Abstract
Objective
To examine the use of platelet-rich plasma (PRP) for treatment of pilonidal disease (PD) and thus provide a reference for clinical application.
Methods
A systematic review of PubMed and the Cochrane Library was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses. We considered all studies that reported the use of PRP for treatment of PD. Extracted data included the first author’s name, year of publication, study type, number of included patients, inclusion and exclusion criteria, interventions, anesthesia, application of PRP (source, preparation, dose, and operation), antibiotics, follow-up time, therapeutic outcomes, and adverse events.
Results
In total, eight randomized controlled trials and one prospective cohort study involving 809 patients were included. PRP reduced pain, accelerated healing, and reduced adverse events. The application of combined minimally invasive surgery achieved better results. However, overfilling of the wound with PRP in minimally invasive surgeries was shown to potentially increase the risk of adverse events.
Conclusion
PRP can be used as an adjuvant treatment in PD surgery to improve the therapeutic effect and reduce adverse events. The optimal combination of PRP and various factors is an important direction of future research.
Keywords
Introduction
Pilonidal disease (PD) is usually acquired and closely associated with the presence of hair in the gluteal cleft; thus, it is also called pilonidal sinus or pilonidal sinus disease. 1 Surgical excision is the traditional standard treatment for PD and is generally divided into two categories: excision of diseased tissue with primary closure (including midline or off-midline sutures and flap techniques) and excision with healing by secondary intention (including open healing and marsupialization).2–4 However, the benefits of open healing versus primary closure are controversial and include factors such as the duration of time required for healing, duration of time unable to perform daily activities or work, surgical site infection, and recurrence. In recent years, new minimally invasive procedures such as laser pilonidoplasty have shown good mid-term outcomes in patients with early PD. 5 Some studies have used advanced dressings to provide an optimal environment for wound healing, typically by optimizing wound cleansing and re-epithelization. 6
The use of blood derivatives in promoting wound healing began with fibrin glue, which is composed of concentrated fibrinogen (polymerization induced by thrombin and calcium). 7 In patients with chronic PD without abscess formation, fibrin glue may be effective as a primary or adjunctive treatment. 2 In the past dozen of years, for PD, a relatively novel blood product called platelet-rich plasma (PRP) has also been developed for PD, and it has gained more positive results.
PRP is a type of platelet concentrate that was originally used for the treatment and prevention of hemorrhage due to severe thrombopenia. Platelet concentrates are divided into four categories according to their leukocyte and fibrin content: pure platelet-rich plasma, leukocyte- and platelet-rich plasma, pure platelet-rich fibrin, and leukocyte- and platelet-rich fibrin. 8 Previous studies have shown that PRP significantly improves wound healing in chronic diabetic ulcers, venous ulcers, pressure ulcers, leprosy ulcers, acute traumatic wounds, and ulcers of multifactorial etiologies. 9 Although some studies have also focused on the application of PRP in anorectal disease, these studies vary greatly in their trial design, material preparation, and application methods, and a summary of the common features of these studies is lacking.
In this review, we examined the recent advances in the application of PRP in PD to provide a reference for its clinical application.
Methods
All studies reporting the use of PRP for the treatment of PD up to July 2023 were considered. The review was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, 10 and the PRISMA checklist is available in this review. PubMed and the Cochrane Library were searched using the Medical Subject Headings terms “platelet-rich plasma” and “pilonidal disease.” Additional studies were identified by searching the reference lists of eligible articles. Case reports were excluded from the review. All studies were reviewed by two independent reviewers. Any discrepancies in selection were discussed until a final consensus was reached. The extracted data included the first author’s name, year of publication, study type, number of included patients, inclusion and exclusion criteria, interventions, anesthesia, application of PRP (source, preparation, dose, and operation), antibiotics, follow-up time, therapeutic outcomes, and adverse events. The Cochrane Handbook Version 5.2.0 and RevMan 5.3 were used to assess the biases of selection, performance, detection, attrition, and reporting of randomized controlled trials. This assessment was independently performed by the same two reviewers, who reached a consensus on all items. We also conducted a summary independent-samples t test of the healing time of all studies that employed the same surgical methods. For this purpose, we selected studies in which there were no significant differences in the control groups but in which there were significant differences in PRP groups to explore the effect of different PRP application methods on efficacy. Finally, we summarized the number of adverse events in all studies and calculated the total incidence. Figures were created or modified with Adobe Illustrator Version 2019 (Adobe Inc., Mountain View, CA, USA). Statistical analysis was performed using SPSS Version 25.0 (IBM Corp., Armonk, NY, USA). A two-tailed test was used, and a p value of <0.05 was considered statistically significant. In error, we did not prospectively register this trial; however, we registered it retrospectively at INPLASY (https://inplasy.com/) (registration number INPLASY2023100070).
Neither ethics approval nor patient consent for publication was required because of the nature of this study (systematic review).
Results
Nine studies were included in this review: eight randomized controlled trials11–18 and one prospective cohort study. 19 A PRISMA flowchart of the study selection process and literature search is shown in Figure 1. In total, 809 patients among all 9 studies were analyzed, with the year of publication ranging from 2009 to 2022. The patients’ basic characteristics, therapeutic outcomes, and adverse events are presented in Tables 1, 2, and 3, respectively.
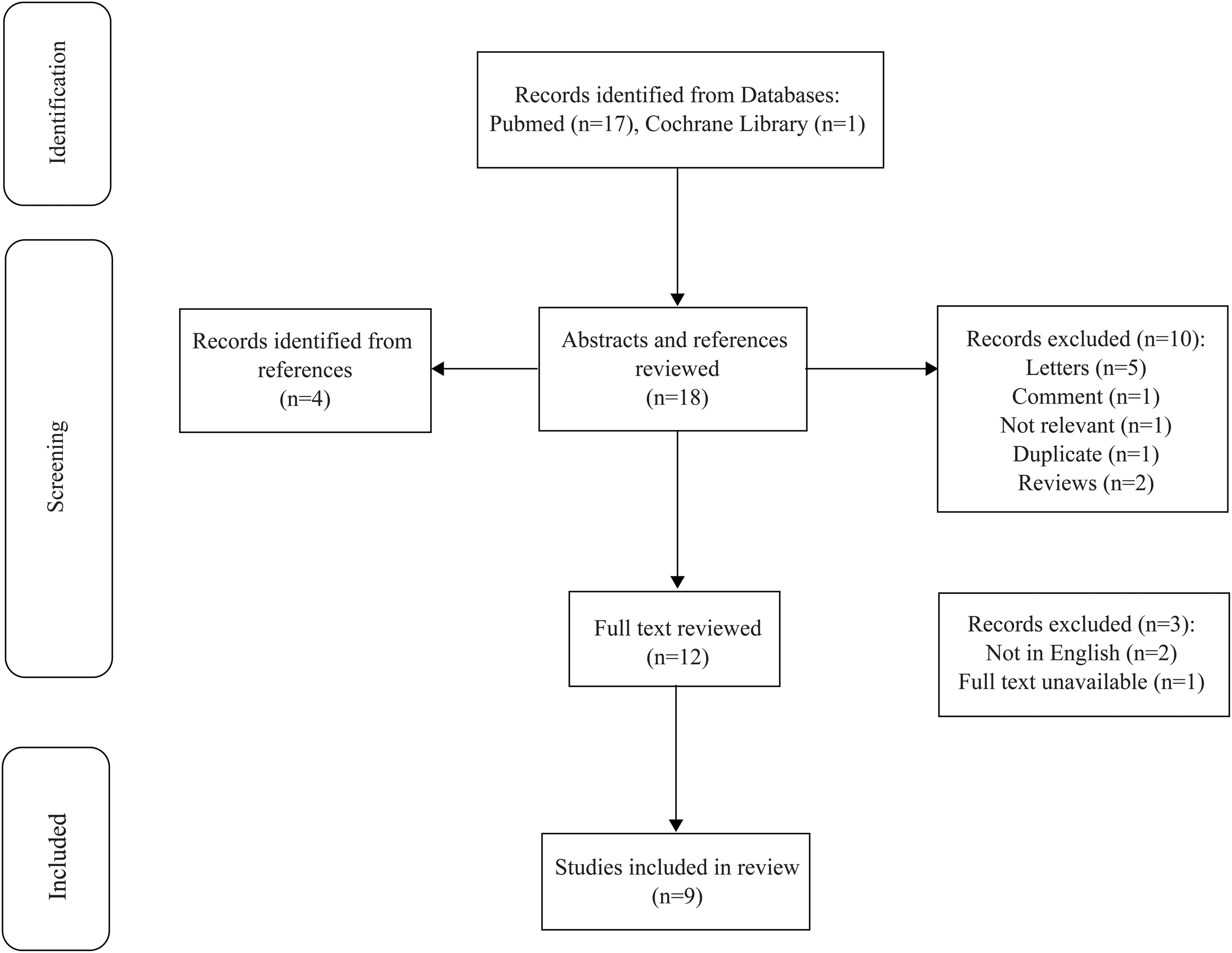
PRISMA diagram.
Characteristics of included studies.

PRP platelet-rich plasma, RCT randomized controlled trial, Max. maximum, NA not available, PRP-FG platelet-rich plasma and fibrin glue.
Main therapeutic outcomes.

PRP platelet-rich plasma, NA not available, a,bnot significant, cduration of pain, dopen surgery + PRP group, ecurettage + PRP group, fquantity of analgesics needed.
Adverse events.

PRP platelet-rich plasma, NA not available, a,b,c,enot significant, ddelayed healing.
For the preparation of PRP, autologous blood was used in eight studies (except one that was not clearly reported 17 ) and kits were used in two studies.13,17 Spyridakis et al. 11 and Gohar et al. 15 measured the wound volume and found that the accelerated healing process in the PRP group started on approximately postoperative day 5 and lasted almost until the time of healing, with PRP being applied on postoperative days 4 and 12. Mohammadi et al. 13 performed biopsies a few days before complete healing and found that wounds in patients treated with PRP contained collagen sediment (no collagen deposits were found in the control group), more CD34+ cells, and higher angiogenesis rates. In their study, two patients in the PRP group reported three episodes of recurrence, all of which ended in complete healing during postoperative weeks 6 and 8. 13 Sevinc et al. 19 performed a single-arm cohort study in 2020 comparing the outcomes of recurrent cases and primary cases. They found no difference in the duration of time required for healing, number of applications of PRP, or recurrence rate. Boztug et al. 16 established three groups: the open surgery group, open surgery + PRP group, and curettage + PRP group. The post hoc analysis results suggested that the curettage + PRP group had the shortest durations of time until healing and pain-free walking as well as the smallest cavity volume. The results also showed that the recovery time per unit of cavity volume was shortest in the open surgery + PRP group, and that in the open surgery group was shorter than that in the curettage + PRP group, but not significantly. Furthermore, they found significantly lower pain scores in the open surgery + PRP group and curettage + PRP group than in the open surgery group from visits 2 to 5, and the open surgery + PRP group showed significant improvements in postoperative symptoms. 16 Kurt et al. 17 combined laser pilonidoplasty with PRP for treatment of PD and found no significant difference in the operative time or length of hospital stay compared with the control group. Sevinc et al. 18 also conducted a 5-year randomized controlled trial with a sample size of 200. In their study, a patient’s wound healing was considered successful if the wound remained closed for at least 1 month with no complaints or formation of abscesses or new pits after one session of curettage combined with phenol or PRP application. When these criteria were not met, a subsequent treatment using the same approach was performed. The results showed healing rates of 53% (1 application), 89% (2 applications), 94% (3 applications), 98% (4 applications), and 100% (5 applications) in the control group. 18 In two studies that clearly reported the healing rate,18,19 the healing rate after PRP application was 98.32% (234/238) and 84.00% (84/100) in the control group. Moreover, a correlation was observed between the frequency of PRP application and the healing rate.
The total incidence of adverse events in traditional surgery and minimally invasive procedures is shown in Figure 2. Traditional surgery included open excision with or without primary closure, and minimally invasive procedures included excision, curettage, and laser pilonidoplasty. The results of the summary independent-samples t test showed that only the healing time in the control groups of the studies by Gohar et al. 15 and Boztug et al. 16 had no significant differences, whereas the healing time in the PRP group of the study by Boztug et al. 16 was significantly shorter (p = 0.04).

Incidence of adverse events.
The risk of bias in the eight randomized controlled trials are presented in Figures 3 and 4. Because of the specificity of surgical clinical trials, all studies had a high risk of performance bias. Most studies did not use an explicit approach to reduce the bias of selection, detection, attrition, reporting, and other aspects.

Risk-of-bias graph of included studies (randomized controlled trials).

Risk-of-bias summary of included studies (randomized controlled trials).
Discussion
PRP preparation
PRP usually contains platelet concentrations 2 to 3 times above the average levels. 20 It also contains many growth factors, cytokines, chemokines, and cell-adhesion molecules, which lead to the activation and synthesis of essential products involved in the healing process and tissue proliferation and regeneration.21,22 Furthermore, the platelet-derived proteins contained in PRP can lead to the formation of a fibrin scaffold that serves a dual purpose by acting as a matrix that promotes healing and facilitating the crucial role of platelets in coagulation.23,24 Therefore, the standardized PRP preparation process is very important and directly related to the efficacy of treatment.
Several convenient and safe PRP commercial systems are available on the market and can be applied in clinical practice. Two examples are the kits used in two of the studies in the present review.13,17 Such commercial systems show no increase in adverse events compared with self-produced PRP.
In almost all studies included in this review, autologous blood was used to make PRP. However, in clinical practice, we sometimes encounter patients who cannot receive an autologous blood supply and we must therefore consider using allogeneic blood to make PRP. In general, autologous PRP has been less thoroughly investigated in terms of the risk of immune reactions, and cross-contamination is a significant obstacle. Local application of autologous PRP may prevent the PRP from entering the circulatory system, thus minimizing exposure to most alloantigens; this may be beneficial in reducing immunogenicity. 25 Using blood from immediate family members is also a possible option. 26
The final yield of PRP is about one-fifth the blood volume used, making it necessary to pre-emptively estimate the consumption according to the treatment requirements. The blood consumption in a study by Mehrabi et al. 12 was five times the volume of the cavity, and the authors set a maximum limit of 250 mL. Although some other trials also accurately measured the volume of the cavity by filling it with 0.9% normal saline, these experiments only used the volume to assess the treatment effect. In the present review, the blood consumption per treatment session varied from 27 to 90 mL, showing no obvious correlation with the way the PRP was used (injection, filling, or a combination of both), and it was only affected by the cavity volume when used to fully fill the cavity. The composition and function of PRP sometimes varied greatly between storage conditions and storage forms,27,28 and the effect of these differences on wound healing remains to be investigated. 29 In the present review, PRP in patients with PD was generally applied soon after blood collection to avoid unknown risks.
Surgery and PRP application
Previous studies have shown that open surgery for treatment of PD is associated with a lower recurrence rate and longer healing time. 2 This was also reflected in the present review (Table 2, Table 3, and Figure 2). However, studies in recent years have shown that minimally invasive surgical methods (including minimally invasive excision, 19 curettage,16,18 laser pilonidoplasty, 17 and others) are associated with faster postoperative recovery and wound healing, a quicker return to normal daily activities, and higher patient satisfaction.30–34 In this review, the laser pilonidoplasty group was limited by its long healing time. Additionally, PRP in combination with minimally invasive surgery had a shorter healing time than PRP combined with general open surgery, which is consistent with the results of some previous studies that did not involve the use of PRP. 35 In the application of PRP to the surgical wound of wide excision with primary closure, the only positive treatment result seemed to be pain relief. 14 This treatment combination was less effective in promoting wound healing in PD than in other types of wounds or ulcers,36,37 which may have been due to the small sample size. Regardless of the surgical method and application method, the addition of PRP to other treatment methods can accelerate healing, reduce the severity of pain, or shorten the duration of pain.
The recovery time per unit of cavity volume was used to assess the healing process. The recovery time was shortest among patients who underwent open surgery combined with PRP, followed by patients who underwent open surgery without PRP and patients who underwent curettage combined with PRP. 16 In other words, among patients treated with PRP, open wounds healed faster than minimally invasive wounds over the same unit of time. The observed shorter healing time of minimally invasive wounds was mainly attributed to their smaller cavity size. Therefore, we believe that PRP is only one factor that accelerates the healing process of PD wounds and that surgery itself plays a decisive role. After the local application of PRP, β-fibroblast growth factor, vascular endothelial growth factor, platelet-derived growth factor BB, and insulin-like growth factor-1 increased to varying degrees over at least the next 4 days; among these, vascular endothelial growth factor showed the longest duration of increase. 38 These findings suggest that PRP activates biological pathways to release growth factors rather than simply delivering growth factors that are present in the concentrate. 39 This is why the effect of PRP in promoting wound healing can work quickly and last for a long time. The biopsy results also validated this at both the cell and tissue levels. 13 In addition, the speed of healing was faster in early stages and slower in later stages; thus, the difference in the mean wound volume between two consecutive time periods (dv/dt) can be calculated to represent this change more accurately. 11 To enhance the long-term effectiveness of PRP therapy and minimize the risks associated with collecting a large volume of blood at once, it may be beneficial to administer multiple applications over time.
The healing time in the PRP group in the study by Boztug et al. 16 was shorter than that in the study by Gohar et al. 15 Although this difference does not necessarily negate the possibility of increased PRP applications being a contributing factor, it raises the possibility that the injections themselves may have played a role in influencing the outcome.25,40 PRP is usually injected to a 13-mm depth in the wound area (0.1 mL/cm2) after cleaning the wound.13,15,17
Prognosis and adverse events
Minimally invasive surgery combined with PRP technologies can repeatedly performed under local anesthesia and in an outpatient setting, and the success rate of a single application can exceed 80%.18,19 If applied several times, the success rate can increase to >95%. Therefore, when comparing PRP with other one-time therapies, it is important to pay attention to the difference in effectiveness associated with the number of treatments. The other studies in this review did not explicitly report the final healing rates, and although they appear to have achieved very positive results, we believe that the healing rates rather than the adverse event rates should be considered first. Unfortunately, except for the shorter healing time and better cosmetic results of laser pilonidoplasty, PRP did not achieve positive results in terms of pain scores, the time until return to normal life or work, the operative time, or the length of hospital stay. 17 To date, only one study with a sample size of 50 patients has reported this technology, and more tests may be needed in the future to find more positive results. The short-term healing effect was worse in recurrent cases than in primary cases, but there was no significant difference in the overall treatment outcome between the two groups. 19 We believe that cases of recurrence and pilonidal abscesses should not be excluded from the scope of treatment. For cases involving abscess formation, prior treatment such as drainage and antibiotic use is required.
Because some studies lacked a control group16,19 and the duration of follow-up and the criteria for including adverse events varied among the studies, we believe that Figure 2 shows only a preliminary outcome with unavoidable bias. Overall, PRP use seems to reduce the incidence of adverse events, which is not completely inconsistent with and sometimes contradictory to the results of individual studies. We identified some beneficial methods to avoid PRP-associated adverse events from existing studies.
The operator should ensure the quality and adaptability of the PRP by verifying that the blood provider is healthy and that their physiological state is similar to that of the host. 25 PRP has an inhibitory effect on methicillin-resistant Staphylococcus aureus, Enterococcus faecalis, Pseudomonas aeruginosa, and other bacteria, and the use of thrombin as an activator can enhance the strength of the antibacterial effect.41–43 Blood providers should also avoid using drugs that affect the immune activity and blood components. This is because the antimicrobial protein content of PRP may be affected 44 and because PRP itself may provide favorable conditions for bacterial growth. 12 Although the clinical value of perioperative prophylactic antibiotic therapy in PD surgery is not clear,2,45 Mehrabi et al. 12 suggested a new approach: adding antibiotics to PRP to increase the antibacterial effect.
Boztug et al. 16 reported that four of their nine patients in the curettage + PRP group had abscesses in the second week of follow-up; therefore, they suspended the group. The authors considered that the abscesses had developed because PRP was trapped in the cavity and was not dispersed and drained sufficiently. 16 In a recent study, when Sevinc et al. 18 placed the PRP gel directly into the cavity after curettage, no aforementioned adverse events occurred after a mean follow-up of 43.1 months; however, the recurrence rate was 4%. Therefore, PRP gel with or without injection can be chosen in minimally invasive surgery or for wounds healing by primary intention to avoid overfilling the wound. During the perioperative period, except for routine measures such as weekly hair removal and use of analgesics (usually 500 mg paracetamol), all included studies recommended replacing the dressing at least 24 to 48 hours after PRP use, followed by regular wound cleaning and dressing replacement thereafter. Furthermore, the administration of drugs that compromise cell viability and platelet function, such as anesthetics, steroids, and nonsteroidal anti-inflammatory drugs, may have negative effects on PRP therapy.15,25
This review is limited by heterogeneity, which was primarily due to the lack of standardization of the application methods and evaluation indicators of PRP. This is also why we were unable to conduct a quantitative meta-analysis. Even when such analyses have been performed, 46 we maintain that a meta-analysis of these highly heterogeneous studies may not be appropriate. None of the studies included in this review reported economic costs, which affected our evaluation of the practical significance of promoting this therapy.
Conclusion
The decisive treatment for PD is surgery. As an adjuvant treatment measure, PRP can promote healing, reduce pain, shorten the duration of pain, accelerate the return to normal life, and reduce psychological stress. Preliminary results showed that PRP can also reduce the recurrence rate, wound infection rate, and incidence of other adverse events. Cases involving recurrence and pilonidal abscesses can be included in the treatment. Minimally invasive surgery combined with multiple applications of PRP have achieved more positive results. PRP is influenced by various factors such as the quality of preparation, surgical methods, application techniques, number of applications, and their combinations. These factors may sometimes contribute to a negative effect. The application of PRP in PD is still in the exploratory stage, and future studies with larger samples should be conducted to explore the optimal combination of PRP and various factors to promote standardization of this therapy.
Footnotes
Acknowledgements
The authors acknowledge Miss He, Dr. Zhang, and Dr. Mei for their general support during this study.
Author contributions
Conception: Yu Zhuang. Design: Yu Zhuang and Wen-zhe Feng. Acquisition of data: Yu Zhuang. Analysis and interpretation of data, drafting of the manuscript, critical revision of the manuscript, and final approval of the version to be published: Yu Zhuang and Wen-zhe Feng.
Data availability statement
All data used in this article are from publicly available studies.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
