Abstract
Objective
To examine the effects of the temporal inverted internal limiting membrane (ILM) flap technique for foveal reconstruction in patients with highly myopic macular hole-associated retinal detachment (MHRD).
Methods
A retrospective case series analysis of four patients (four eyes) with MHRD was conducted. The foveal optical coherence tomography changes following treatment using the temporal inverted ILM flap technique were evaluated.
Results
In Patient 1, the ILM bridged the macular hole and residual subretinal fluid on postoperative day 6, and complete retinal reattachment was achieved at 19 months. Patient 2 exhibited reduced retinal detachment, with visible ILM inversion and macular hole closure after 14 days. In Patient 3, macular hole closure and fovea formation had occurred by day 25, and the ILM flap was visible. At 2 months, the outer collagenous layer connection in the central fovea and recovery of the external limiting membrane and ellipsoid zone were observed. Patient 4 had a “white hole” MHRD, with macular hole closure achieved on postoperative day 20, albeit with a suboptimal foveal shape.
Conclusion
The temporal inverted ILM flap technique in conjunction with vitrectomy facilitates foveal reconstruction in patients with successful treatment of MHRD, and this reconstruction process can be observed by optical coherence tomography.
Keywords
Introduction
In 1991, Kelly and Wendel 1 used vitrectomy with inverted internal limiting membrane (ILM) removal to treat macular holes (MHs), achieving visually and anatomically satisfactory outcomes. This approach has since become an important part of the surgical repair of idiopathic MHs and is currently used to treat MHs and MH-associated retinal detachment (MHRD) with high myopia. 2 However, although inverted ILM removal can achieve satisfactory results in treating idiopathic MHs, 3 the U-shaped closure is most conducive to restoring optimal function in only 45% of cases. Furthermore, large MHs and highly myopic MHs, especially MHRD, often increase the risk of surgical failure. 4
In 2009, Michalewska et al. 4 introduced vitrectomy with the inverted ILM flap technique, which includes retaining the entire 360° of the ILM with a diameter of approximately 3 mm attached to the edge of the MH. The residual ILM is then inverted to cover the MH, increasing the closure rate of MHs of >400 μm in diameter and improving anatomical recovery and function.4–9 This approach has since been extended to serve as an effective complement to the treatment of highly myopic MHs, promoting MH closure, fovea reconstruction, and improved visual acuity.10–13 Furthermore, as a new surgical strategy, it can greatly improve the treatment success rate of MHRD with high myopia, achieve significantly better retinal reattachment and MH closure rates, and provide better visual function recovery compared with vitrectomy alone combined with ILM peeling.14,15
ILM peeling may lead to complications such as paracentric neuroretinal holes and optic nerve fiber layer separation. Excessive glial hyperplasia may also occur, adversely affecting retinal ellipsoid zone (EZ) recovery and postoperative visual function improvement.7,10,16,17 Michalewska et al. 16 adopted a modified form of limiting the peeling of the ILM to the temporal side of the fovea and inverting it to cover the MH. This new approach, which they termed the temporal inverted ILM flap technique, leaves the nasal-side ILM intact, thus reducing the risk of surgical trauma in the papillomacular bundle area. Repairing stage IV MHs has the same anatomical reattachment effect and the same or even better effect on the recovery of visual function compared with the traditional classical flipped ILM flap technique.17,18 In the present study, we reviewed and analyzed the temporal inverted ILM flap technique in treating four typical cases of MHRD with high myopia and examined the different approaches to foveal reconstruction.
Patients and methods
We performed a retrospective case series analysis of the temporal inverted ILM flap technique in four consecutive patients with typical high myopic MHRD treated from October 2014 to October 2020 at the Department of Ophthalmology, Harbin Bright Eye Hospital (Harbin, China), a tertiary hospital. The study was approved by the ethics committee of Harbin Bright Eye Hospital (approval #2022017). Verbal informed consent for both treatment and publication was obtained from all four patients. The reporting of this study conforms to the STROBE guidelines. 19
Ophthalmic findings at the time of presentation were obtained from the patients’ charts and included best-corrected visual acuity (BCVA), axial length, B-scan ultrasonography findings, laser fundus photography findings, intraocular pressure (IOP), and optical coherence tomography (OCT) findings. The OCT devices were an Optovue OCT system (Visionix, Pont-de-l'Arche, Normandie, France) and ZEISS CIRRUS HD-OCT 5000 (ZEISS Group, Oberkochen, Germany), using a macular cube of 512 × 128. Age, sex, preoperative and postoperative BCVA, axial length, IOP, subretinal fluid (SRF) drainage, type of tamponade, follow-up, time to reattachment, time to macular closure, and final foveal status were collected.
The surgical method was as follows. Intraoperative staining with 20% triamcinolone acetonide was performed to completely remove the vitreous, followed by ILM staining with indocyanine green (diluted to 0.125 mg/mL with 5% glucose water for approximately 20 seconds). After creating a semicircular ILM notch from the temporal edge of the MH, the ILM flap was flipped toward the nasal side to completely cover the MH (Figure 1(a)–(c)), and a low-molecular-weight viscoelastic agent was used for fixation (Figure 1(d)). Postoperative OCT was then utilized to assess alterations, determining successful reattachment of the retina and reconstruction of the fovea following MH closure.
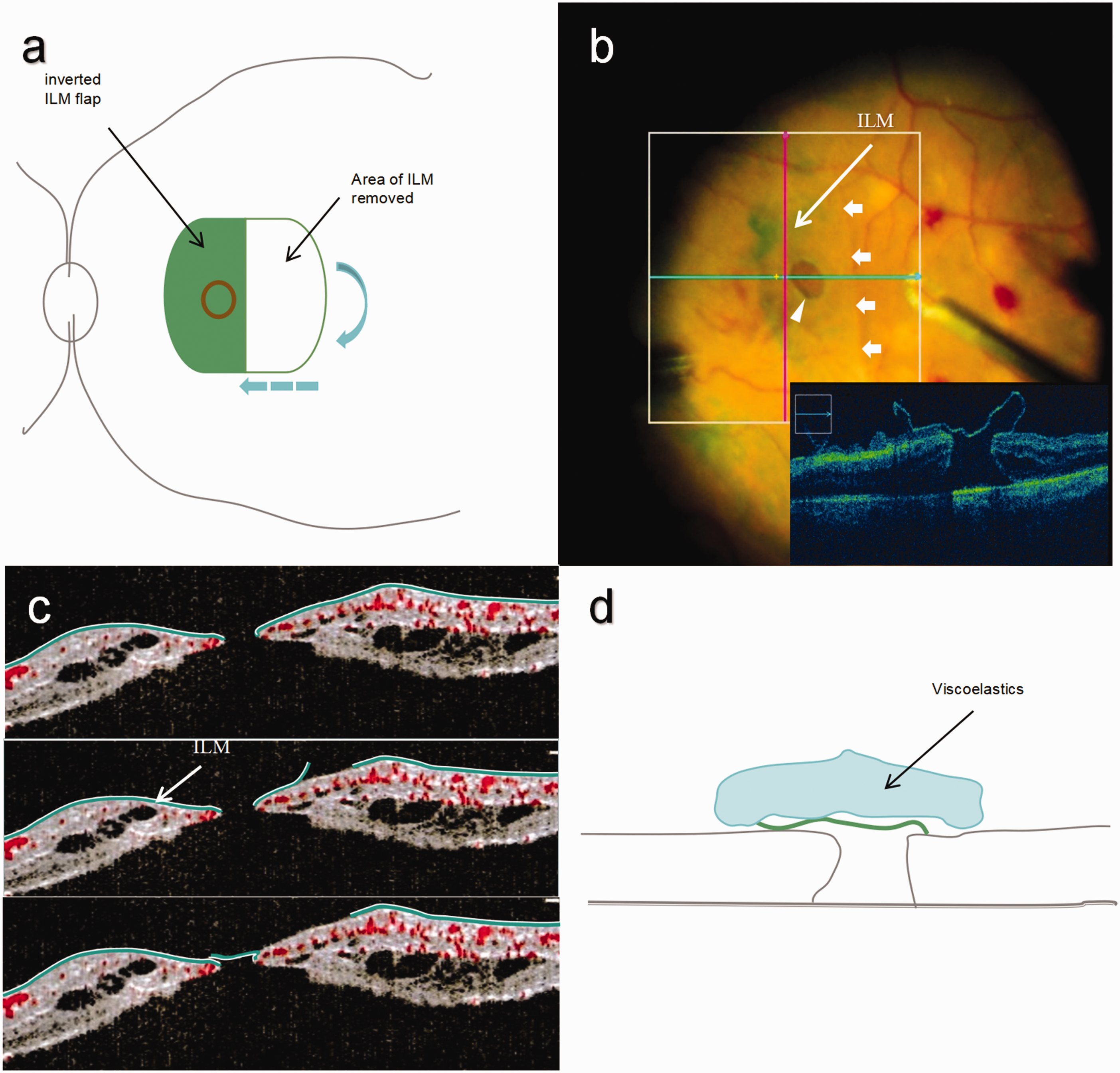
Schematic diagram of temporal inverted limiting membrane (ILM) flap technique. (a) After creating a semicircular ILM notch (blue arrow) from the temporal edge of the macular hole (MH) (red circle), the indocyanine green-stained ILM flap that formed on the temporal side of the macula was flipped toward the nasal side (blue dotted arrow) to cover the MH completely. (b) Cross-sectional view of the temporal inverted ILM flap technique (intraoperative optical coherence tomography). The figure shows the temporal-side ILM flap flip line (white arrow) toward the nasal retinal reversal covering the MH (white triangle). (c) The ILM peels off to the edge of the MH. The ILM part facing the vitreous is white, while the ILM part facing the retinal tissue is green. The peripheral parts of the ILM were trimmed. The MH was closed with an inverted ILM flap. The ILM flap facing the vitreous is green, while the ILM flap facing the retinal pigment epithelium is white. This indicates that the ILM flap was inverted and (d) A low-molecular-weight viscoelastic agent was used for fixation (arrow).
Results
Patient 1
Patient 1 was a 66-year-old woman (Table 1) who presented to our hospital because of a 3-day history of vision loss in the left eye. She had a >40-year history of high myopia. The spherical equivalent BCVA in the left eye before surgery was −9.50 = 0.01. Laser fundus photography (Daytona; Optos, Dunfermline, Scotland) showed a tessellated fundus, an atrophic arc next to the optic disc, diffuse atrophy of the posterior polar choroid, and superficial retinal detachment in the macular area (Figure 2(a) and Supplementary Figure S1). B-scan ultrasonography revealed speckled vitreous body opacity, posterior scleral staphyloma, superficial retinal detachment, and axial length of 29.32 mm. The IOP in the left eye was 16 mmHg (1 mmHg =0.133 kPa). OCT showed separation of the macular neuroretina from the pigmented epithelial layer (Figure 3(a)). The patient underwent vitrectomy. During the surgery, the vitreous was completely removed, and Brilliant Blue-Green ILM stain (diluted to 0.125 mg/mL with 5% glucose solution for approximately 20 seconds) was then applied. A temporal ILM flap was created using a retinal brush and ILM forceps. After performing a semicircular ILM incision at the temporal edge of the MH, the ILM flap was flipped toward the nasal side to completely cover the MH, and low-molecular-weight viscoelastic agents were used for fixation. Intraoperative drainage of the SRF was not performed because the postoperative macular hole was well-covered by the ILM, and removing the SRF at this time had a risk of flipping the ILM. Silicone oil was used for filling.
Patients’ characteristics

BCVA: best-corrected visual acuity; IOP: intraocular pressure; SRF: subretinal fluid; OCL: outer collagenous layer.
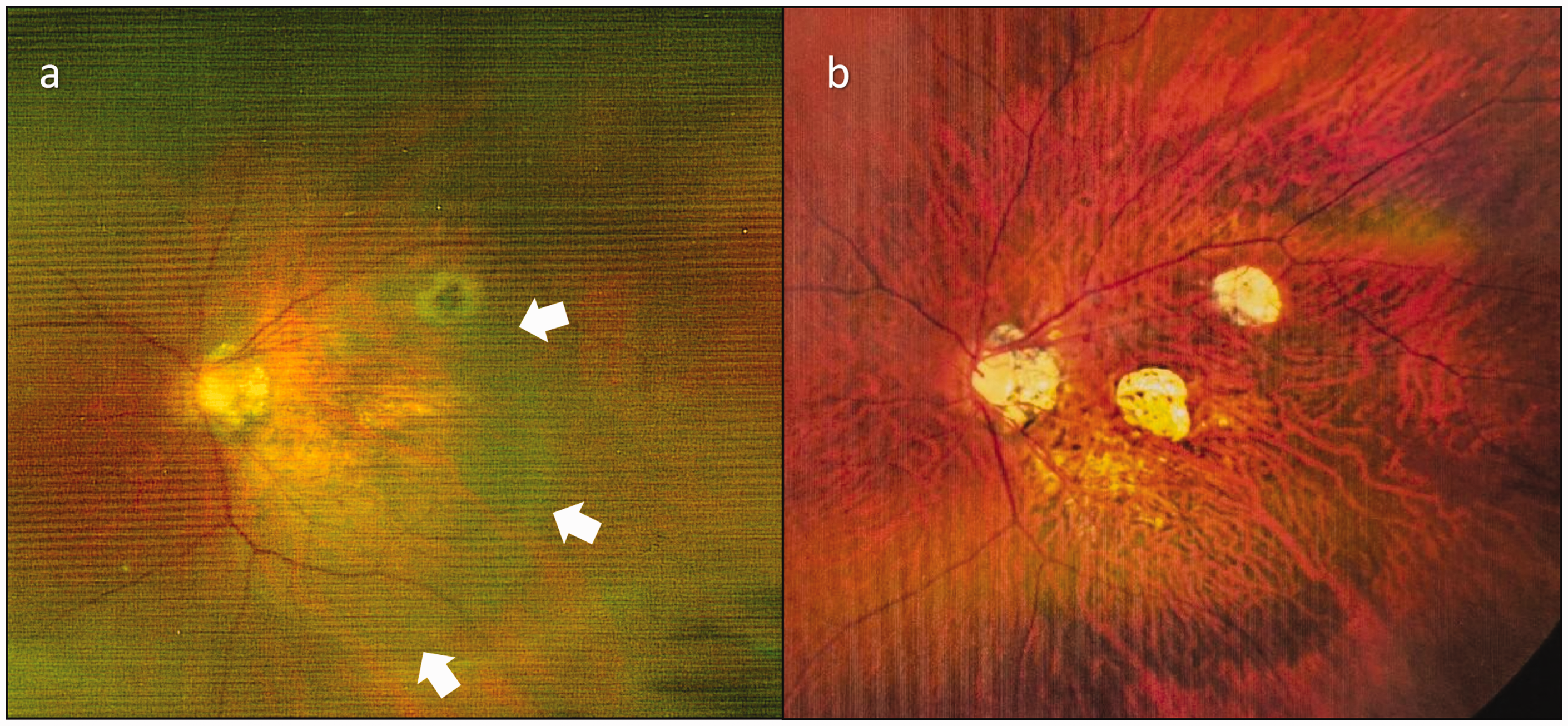
Patient 1, fundus image before and after left eye surgery. (a) Photograph of the preoperative superficially detached fundus of the macular area (white arrows) and (b) Nineteen months after surgery.

Patient 1, optical coherence tomography images. (a, c–f) Horizontal scan. (b) Vertical scan. (a) Preoperative loss of the full retinal thickness in the macular area, macula hole formation, and retinal detachment. (b) On postoperative day 6, the macular hole margin was connected by an inverted limiting membrane flap bridge. (c) Twenty-three days after surgery, the integrity of the nerve fiber layer of the central retina of the macula was restored. The retina was still detached. (d) Eighty-three days after surgery, the center of the macula was completely reduced, and the retinal temporal side had persistent subretinal fluid. (e) A small amount of subretinal fluid remained on the temporal side of the retina 18 months after surgery and (f) One month after silicone oil removal (approximately 19 months after application of the temporal inverted limiting membrane flap), the retina was completely reduced, the subretinal fluid had disappeared, and the macular fovea was formed.
The postoperative OCT examination findings were as follows. On postoperative day 6, the MH was covered by the ILM flap, forming a bridge-like connection. The nasal macular retina was basically reduced, and the temporal retina, including the center of the macula, was still detached. On postoperative day 23, OCT showed restoration of the nerve fiber layer integrity in the central macular retina. The center of the macula and the temporal retina of the macula were still detached. On postoperative day 83, OCT showed complete reattachment of the macular center and persistent SRF under the temporal side retina. The retina was basically reduced, and a small amount of SRF remained on the temporal side of the retina at 18 months. The patient underwent cataract phacoemulsification removal, intraocular lens implantation, and silicone oil removal. One month after surgery (about 19 months after ILM reversal), the retina was completely reduced and the macular fovea was formed (Figure 3(f)). The contraction and fusion of residual Müller cell structures in the flipped ILM valve led to closure of the bridging hole (Figure 3(d)). Over time, OCT showed that the bridged MH gradually closed, the SRF gradually disappeared, and the macular structure was reconstructed. By removing the temporal portion of the ILM around the hole (considered a challenging aspect of the retina because of its rigidity), the proliferation of glial tissue around the hole was simultaneously removed. This led to retinal relaxation, relieving the traction causing the hole and promoting the process of central fovea reconstruction. Fundus images (ZEISS CLARUS 500; Zeiss Group) showed retinal reattachment with two limited choroidal atrophic plaques at the posterior pole (Figure 2(b)). The BCVA in the left eye was 0.04.
Patient 2
Patient 2 was a 55-year-old woman (Table 1) with a chief complaint of unclear vision in her left eye for approximately 2 weeks. The patient had a history of high myopia. The initial visual acuity of her left eye was hand movements. Corrective vision resulted in no improvement. Fundus photography showed a tessellated fundus, an atrophic arc next to the optic disc, diffuse atrophy of the posterior polar choroid, and superficial retinal detachment in the macular area and MHs (Figure 4 and Supplementary Figure S1). B-scan ultrasonography showed speckled vitreous body opacity, posterior scleral staphyloma, and superficial retinal detachment. The IOP in the left eye was 10 mmHg. The patient underwent vitrectomy, during which the vitreous was removed completely and Brilliant Blue-Green ILM stain was applied. The SRF was adequately excluded through the MH following application of a temporal inverted ILM flap under perfluorocarbon gas–liquid exchange and silicone oil filling. The postoperative OCT examination showed the following. On postoperative day 4, the MH was still present; the maximum diameter above the hole was 797 μm, and the bottom of the hole was 407 μm. The ILM was flipped to the inside of the MH, and ILM folds were visible. At 14 days postoperatively the MH had basically disappeared, and the macular center was connected by hyperreflective matter. At 52 days, the MH had healed, and the outer retinal layer in the center of the fovea was missing. At 3 months, fovea formation of the outer layer of the retina was observed (Figure 4(e)). A flipped ILM was observed lining the inner wall and bottom of the MHRD (Figure 4(b)), and both sides of the retinal nerve tissue extended into the hole, resulting in complete healing of the hole. Cataract phacoemulsification removal, intraocular lens implantation, and silicone oil removal were performed, and the postoperative BCVA was 0.1.

Patient 2. (a) Heidelberg laser fundus photography before surgery. Postoperative optical coherence tomography image (horizontal scan). (b) On the fourth day, the hole was still visible; the maximum diameter above the hole was 797 µm. (b) The inverted limiting membrane was flipped to the inside of the macular hole; the inverted limiting membrane fold (white arrow) was visible, and the retina was completely reduced. (c) On day 14, the macular hole had basically disappeared, and the macular center was connected by hyperreflective matter. (d) On day 52, the macular hole had healed, and the outer retinal layer in the center of the fovea was missing and (e) Formation of the outer retinal layer was seen in the fovea 3 months after surgery.
Patient 3
Patient 3 was a 57-year-old woman (Table 1) who presented with a 30-day history of vision loss in her left eye. The patient had been highly myopic in the past (eye axis of 28.15 mm). The visual acuity of the left eye before the operation was 0.02, and it had not improved with correction. Fundus color photography showed a tessellated fundus, diffuse choroidal atrophy in the posterior pole, and peripapillary atrophy. Ultrasound revealed speckled opacity (Supplementary Figure S1). The IOP in the left eye was 13 mmHg. OCT examination revealed loss of the full thickness of the retina and fracture in the macular area, maximum hole diameter of 598 μm, cystic lesions in the Henle fiber layer of the hole wall, and local retinal detachment. Surgery was performed. Complete removal of the vitreous body was surgically performed, followed by staining of the ILM. During vitrectomy, posterior-pole retinal detachment occurred with an approximate extent of 3 prism diopters. A temporal ILM flap was created using a retinal brush and intraocular forceps, and an enlarged scope of retinal detachment occurred during the ILM flap creation process (approximately 2 disc diameters around the hole). After thoroughly removing the SRF around the MH, the ILM flap was flipped nasally to completely cover the MH. A low-molecular-weight viscoelastic agent (0.7 mL of C3F8) was used for fixation with the assistance of heavy water. On postoperative day 25, Heidelberg laser fundus photography showed that approximately half of the gas remained, and the retina was well reduced. Additionally, OCT showed that the hole was closed on postoperative day 25 and that the fovea was well formed. The ILM flap and its lower gaps were visible on the surface. The foveal center outer collagenous layer was interrupted, and the macular area external limiting membrane (ELM) and EZ were missing. At 2 months, hyperplasia was observed under the ILM, the spaces below the ILM remained, the central fovea outer collagenous layer was connected, and the ELM and EZ were restored (Figure 5(c), (d)). The BCVA was 0.3.
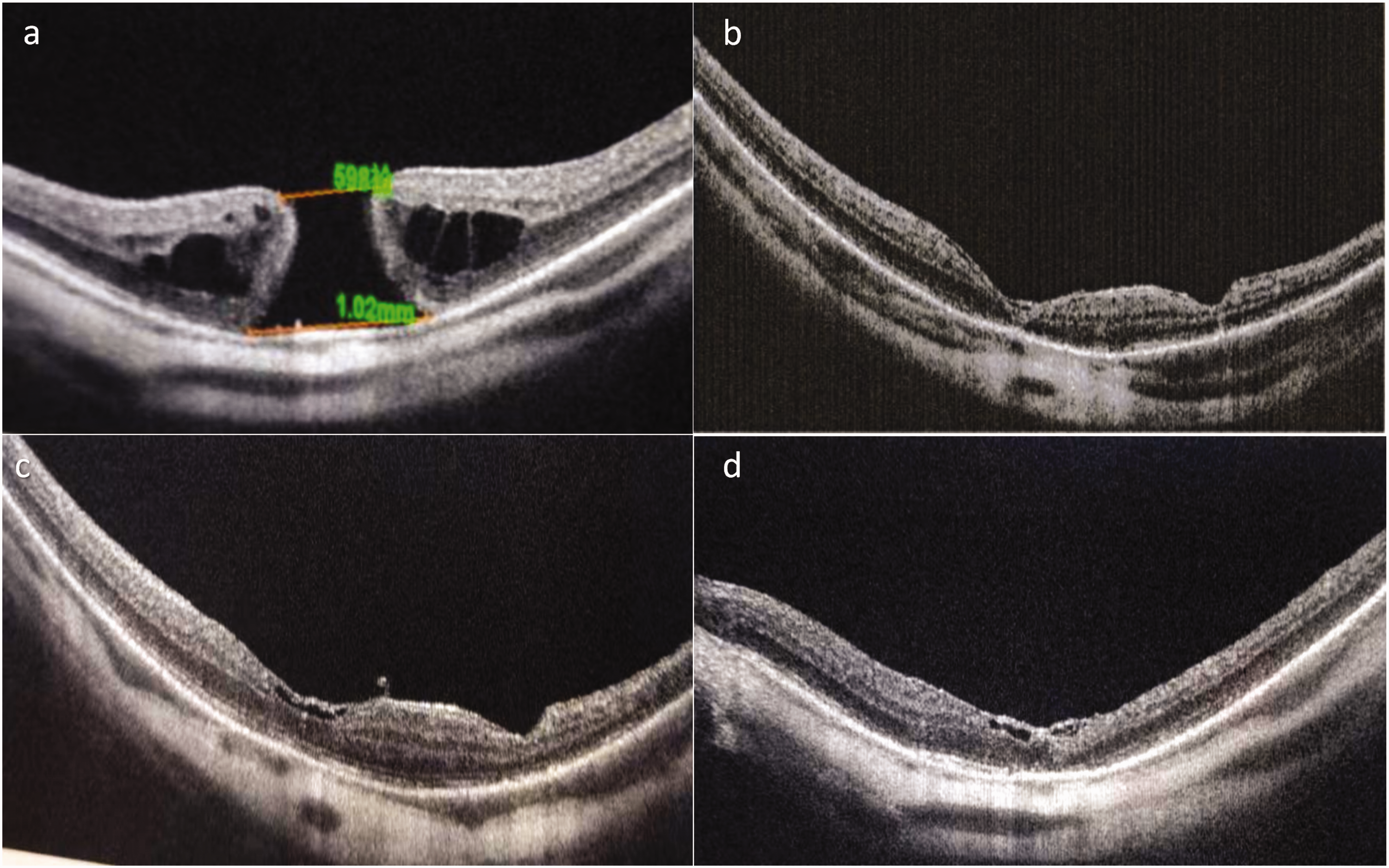
Patient 3. Optical coherence tomography images. (a–c) Horizontal scan. (d) Vertical scan. (a) Before surgery, the maximum diameter of the hole was 598 µm. (b) On day 25 after surgery, the hole was closed and the fovea was well formed and (c, d) Two months after surgery, hyperplasia was observed under the internal limiting membrane, the spaces below the internal limiting membrane remained. The central fovea outer collagenous layer was connected, and the external limiting membrane and ellipsoid zone were restored.
Patient 4
Patient 4 was a 61-year-old woman (Table 1) who presented with a 20-day history of vision loss in her left eye. The patient had been highly myopic in the past. The vision in her left eye was limited to hand movements and could not be improved by correction. Heidelberg laser fundus photography showed a tessellated fundus, diffuse atrophy of the posterior polar choroid, and a macular white hole (i.e., localized area of the sclera appearing white, making it difficult to observe retinal fissures against the white background) (Figure 6(a)). B-scan ultrasonography showed speckled vitreous body opacity, posterior scleral staphyloma, and superficial retinal detachment (
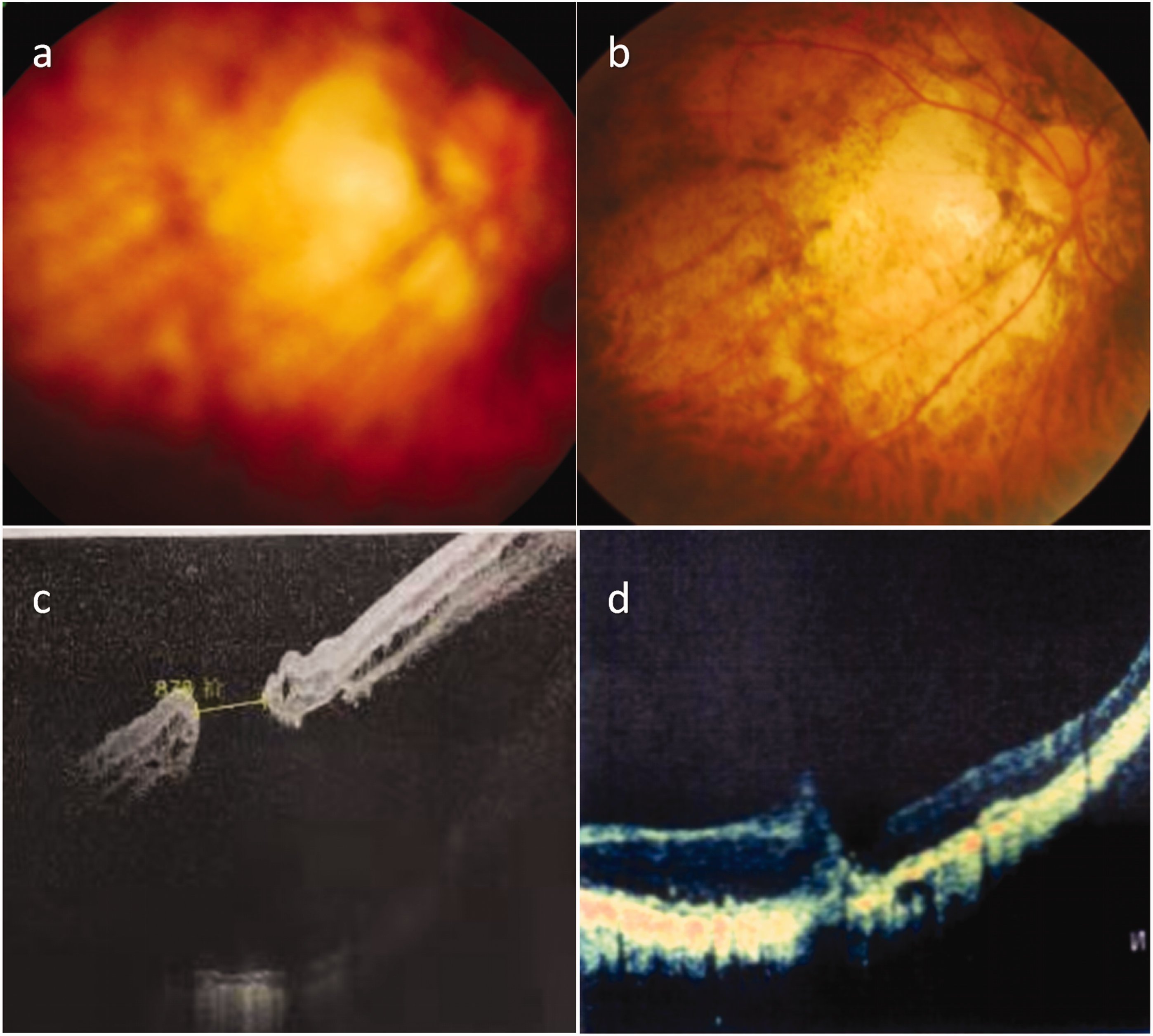
Patient 4. (a) Fundus photography shows a tessellated fundus, diffuse atrophy of the posterior polar choroid, and a macular white hole. (b) On day 20, fundus photography showed good retinal reattachment and diffuse choroidal atrophy at the posterior pole and white fundus. (c) Optical coherence tomography showed preoperative retinal detachment and (d) Twenty days after surgery, hole closure and scarring were evident below the poor foveal morphology.
Discussion
MHRD in highly myopic eyes is a challenging condition for vitreoretinal surgeons. Anatomic reduction can be achieved by vitrectomy, retinal ILM extraction, and filling with gas or silicone oil; however, only a limited MH closure rate of approximately 47% can be achieved. 20 The use of vitrectomy and inverted ILM flap technology to treat highly myopic MHRD can greatly improve the success rate of surgery. In previous studies, the postoperative MH closure rate and retinal reattachment rate were significantly higher than those achieved with vitrectomy alone combined with ILM peeling.14,15 Ho et al. 21 treated MHRD with high myopia using a modified C-shaped temporal inverted ILM flap technique and performed a 12-month follow-up. Although persistent superficial SRF was observed in one case 11 months after surgery, 100% of the MHs eventually closed, and retinal reattachment was achieved. The BCVA significantly improved and the macular structure was regained in 94.4% of the eyes. Additionally, the foveal EZ recovered in 77.8% of patients. Vitrectomy combined with a modified C-shaped temporal inverted ILM flap technique is considered an ideal surgical method to help rebuild the foveolar architecture. The normal fluid flow from the vitreous cavity to the choroid is driven by osmotic pressure, hydrostatic pressure, and active transport mechanisms. The retina provides some resistance to fluid movement from the vitreous to the choroid, promoting adhesion of the retina to the retinal pigment epithelium (RPE) and thus preventing fluid from passing through the retina. In the MH state, the barrier disappears, leading to retinal detachment. 22
Retinal and scleral mismatch can be due to posterior staphyloma in patients with high myopia. The relatively insufficient area of the retina leads to increased tension, making it difficult for MHs to heal under tension. Inverted ILM flaps repair the defective retina and can completely release the tangential traction of the ILM.23,24 Classic inverted ILM flap techniques result in irregular ILM shrinkage and abnormal foveal structures 3 months postoperatively. The method applied in the present study involves temporal ILM coverage of the MH, which allows for the release of more traction from the temporal side. As a single-layer structure, the ILM flap covering the hole contributes to better restoration of the foveal structure. Simultaneous preservation of the nasal papillomacular bundle may help to achieve better surgical outcomes.
In Patient 1, OCT on postoperative day 6 revealed a single-layer ILM that completely covered the MH. By day 23, the integrity of the retinal nerve fiber layer in the center of the macula had been restored. However, detachment persisted in the center of the macula and the temporal retina of the macula. On postoperative day 83, central macular reattachment and sustained SRF under the temporal side of the retina were observed. A small amount of SRF remained on the temporal side at 18 months, and complete retinal reattachment occurred about 19 months after coverage with the ILM. These findings indicate that application of the ILM to cover the MH and restore the integrity of the retina was effective in treating MHRD. Covering the MH with a single layer of ILM prevents fluid from flowing from the vitreous cavity to the retina, creating a relatively closed environment at the hole. In addition, a completely sealed macula allows SRF to be progressively absorbed by the RPE pump. Although the function of the RPE pump is impaired in some highly myopic individuals, closure of the MH does not necessarily require retinal reattachment.21,25
The Müller cell cone (Mcc) is an inverted cone-shaped region of Müller cells that constitutes the medial part of the fovea. Cross-sectional apices are located in the outer membrane and the base forms the base of the fovea, and the region extending into the surrounding slope area. The long outer cone fibers extend anteriorly and outward from the outer membrane and radiate to the surrounding optic cone nucleus, which may be the main supporting structure of the fovea.26,27 The Mcc may be quite vulnerable in highly myopic eyes because of extreme elongation of the axial length and chorioretinal atrophy, and there may be significant differences in the mechanism of formation of highly myopic MH versus idiopathic MH. 28 Posterior vitreous detachment occurs earlier in highly myopic eyes but is often incomplete. Vitreomacular interface abnormalities (e.g., epiretinal membrane, residual vitreous cortex after posterior vitreous detachment, and incomplete posterior vitreous detachment) and ocular deformities are considered to be the two main mechanisms of MH formation in patients with high myopia. 28 When an MH is formed, the posterior vitreous body becomes attached to the fovea. This attachment exerts forces on the fovea in both the forward and backward directions, particularly at the site of low mechanical stability where there is no cellular connection between the Müller cell layer and the outer nuclear layer (ONL). Detachment of the ONL from the Müller cell layer within the fovea leads to extension and destruction of the Mcc stalk. This process leads to the formation of a large pseudocyst in the fovea and a horizontal gap between the central ONL. This gap is further manifested within the central ONL. Disruption of the Müller cell layer in the fovea produces a full-thickness hole. Subsequent enlargement of the cystic cavity of the fovea creates a protrusion of the retinal tissue and the center (ELM) around the hole, resulting in narrowing of the pores at the level inside the ONL.29,30
MHs of <400 μm can occasionally close spontaneously through a mechanism that may be mediated by Müller cells. In addition, the annular contraction of the lateral process of Müller cells surrounding the hole at the OPL level and the annular contraction of the Müller cell structure enveloping photoreceptor cells located in the ELM lead to a centripetal shift of the central photoreceptor cell body, guiding the centripetal shift of the foveal wall. The mechanism of MH closure in Patient 1 may have included traction and fusion of residual Müller cones, whereby Müller cell structures cause pore bridge closure (Figure 3(d)). Assuming remnants of the Müller cell layer exist in the fovea, these cells remaining in the foveal wall regenerate to form an Mcc after the hole is closed. When the MH diameter is larger than the foveal diameter (average of approximately 350 μm), nonfunctional closure of the MH may occur; i.e., the ONL shifts toward the center, the hole closes only at the ELM, and no new Mcc forms. 29 After the hole is closed, the fovea has two regeneration modes: either regular regeneration with photoreceptors in the center or irregular regeneration, where the center does not contain photoreceptors and the foveal center is filled with tissue formed by Müller cells and RPE cells. This may be one of the reasons why vision only slightly improves after surgery despite successful closure of the hole. 30
The MH ILM removal technology completely strips away the structure of glial tissue proliferation around the hole (the hardest part of the retinal ILM), resulting in retinal relaxation. The traction force causing the MH is alleviated, triggering activation of epidermal growth factor receptor and upregulation of glial fibrillary acidic protein by damaging Müller cells. 31 Activation of Müller cells and stimulation of reparative glial cell regeneration are the mechanisms of MH closure.2,21 The U-shaped closure rate of ILM peeling is 100% only when the diameter of the hole is <300 μm. 9 Larger nerve defects are difficult to connect by glial tissue. 2 Inverted ILM flap or transplantation techniques, which potentially act through the tear of the terminal foot of Müller cells during ILM peeling, lead to activation of Müller cells and stimulate regeneration. The three main components of the ILM, namely type IV collagen, fibronectin, and laminin, can promote proliferation of ILM cells, and type IV collagen and fibronectin can enhance their migration. The small amount of ILM retained around the MH after ILM flipping can cause glial cell proliferation in the retina and on the surface of the ILM. The flipped and transplanted ILM flap simultaneously fills the MH space, and the Müller cell fragments it contains are activated to proliferate and serve as a scaffold for the proliferation and migration of Müller cells in the MH wall. 27
Müller cells retained on the surface of the ILM flap placed in the MH and Müller cells that have migrated through activation secrete neurotrophic factors and basic fibroblast growth factor to promote the survival of retinal neurons and photoreceptor cells. Müller cell proliferation induced by an inverted ILM flap and the release of factors such as neurotrophic factors and basic fibroblast growth factor lead to the closure of MHs. 32 The disadvantage of the inverted ILM flap technique and autologous ILM transplantation is that continuous activation of glial cells may lead to excessive proliferation of glial cells and a poor visual prognosis. 32 If the Mcc is destroyed before surgery, the Mcc residue is missing. In addition, irreversible damage to the photoreceptor layer caused by atrophy may lead to direct contact between the ONL and RPE, preventing normal and regular foveal regeneration. Moreover, retraction of the outer layer of Müller cells that envelops the photoreceptor cells can lead to centrifugal displacement of photoreceptor cells. 33 In the temporal inverted ILM flap technique, a large monolayer ILM flap provides a more stable structure for glial cell proliferation. As glial cells present and adhere to the ILM flap, sticking to the retinal surface due to ILM turnover and being unable to migrate to the fully sealed MH space, the proliferation of glial cells within the MH wall also has an important role in MH closure. The temporal inverted ILM flap provides a basement membrane for MH wall glial cell proliferation. MH closure begins at the top of the MH space under the covered ILM flap; however, excessive proliferation of glial cells affects the recovery of the extraretinal band.4,17 Ho et al. 21 suggested that vitrectomy combined with a modified C-shaped temporal inverted ILM flap can help restore the foveal structure. They proposed that the Mcc could be repaired after MHRD surgery. In the present study, Patient 2 had an inverted ILM lining the inner wall of the MHRD and the bottom of the hole (Figure 4(b)). Although a small ILM flap that migrates to the base of the MH may present a barrier to natural closure of the MH and functional recovery of the outer retina, 17 the MH healing was not affected in this case. The retinal nerve tissue on both sides crawled into the hole, thus allowing the MH to heal completely. This indicates that turning over a large smooth ILM flap can still act as an ILM and promote healing of the hole even if it falls into the bottom of the hole. In Patients 1 and 2, the hole was in the outer plexiform layer and the ONL; thus, the MH closure mechanism was an active rather than passive process. This is identical to the spontaneous closure mechanism of the MH (i.e., a single-layer ILM flap covering the MH leads to Müller cell proliferation and induces spontaneous closure mediated by Müller cells). Similar mechanisms are involved in MH bridge closure, foveal reconstruction, and Mcc repair. Bringmann et al. 30 suggested that contact between the central ONL and RPE may hinder the normal regeneration process of the foveal region. In Patient 2 of the present study, the contact between the central ONL and RPE after application of the monolayer inverted ILM did not prevent regular regeneration of the fovea (Figure 4(e)). In Patient 3, the covering ILM flap was suspended, and the visible space with the retina below and the lower macular fovea was reconstructed. Two months after surgery, the ILM flap was characterized by hyperplasia and thickening, and its center was connected to the fovea, while ELM and EZ recovery were visible in the outer layer of the fovea (Figure 5(c), (d)). We believe that this type of macular fovea reconstruction was not related to the ILM flap but positively affected later Mcc repair. Even if the macular fovea is poorly repaired or an irregular nonfunctional foveal structure appears, use of the single temporal layer inverted ILM flap technique for treatment of MHs in high myopia is still clinically significant because it can promote retinal reattachment, reduce the central scotoma, and improve certain aspects of visual function. Patient 4 had an MH with retinal detachment and choroidal atrophy (white hole), and retinal reattachment and improvement of poor visual acuity were still achieved after surgery. Treating macular white-hole retinal detachment is very challenging because the ILM flap is difficult to form, hindering coverage of the MH with the ILM. In addition, white holes can hardly form effective retinal adhesions. The cause of the poor formation of the white-hole fovea may be the presence of a scar under the macular fovea before retinal detachment. The ELM and EZ recovery observed by OCT in such patients is poor, which may be related to a short retinal detachment time or short observation time. Michalewska et al. 11 found that the retinal foveal structural repair process persisted for at least 12 months in patients who had MHs without retinal detachment. In Patient 1 of the present study, the postoperative observation time was 19 months, after which time the ELM and EZ remained poorly recovered. The outer layer of the MHRD may require prolonged recovery, or that part of the outer layer may not be able to fully recover. Moreover, most highly myopic MHRDs are combined with scleral staphyloma and retinal choroidal atrophy, resulting in poorer retinal recovery. During the observation period in the present study, Patient 1 exhibited aggravation of posterior polar limited choroidal retinal atrophy and enlargement of the atrophy area while the retinal structure deteriorated.
Surgical repair of MHRD is a challenging procedure, and fixing the inverted ILM during surgery can be difficult. There is a risk of ILM displacement or loss during gas–fluid exchange or silicone oil filling. Through experimentation and practice, we developed a technique for creating an ILM flap using a diamond-dusted membrane scraper during surgery; the flap is then secured with heavy water and adhesive to prevent its displacement. This technique has improved the treatment success rate to approximately 80%. Most unsuccessful cases are due to ILM displacement and detachment. Nevertheless, smaller MHs can heal, whereas larger ones may require additional surgery. The process of MH reconstruction involves providing a scaffold structure for repair. We have also attempted treatments involving amniotic membrane coverage and filling the lens capsule, resulting in a central foveal reconstruction process similar to ILM flipping. In most cases, MHs can heal successfully.
This study had two main limitations. First, it involved only four patients, all of whom were treated at the same institution by the same surgeons. Second, the study was retrospective in nature, which limited the available data to those within the patients’ charts.
In conclusion, in patients successfully treated by vitrectomy combined with the ILM flap technique, the process of foveal reconstruction can be observed by OCT. Reconstruction of the macular fovea can occur in four ways. (1) In bridge-healing, the MH can be bridged by closure over the residual SRF and does not necessarily require retinal reattachment. 17 The single-layer temporal inverted ILM flap technique provides a scaffold for retinal gliosis and may promote bridge formation between MH walls under the ILM flap. (2) With in situ healing, the ILM is lined with the inner surface of the MH, and the retina around the MH gradually transitions and heals. (3) With independent healing of the ILM flap, MH healing may not be related to ILM flap coverage but may instead be related to later foveal Mcc repair. (4) Finally, poor foveal healing may occur.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231223635 - Supplemental material for Temporal inverted internal limiting membrane flap technique for myopic macular hole retinal detachment reconstruction
Supplemental material, sj-pdf-1-imr-10.1177_03000605231223635 for Temporal inverted internal limiting membrane flap technique for myopic macular hole retinal detachment reconstruction by Yan Teng and Xiaoyu Zhang in Journal of International Medical Research
Footnotes
Authors’ contributions
Both authors participated in case collection, data collection, data analysis, and manuscript writing.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare that they have no competing interests.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
