondary to periodontal pathogens can contribute to IGF onset.
Keywords
Introduction
Gingival fibromatosis is a rare type of benign tumor. Its current incidence is 1 in 175,000 with no sex-related difference. 1 The main clinical manifestations are painless gingival swelling and diffuse growth, which can cover most or all of the tooth surface. Therefore, gingival fibromatosis often affects the patient’s chewing function and facial appearance. Histopathologic features include excessive extracellular matrix deposition with abundant collagen fibers (e.g., type I collagen) in gingival tissue. Additionally, fibroblasts and a few blood vessels are evident. 2 Gingival fibromatosis may be accompanied by gingival inflammation, and periodontitis may also occur in some susceptible individuals. Gingival fibromatosis and aggressive periodontitis can reportedly coexist. 3 Because the etiology of gingival fibromatosis is unknown, the condition is termed idiopathic gingival fibromatosis (IGF); however, it is known to be associated with genetic factors and is therefore also termed hereditary gingival fibromatosis. 4 Most patients with IGF demonstrate a dominant inheritance pattern, but a few exhibit a recessive inheritance pattern. 5
The underlying pathogenesis of IGF remains unclear. Gingival hyperplasia usually occurs during the eruption of permanent teeth, but it may also occur at the beginning of the eruption of deciduous teeth. 6 However, no reports have described IGF in patients with missing teeth, suggesting that the presence of teeth is required for the development of IGF. 7 The presence of teeth appears to be a common factor in both periodontitis and IGF, and both diseases disappear after tooth loss. Periodontitis is a complex infectious disease in which periodontal pathogens such as Porphyromonas gingivalis and Fusobacterium nucleatum play an important role. 8 However, periodontal infections have not been definitively linked to IGF. Excessive extracellular matrix deposition is closely associated with the involvement of key intracellular signaling molecules (e.g., transforming growth factor beta), but pathogenic microorganisms and inflammatory responses to those microorganisms also make important contributions. 2 Gingival hyperplasia leads to the formation of periodontal pseudo-pockets followed by changes in the subgingival biofilm (SB) microbiome. 9 Alternatively, changes in the SB may lead to the formation of periodontal pockets and the onset of gingival hyperplasia. Overall, the role of microorganisms in the pathogenesis of IGF requires further exploration.
We herein describe a patient who developed IGF along with recurrent gingival swelling prior to clinically significant gingival growth. Before periodontal surgery, we collected SB specimens and gingival crevicular fluid (GCF) from the patient; we then explored the changes in the microbiome within each type of sample. Our findings provide insights to support further research into the association between the SB microbiome and the etiology of IGF.
Case report
Diagnosis
A 27-year-old man presented to the periodontal department in our hospital for evaluation of gingival swelling, which was affecting his facial appearance and eating habits; he had no other obvious symptoms. Eight months prior to presentation, the patient had developed substantial gingival swelling and bleeding around the anterior maxillary teeth. The swollen area was sensitive to manual pressure, and the swelling decreased after treatment with anti-inflammatory medication. Six months prior to presentation, the gingival swelling recurred; the texture of the swelling was firm, but no bleeding was present. The patient reported brushing his teeth twice per day. He had a 5-year history of smoking 15 to 20 cigarettes per day, and he had no other medical problems. He had no family history of relevant genetic disorders.
Oral and maxillofacial examinations revealed maxillofacial symmetry, and the upper and lower ratios were coordinated. The pattern and degree of mouth opening were normal, and the posterior teeth exhibited neutral alignment. There were no ulcers in the oral mucosa and no obvious bleeding on probing. Oral hygiene was poor with dental calculus, soft tartar, and plaque accumulation. The gingiva was obviously hyperplastic and swollen with a firm texture; the swelling involved the gingival papilla and attached gingiva and covered the middle third of the tooth surface (Figure 1). No teeth displayed attachment loss. Orthopantomography showed no obvious alveolar bone resorption, and the alveolar crest position was normal (Supplementary Figure 1). Upon general examination, the patient showed no signs of hirsutism or intellectual disability. Hematologic analysis revealed no significant abnormalities, and the patient was not taking drugs known to cause excessive gingival growth. He reported experiencing one episode of gingivitis-associated gingival swelling and bleeding. However, when the patient sought medical attention, the texture of the gingiva was tough and significantly enlarged, and the attached gingivae of all teeth were affected. In addition, the patient had no attachment loss or obvious bleeding on probing. Therefore, the patient’s gingival hyperplasia and swelling were diagnosed as IGF.
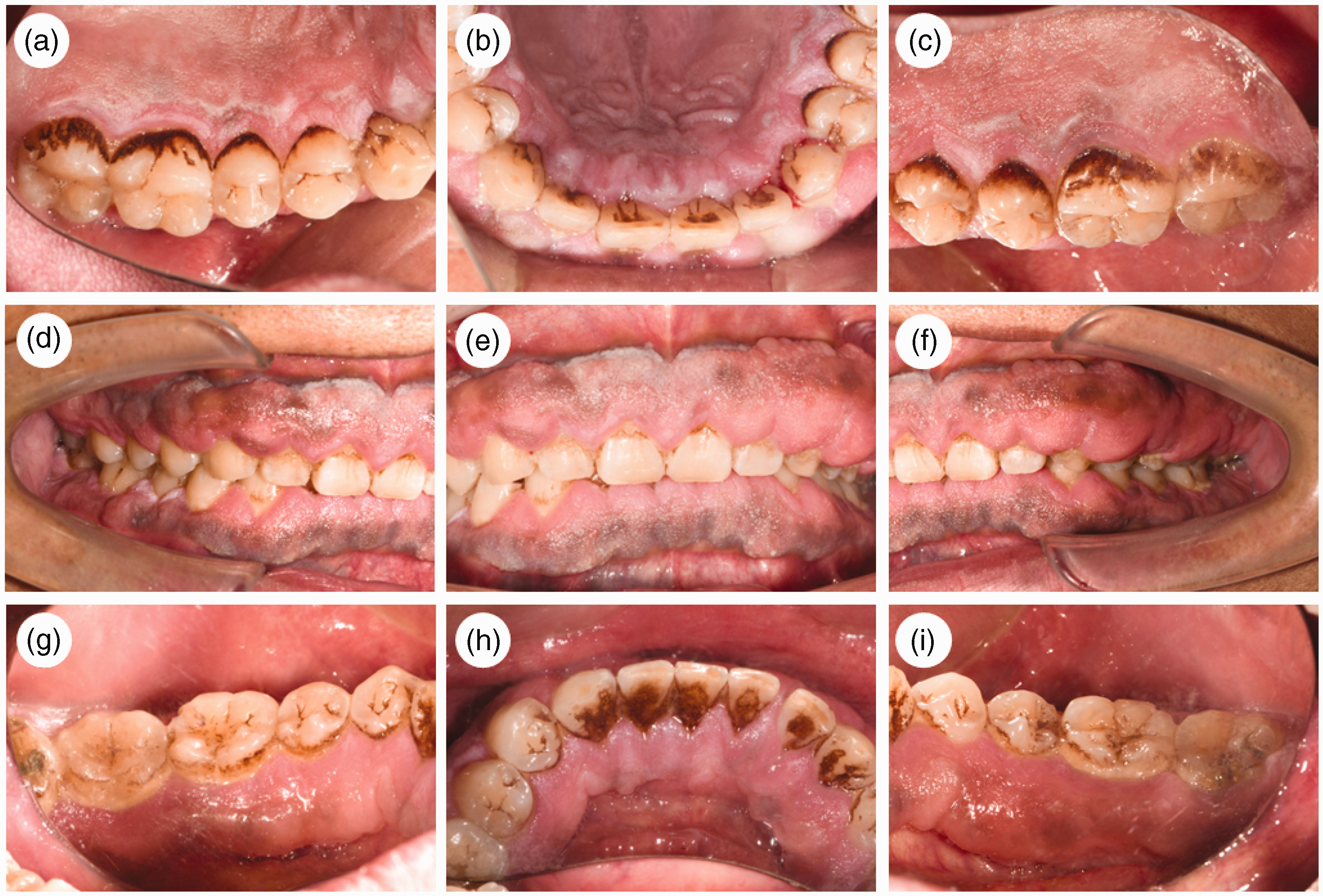
Photographs of patient’s dentition and gingiva at the first visit.
Histopathologic findings
Gingival tissues were collected from buccal and lingual sites during surgery and immediately fixed in formaldehyde solution. The specimens were then sent to the tissue pathology laboratory for routine dehydration, transparency, paraffin embedding, and paraffin sectioning. Serial sections of 5-µm thickness were prepared, stained with hematoxylin and eosin, and visualized under a microscope.
Histopathologic evaluation of the gingival tissues revealed gingival fibroplasia characterized by thickened epithelium, elongated reticular cristae, and densely packed collagen bundles containing fibroblasts. The tissues also displayed significant inflammatory cell infiltration between collagen fiber bundles, and multiple inflammatory cell foci were present (mainly plasma cells with small numbers of other cells such as neutrophils and macrophages). The firm, rice-sized growths on the gingival surface consisted of a continuous, intact, regular epithelial layer surrounded by connective tissue containing numerous collagen fibers along with fibroblasts (Supplementary Figure 2). The differential diagnosis of gingival hyperplasia that may be accompanied by inflammatory response is shown in Supplementary Table 1.
Subgingival microbiome analysis
Before surgery, we collected GCF and SB samples from three teeth in different areas of the dentition (mesiobuccal and mesiolingual aspects of teeth 16, 11, and 36), stored them on dry ice, and sent them to a sequencing laboratory for microbial 16S rDNA sequencing and subgingival community structure analysis.
As shown in the bar chart of community composition, Porphyromonas, Fusobacterium, and Prevotella were the most abundant genera in the GCF and SB samples. Fretibacterium and Treponema demonstrated higher abundances in the SB than GCF samples, whereas Streptococcus demonstrated higher abundance in the GCF samples (Supplementary Figure 3(a)). Porphyromonas gingivalis and Fusobacterium nucleatum were the most abundant species in both types of samples, followed by Porphyromonas endodontalis. The abundance of uncultured Synergistetes was higher in the SB than GCF samples, and the abundance of unclassified Veillonella was higher in the GCF samples (Supplementary Figure 3(b)).
Treatment
Considering the patient’s disease characteristics, we held a discussion with the patient and then established a three-step treatment plan: initial periodontal therapy, maxillary gingivectomy, and mandibular gingivectomy. For basic periodontal treatment, the patient underwent supragingival ultrasonic cleaning of all teeth. One week later, he underwent subgingival scaling and a series of irrigation procedures with hydrogen peroxide and normal saline (Supplementary Figure 4). He was then observed for 2 months prior to subsequent treatment. The maxillary gingivectomy procedure was performed under local anesthesia. After routine disinfection and local anesthesia, a periodontal probe was used to detect the depth of the periodontal pockets, and methylene blue was used to mark the bottom of the gingival groove. Above this mark, a primary incision was made perpendicular to the tooth surface; this was followed by an internal oblique incision and reduction of the gingival thickness. The remaining gingival tissue, pathological granulation tissue, and residual dental calculus were then removed, and the surgical wound was tightly sutured (Figure 2). Oral antibiotic treatment was prescribed (amoxicillin, three times daily for 3 days). Analgesic medication (e.g., ibuprofen) was permitted in the event of severe pain. The sutures were removed 1 week after surgery, and the patient received instructions regarding routine oral hygiene. The mandibular gingivectomy procedure was similar to the maxillary gingivectomy procedure (Supplementary Figure 5).
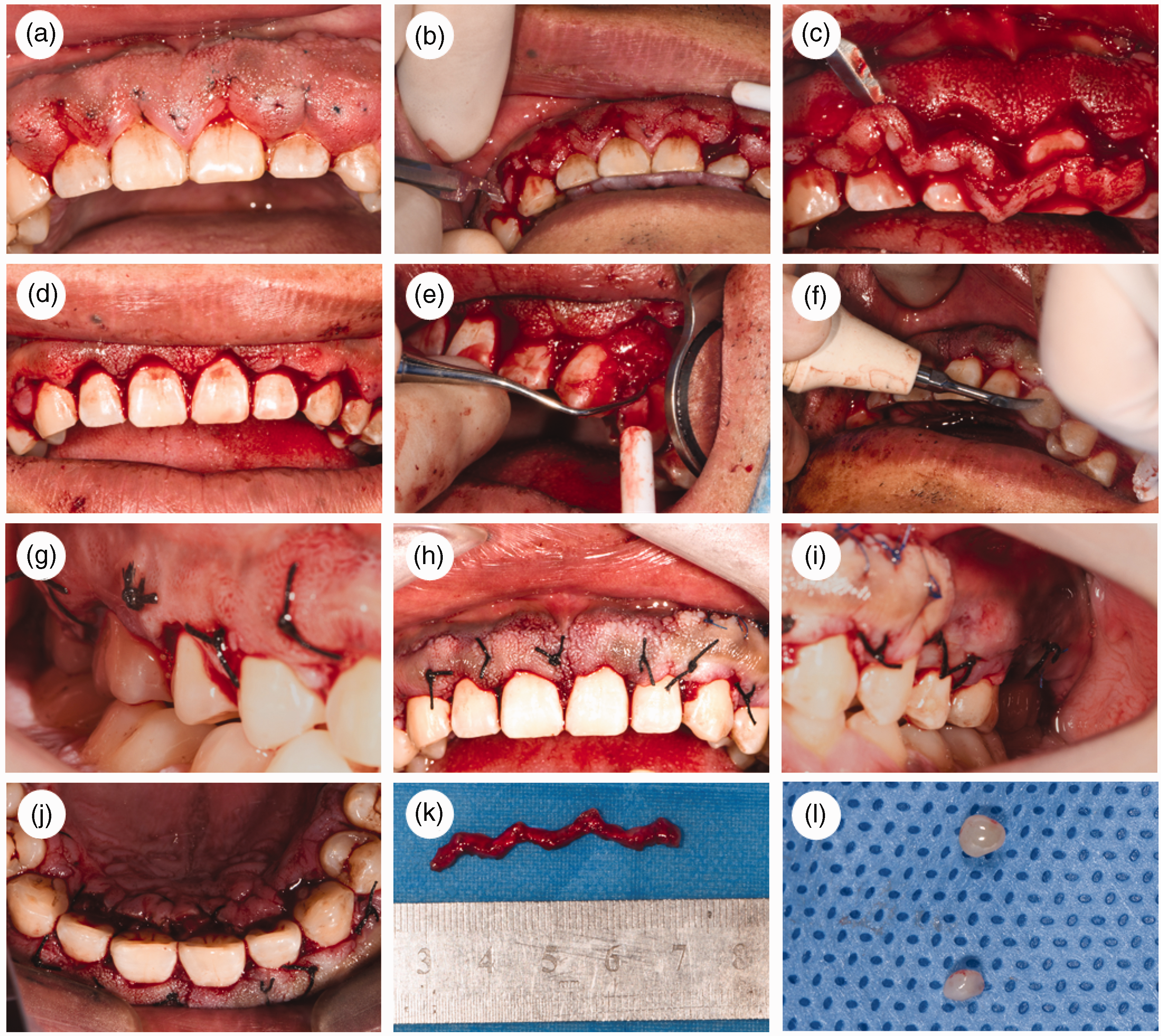
Maxillary gingivectomy. (a) Marking point. (b) Primary incision. (c) Removal of excess gingiva. (d) Second incision to reduce gingival thickness. (e) Removal of remaining gingiva and granulation tissue. (f) Ultrasound to remove remaining dental calculus. (g–j) Views after suturing. (k) Gingival tissue proliferation and (l) two firm, rice-sized growths on the surface of the swollen gingiva.
Follow-up
The patient returned to the clinic at 6-month intervals. He felt that the esthetic appearance of the gingiva had considerably improved, and he reported no discomfort. Oral examinations revealed no recurrence of the gingival hyperplasia or swelling, and the patient’s oral health was good (Figure 3).

Follow-up findings 1 year after surgery.
Ethics statements
Written informed consent was obtained from the patient to publish her anonymized data. The reporting of this study conforms to the CARE guidelines. 10 We obtained the patient’s written consent prior to treatment. This study was approved by the Ethics Committee of the Stomatological Hospital of Lanzhou University (No. LZUKQ-2023-020) and was conducted in accordance with the ethical standards of the Declaration of Helsinki.
Discussion
In this case, the clinical manifestations were gingival swelling and hyperplasia. The patient had no family history of gingival fibromatosis, systemic diseases, or other potential triggers of these gingival abnormalities. Considering these clinical factors and pathologic findings (numerous collagen fiber components), the patient was diagnosed with IGF. In most cases, the onset of IGF occurs during eruption of the permanent teeth in adolescents; however, it may also occur during eruption of the deciduous teeth in children. 4 In this case, the patient did not develop obvious gingival swelling and hyperplasia until the age of 27 years. These differences merit careful consideration because they may provide insights into the etiology of IGF.
Our patient had a smoking history of at least 5 years and obvious dark brown smoke patches on the surfaces of the teeth. Research has confirmed that smoking is a risk factor for periodontitis, and there is a positive correlation between smoking and the severity of periodontitis. 11 Some studies have shown a dose–response relationship between smoking and the development or recurrence of periodontitis. 12 However, smoking can lead to a decrease in gingival bleeding and GCF volume, likely via smoking-induced vasoconstriction and reduced blood flow. 13 We reviewed several published case reports for comparison with our findings. Most reports did not clearly indicate whether the patient smoked, possibly because the patient was young; however, several descriptions of IGF in men aged >20 years (including our patient) clearly indicated that the patient had a long history of smoking.14,15 There is a need for further research concerning whether smoking is associated with IGF onset.
The etiology of gingival fibromatosis is unknown, and current research generally focuses on links between gingival fibromatosis and various genetic factors (e.g., gene variants). 2 Although genetic factors may be the key underlying cause of gingival fibromatosis, two issues require clarification. First, what pathogenic factors contribute to the onset of gingival fibromatosis in the absence of a clear genetic background? Second, do SB microorganisms and their toxic metabolites, as well as the resulting gingival inflammation, play a role in the pathogenesis of IGF? Our patient reported a history of gingival swelling, tenderness, and bleeding, which are signs of gingival inflammation. To determine whether SB changes had occurred in our patient, we examined the subgingival microbiome, including the microbiome in the GCF and that in the SB attached to the tooth surface. In the SB, the most abundant genera were Porphyromonas and Fusobacterium, and the most abundant species were Porphyromonas gingivalis and Fusobacterium nucleatum. These two bacterial genera are associated with periodontal disease, and their abundances are significantly higher in patients with periodontal disease than in healthy individuals.16,17 Accordingly, proliferative periodontal pseudo-pockets display an altered microbiome, similar to the microbiome in periodontitis. This is consistent with the history of gingival swelling in our patient. The increased abundances of pathogenic microorganisms in periodontitis inevitably lead to an immune response in gingival tissue. The specific role of this response in IGF should be explored in future studies. To our knowledge, the present analysis is the first to demonstrate that the subgingival microbiome in patients with IGF is altered in a manner similar to that in patients with periodontitis, suggesting a link between the two diseases. Previous reports have indicated that mild gingival swelling in patients with IGF can be resolved by improving oral hygiene and that the recurrence of gingival swelling after surgery is influenced by plaque and calculus management, consistent with our observations.7,18 Notably, some patients with IGF also develop aggressive periodontitis; however, the relationship between the subgingival microbiome and IGF has not been explored. The authors of previous reports have suggested that both IGF and aggressive periodontitis have a genetic background.
Previous studies have often revealed limited numbers of inflammatory cells in gingival tissue from patients with IGF (excluding patients with aggressive periodontitis). By contrast, we observed multiple inflammatory cell infiltrates surrounding the abundant collagen fibers in the subepithelial connective tissue. However, our histopathologic results were consistent with our microbiome findings. Specifically, we found that the subgingival microbiome in our patient contained high abundances of the periodontal pathogens Porphyromonas gingivalis and Fusobacterium nucleatum, which can cause inflammation in periodontal tissue.16,17 Our histopathologic results also indirectly confirmed the patient’s report of gingival swelling, which subsided after the use of anti-inflammatory medication. There is a need to explore the relationship between gingival swelling and subsequent gingival fibroplasia. We believe that this relationship may arise because inflammation promotes the development of gingival fibrosis. 19 Our patient displayed a transitional state wherein gingival tissue fibrosis replaces inflamed tissue; eventually, the fibrotic area increases whereas the area of inflammatory cell infiltrates decreases. Nevertheless, the underlying mechanism requires investigation. The inflammatory response can stimulate tissue fibrosis, and the findings in our patient with IGF appear to be consistent with this pathological process. We suspect that the pathogenesis of IGF involves local inflammation as a contributing factor. Importantly, if our patient had presented after prolonged swelling, we may have observed almost entirely fibrous tissue, which would be consistent with previous reports.
Conclusion
In this report, we have described a case of IGF in a patient who underwent gingivectomy and had a good prognosis. Importantly, we found that the subgingival microbiome in this patient was altered in a manner similar to that of periodontitis. Histopathologic examination revealed numerous inflammatory cell infiltrates in the swollen and hyperplastic gingival connective tissue, which may have been related to the onset of gingival fibrosis.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241245302 - Supplemental material for Idiopathic gingival fibromatosis and primary analysis of dominant bacteria in subgingival biofilm: a case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605241245302 for Idiopathic gingival fibromatosis and primary analysis of dominant bacteria in subgingival biofilm: a case report by Jingli Zhu, Xinyue Fan, Lili Ding and Tianzhu Song in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605241245302 - Supplemental material for Idiopathic gingival fibromatosis and primary analysis of dominant bacteria in subgingival biofilm: a case report
Supplemental material, sj-pdf-2-imr-10.1177_03000605241245302 for Idiopathic gingival fibromatosis and primary analysis of dominant bacteria in subgingival biofilm: a case report by Jingli Zhu, Xinyue Fan, Lili Ding and Tianzhu Song in Journal of International Medical Research
Footnotes
Acknowledgements
We would like to thank all individuals who participated in this study.
Authors’ contributions
Jingli Zhu contributed to the data collection and analysis, diagnosis, treatment, and manuscript drafting. Xinyue Fan and Lili Ding contributed to the sample collection and data interpretation. Tianzhu Song drafted and critically revised the manuscript. All authors gave final approval of the manuscript and agree to be accountable for all aspects of the work.
Availability of data and materials
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This study was supported by the Gansu Province Science and Technology Plan Project (23JRRA721) awarded to Tianzhu Song and the Clinical Team Project of Stomatology Hospital of Lanzhou University (lzukqky-2022-t07) awarded to Jingli Zhu.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
