Abstract
Portal vein gas accumulation and intestinal pneumatosis are uncommon signs indicating a high mortality risk in cases of intestinal ischemic necrosis. However, the widespread use of computed tomography has led to an increase in detection of benign lesions. We report a case of portal vein gas accumulation resulting from organophosphorus pesticide poisoning. A male patient was brought to the hospital in a comatose state with bilateral pupils that measured 1.0 mm, and he showed shortness of breath and wet rattles in the lungs. A cholinesterase concentration of 214 U/L was detected on an auxiliary examination. The patient was diagnosed with organophosphorus pesticide poisoning and underwent mechanical ventilation, hemoperfusion, and continuous renal replacement therapy according to the poisoning guidelines. On the fifth day, considerable abdominal distension was observed. An abdominal computed tomography scan revealed dilation of the small bowel and ascending colon with fluid and gas accumulation, as well as gas within the intestinal wall and hepatic veins. Although portal vein gas and intestinal pneumatosis are a sign of mortality requiring immediate surgical intervention, an increasing number of benign cases suggests potential benefits of conservative treatment approaches.
Keywords
Introduction
Hepatic portal vein gas (PVG) is a rare radiographic finding characterized by an abnormal accumulation of gas within the portal vein and its intrahepatic branches. PVG is typically caused by various underlying conditions and its prognosis depends on the underlying condition. PVG has historically been considered a sign of poor prognosis because it is most commonly associated with intestinal ischemia and necrosis. PVG can also occur in non-gastrointestinal conditions, such as septicemia and abdominal infections. In recent years, there has been an increase in the identification of benign causes for PVG. 1 We report a case of development of PVG as a result of secondary organophosphate pesticide poisoning.
Case presentation
A 68-year-old male retiree with a previously unremarkable medical history was admitted after being discovered in a comatose state for 3 hours. Three hours previously, he was found unconscious and unable to breathe by his family members. He was lying on the ground, foaming at the mouth, and there was a smell of pesticide in the air. An empty bottle of dichlorvos was found beside him.
When the patient was brought to the hospital for emergency treatment, he was experiencing respiratory distress, with bilateral moist rales in his lungs and a non-invasive oxygen saturation level of 85%. To manage his condition, he underwent endotracheal intubation for ventilation and gastric lavage. The results of a cholinesterase exam showed a concentration of 214 U/L (normal: 35–135 IU/L). Additionally, he received an intravenous infusion of 1.0 pralidoxime iodide and an atropine injection of 0.5 mg intravenously every 10 minutes. After undergoing a head, thorax, and abdominal computed tomography (CT) scan, he was transferred to the hospital for treatment. A physical examination on admission showed the following: body temperature, 34°C; pulse, 128 beats/minute; respiratory rate, 23 beats/minute; blood pressure, 71/45 mmHg; and critical care pain observation tool score, 0. The patient was confused and both pupils were 1.0 mm. The reflex of light was dull, he was free from perspiration, and his skin was red. The patient was intubated via an orotracheal route and received ventilatory support from a mechanical ventilator. There was no cyanosis of the lips. On listening to the lungs, coarse breath sounds and moist rattles were heard. The heart rhythm was irregular. Upon palpation, the abdomen was soft and bowel sounds were diminished. There was no noticeable swelling or concave deformities in either lower limb, and a bilateral Pap smear test showed a negative result. An auxiliary examination showed the following: cholinesterase concentration of 214 U/L, white blood cell count of 13.2 × 109/L (3.5–9.5 × 109/L), 59% neutrophils (40%–5%), C-reactive protein concentration of 0.49 mg/L (0–6 mg/L), and fibrinogen concentration of 3.29 g/L (2–4 g/L). A blood gas analysis showed the following: pH of 7.3 (7.35–7.45), partial pressure of carbon dioxide of 27 mmHg (35–45 mmHg), partial pressure of oxygen of 80 mmHg (80–100 mmHg), and lactic acid concentration of 4.7 mmol/L (0.5–2.2 mmol/L). A chest and cranial CT exam (including the thorax) showed (1) low-density lesions on both temporal lobes, possibly indicating infarction, and (2) infection in both lungs.
The admission diagnosis comprised the following possibilities: (1) organic phosphorus pesticide poisoning, (2) hypovolemic shock, (3) metabolic acidosis, (4) inhalation pneumonia, and (5) cerebral infarction. After admission, the patient received conventional treatment according to the poisoning guidelines, comprising activated charcoal (30 g) and mannitol (250 mL) through the gastric tube to enhance adsorption, catharsis, and detoxification. Blood perfusion therapy was conducted once daily for a total of three sessions in conjunction with continuous renal replacement therapy. The patient received intravenous administration of pralidoxime iodide (6 g) daily to counteract the effects of poisoning, while atropine injections were provided intravenously to achieve atropinization as quickly as possible. Intravenous piperacillin-tazobactam (4.5 g) was administered every 8 hours to combat inhalation pneumonia and prevent further infection. The patient also received supportive treatment, such as fasting, gastrointestinal decompression, and maintenance of a stable internal environment. High-dose vasoactive medications were administered to raise blood pressure owing to difficulty in maintaining the circulation. Bedside ultrasound showed considerable fluid accumulation in the gastrointestinal tract (Figure 1).

(a) The small intestine is dilated with intestinal effusion and the plicae are clearly visible and (b) The colon is obviously dilated and shows effusion. Hepatorenal recess can be seen with no obvious effusion.
After the initiation of active fluid resuscitation therapy and plasma infusion, the patient gradually stabilized over 2 days, and regained consciousness on the third day. On the fifth day, there was marked widening of the abdomen, disappearance of bowel noises, and no tenderness or rebound pain. An abdominal CT scan showed considerable expansion of the small intestine and colon (Figure 2). Intra-abdominal pressure measured by the intravesical method was 32 mmHg, which suggested bowel obstruction. A subsequent enhanced CT scan showed portal vein gas and hepatic vein gas (Figure 3). After consultations with various departments, including gastrointestinal surgery, interventional medicine, and imaging, there appeared to be insufficient evidence of intestinal necrosis and no indication for exploratory laparotomy. Therefore, the patient’s condition was monitored after decompressing the intestinal obstruction using a catheter (Figure 4).

Abdominal computed tomography shows dilatation of the intestinal cavity without an obvious fluid level.
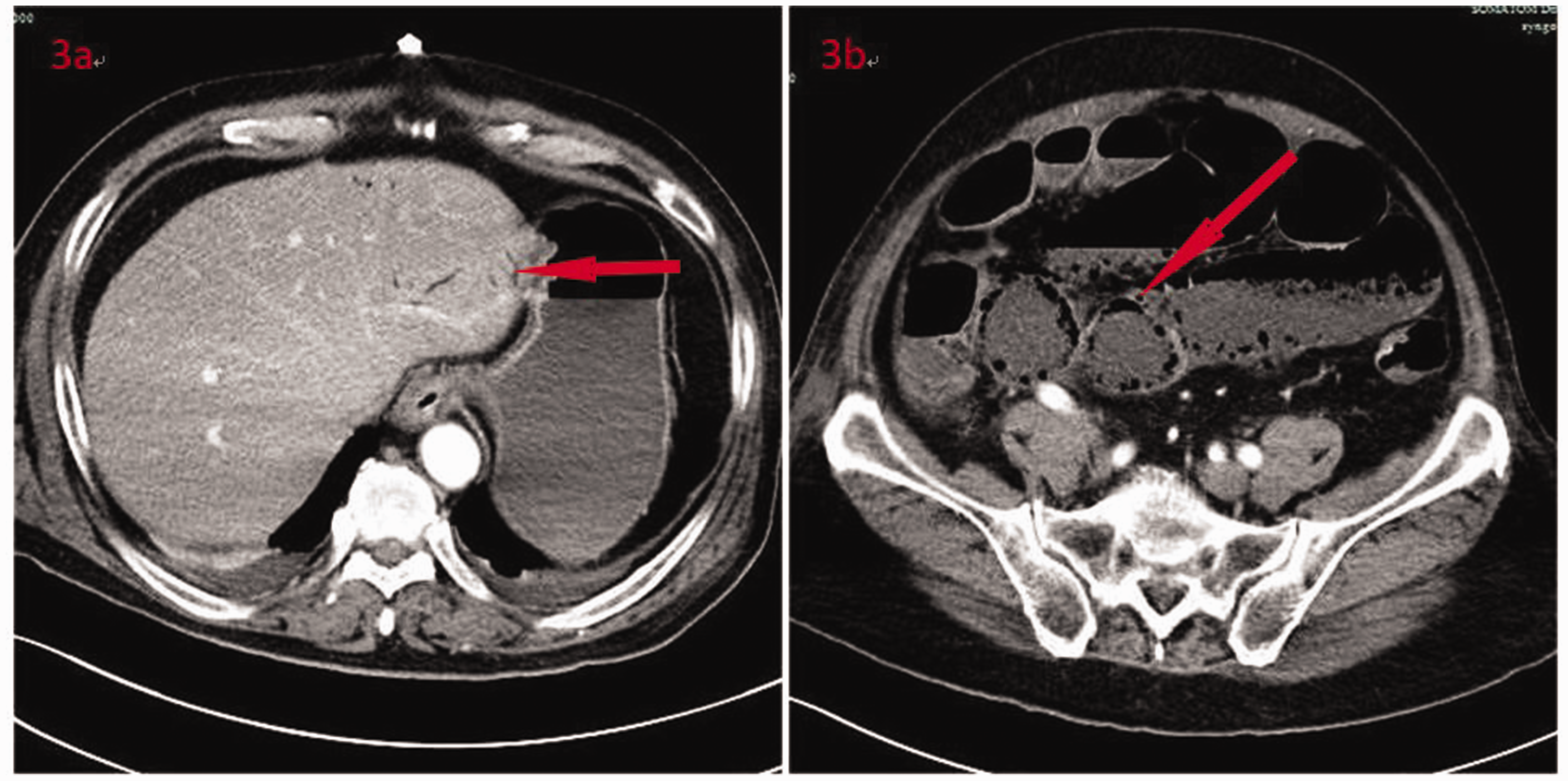
(a) Enhanced abdominal computed tomography shows portal vein gas (arrow) and gastric dilated fluid and (b) extensive dilatation and aeration of the intestinal cavity can be seen, and the fluid–gas level (arrow) and a indicates the presence of intestinal obstruction.
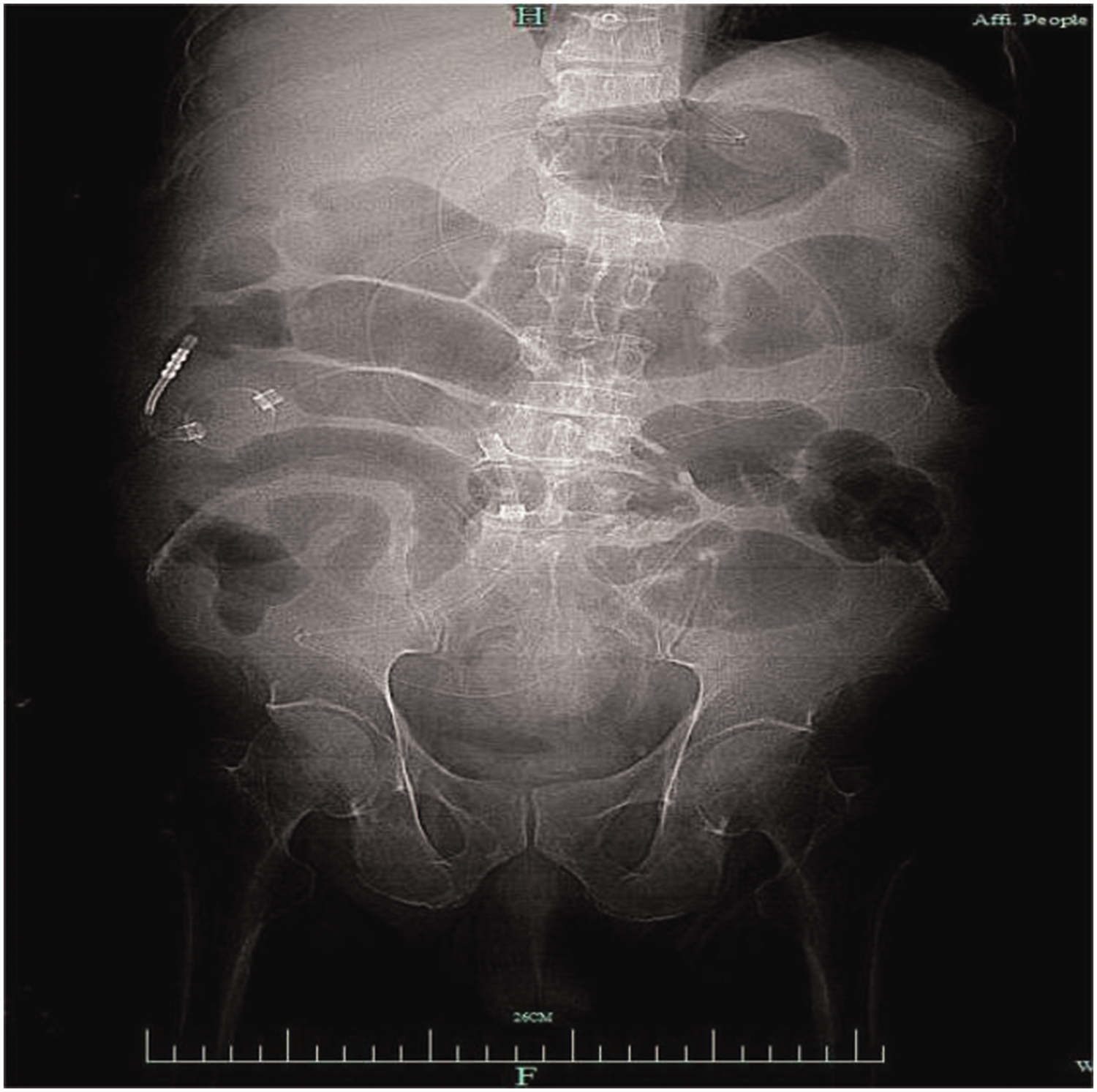
An ileal catheter is placed in the jejunum.
The patient underwent a bowel obstruction catheter insertion surgery, while the administration of atropine and sedative medications was gradually reduced. This treatment led to a gradual relief of abdominal distension without visible signs of peritoneal irritation. Nutritional solution was initiated nasally on day 10 after admission, and tracheal intubation was removed on day 15. The patient’s condition continued to improve, leading to his discharge on day 25.
Discussion
PVG is a rare radiographic finding characterized by abnormal accumulation of gas in the portal vein and its liver branches. This gas can enter the small mesenteric veins and eventually reach the liver through the superior and inferior mesenteric veins and portal veins. PVG can be caused by a variety of underlying factors. The presence of gas within the portal venous system is a rare occurrence. Causes facilitating the entry of intraluminal gas into the portal vein system include ulcer perforation damaging the intestinal wall, increased permeability of the intestinal wall, intestinal distention, and shedding of the intestinal epithelium. 2 Mesenteric ischemia is the most common pathology associated with PVG, but it can also be observed in cases of trauma and transplantation. 1 Pneumatosis in the portal vein and superior mesenteric vein can coexist, with portal phlebitis and liver transplantation commonly associated with the former, and small bowel lesions frequently associated with the latter. Generally, PVG has a good prognosis and does not typically require surgical intervention. However, if there is simultaneous accumulation of gas in the portal vein system and bowel wall, the risk of mesenteric ischemia greatly increases and may necessitate surgical intervention. Patients with this condition have a poor prognosis, with a mortality rate of 75% to 90%. 3 Therefore, in a patient presenting with both PVG and intestinal pneumatosis, there is the possibility of ischemic enteritis with tissue death. The presence of intra-abdominal pressure may indicate the requirement for surgical intervention. However, surgery and anesthesia could worsen intestinal obstruction, which could further increase intra-abdominal pressure.
Changsheng et al. reported a case in which PVG developed after self-administration of organophosphorus pesticides. The case was ultimately confirmed to be associated with intestinal necrosis, indicating a possible association between organophosphorus pesticide poisoning, intestinal necrosis, and the accumulation of PVG. 4 The clinical manifestations of organophosphate intoxication can be categorized into three types of symptoms: muscarine, nicotinic, and neurological symptoms. Common symptoms include increased airway secretion, abdominal pain, diarrhea, miosis, tearing, salivation, facial, limb, and systemic striated muscle twitching, dizziness, and headache. Severe manifestations often present as dyspnea and alterations in consciousness. 5 Currently, there is no specific treatment for organophosphate intoxication, and symptomatic supportive treatment is predominantly used. Organophosphate pesticides have a direct effect on the gastrointestinal tract. 6 Bird et al. 7 suggested that an increase in acetylcholine with organophosphate pesticide poisoning causes the intestine to release epinephrine and norepinephrine by overstimulating the toxic alkali receptor, thus causing intestinal vasospasm and microcirculation disorder. Elevated blood lactic acid concentrations and intestinal wall dilation suggest severe organophosphorus pesticide poisoning and a poor prognosis. 8
Our patient had a medical history of severe hypotension since admission, which carries a high risk of ischemic necrosis. If ischemic necrosis occurs, surgical intervention is necessary to prevent the high risk of delayed treatment and mortality. Moreover, if elevated intra-abdominal pressure remains unrelieved, it can further worsen the hypoperfusion of abdominal organs, leading to liver and kidney failure, posing a life-threatening situation for the patient. Therefore, establishing whether the patient has intestinal necrosis is important because it will determine the subsequent steps in diagnostic and treatment plans, such as the requirement for surgery and the choice of surgical methods. Studies have indicated that ultrasound or CT scans can aid in distinguishing between benign and life-threatening causes of intestinal pneumatosis. 9 However, there is still a lack of specific clinical indicators to determine whether pneumatosis intestinalis associated with PVG is a result of ischemic intestinal necrosis. While previous studies have indicated that intestinal pneumatosis and PVG may be associated with a poor prognosis, potentially indicating intestinal ischemic necrosis, many benign causes have been identified recently. 10 As a result, not all cases of intestinal pneumatosis and PVG necessarily require surgical treatment.11,12 In a study conducted by Lebert et al, among 14 patients with intestinal pneumatosis and PVG, only two patients had intestinal ischemic necrosis during surgical intervention. 13 However, distinguishing between hepatic venous gas combined with intestinal wall gas and determining whether it is benign or ischemic are difficult. 14 In our patient, we observed that despite his distended abdominal wall, there was no muscle tension. Laboratory tests also indicated no increase in inflammatory markers, such as the white blood cell count and C-reactive protein concentrations. Moreover, an enhanced abdominal CT scan showed no obstruction in the mesenteric arteries and no signs of filling defects, which suggested that the likelihood of intestinal ischemic necrosis was not increased. The consensus was reached to implement a comprehensive treatment approach, including decompression through intestinal obstruction catheter, discontinuation of anticholinergic and sedative medications, and promotion of gastrointestinal motility. Close monitoring of the patient’s gastrointestinal function and intra-abdominal pressure was also recommended. After conservative treatment, his bowel obstruction improved, and the intra-abdominal pressure returned to a normal level. We speculate that, in this case, extensive mucosal damage to the intestinal wall occurred as a result of self-administration of organophosphorus pesticides. B-ultrasound showed fluid accumulation in the intestinal cavity, indicating increased permeability of the intestinal wall and evident leakage following fluid resuscitation. Furthermore, the patient’s hypotension and prolonged intestinal ischemia led to intestinal dilation, decreased intestinal peristalsis, and gastrointestinal dysfunction. The administration of anticholinergic and sedative medications further suppressed gastrointestinal motility, leading to the development of paralytic ileus. This condition subsequently resulted in the presence of gas within the intestinal wall and portal vein, similar to the findings reported by Changsheng et al. 4 Severe intestinal obstruction and increased intra-abdominal pressure led to a progressive accumulation of gas in the intestinal cavity. This in turn caused intra-abdominal hypertension, abdominal compartment syndrome, and the absorption of gas from the damaged intestinal wall into the portal vein and hepatic vein.
In summary, the occurrence of PVG in combination with mesenteric ischemic necrotic enteritis is relatively uncommon. While surgery is important for this condition, the presence of gas in the intestinal wall along with PVG is not always indicative of necrotizing enteritis. Making a comprehensive assessment in this situation is crucial. In cases where PVG is associated with non-ischemic necrotizing enteritis, conservative treatment may be a viable option.
The reporting of this study conforms to the CARE guidelines. 15
Footnotes
Acknowledgements
We thank the staff and patient for their contributions and participation in the study.
Author contributions
Huipeng Zhu and Guozhong Chen made equal contributions to writing of the case report, the main experimental work, and data analysis. Huajun Wang was responsible for the coordination, organization, and communication with journal Editors and peers throughout the research. In addition, Huajun Wang was also involved in revision of the knowledge content of the manuscript. Hong Keyan and Liu Shiyi participated in the diagnosis and treatment process, and reviewed and revised the manuscript. All authors have read and approved the manuscript.
Availability of data and materials
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
This study was a case report and did not require ethics approval. The patient provided written informed consent for the publication of this case report and the accompanying images. Copies of the written consent are available for review by the Editor-in-Chief. Consent for treatment was obtained from the patient.
Funding
This article was supported by Ningbo Clinical Ophthalmology Research Center funding (2023-Y7).
