Abstract
Objective
To obtain high-purity nasal epithelial cells (NEC) while avoiding the irritation experienced by patients during nasal biopsies.
Methods
This prospective, observational study enrolled patients undergoing surgical treatment for nasal septum deviation. After general anaesthesia, a novel nasal scraping spoon was used to collect epithelial cells from the mid-part of the inferior turbinate. The cells were evenly plated on six-well plates coated with rat tail collagen. The morphology and growth of the cells were observed at different time-points using an inverted phase-contrast microscope. Immunofluorescent staining of cytokeratin 18 was used to identify NEC. Ki67 staining was used to check cell viability.
Results
This study collected samples from 19 patients during a short procedure. No postoperative complications were observed. Cell samples ranging from 8.31 × 105 to 2.04 × 106 cells/sample were obtained. The culture model was suitable for primary NEC culture as demonstrated by the faster proliferation (5–7 days). There was no fungal or bacterial contamination. Immunofluorescent staining confirmed the presence and proliferative activity of NEC in the cultures.
Conclusion
A novel nasal scraping spoon provided an easy sampling method, avoided nasal injuries and psychological barriers to sampling and sufficient viable NEC to establish primary cultures.
Introduction
Airway epithelial cells can block harmful substances and play a crucial role in the first line of immunological defence. They include upper airway cells such as nasal epithelial cells (NEC) and lower airway cells such as bronchial epithelial cells. Human nasal epithelial cells have been proven to be a good surrogate model for studying nasal diseases. 1 A previous study measured the effect of different cytology brushes on the ciliary beat frequency (CBF) and reported that the choice of brush did not affect the diagnostic accuracy of CBF measurement. 2 Cultured NEC broadly increase our knowledge of the role of epithelial cells in some diseases, such as cystic fibrosis (CF),3,4 primary ciliary dyskinesia, 5 allergic rhinitis 6 and rhinosinusitis. 7
Nasal brushing has been used to obtain epithelial cells to establish primary cell cultures. However, it can be associated with discomfort, such as irritation, reflex lacrimation and/or sneezing. A previous study used a polyester swab or a cytology brush to take samples from the inferior turbinate of healthy adults and then performed cytology, RNA and DNA extraction. 8 Another study reported that curettes were used to scrape the inferior surface of the inferior turbinate for collecting and processing nasal epithelium to diagnose primary ciliary dyskinesia under a transmission electron microscope. 9 At present, nasal brushes are used mainly for the purpose of reducing patient discomfort, but the number of cells collected with them is small. 10 However, some patients still feel that there is a psychological barrier when undergoing nasal brushing. 10
This current study evaluated the use of a novel self-made nasal scraping spoon in patients requiring surgery with general anaesthesia in order to eliminate the discomfort experienced by individuals in obtaining high-purity primary NEC for cell culture and provide a basis for scientific research on nasal sinus diseases.
Patients and methods
Study design and population
This prospective, observational study enrolled consecutive patients in the Department of Otorhinolaryngology, Second Hospital of Dalian Medical University, Dalian, Liaoning Province, China between June 2022 and December 2022. All participants had a deviated nasal septum without sinus inflammation on computed tomography scanning. Patients with allergic rhinitis, chronic rhinitis, sinus mycosis and cancerous diseases were excluded. All participants underwent allergen screening and blood routine examination and had not taken oral or nasal topical glucocorticoids and systemic antimicrobials within 1 month of surgery. There were no other inclusion/exclusion criteria in this study.
This study was approved by the ethics committee at the Second Hospital of Dalian Medical University, Dalian, Liaoning Province, China (Ethics approval code: 2022-167, approval date: 29 December 2022) and it was conducted according to the ethical principles for medical research formulated in the World Medical Association Declaration of Helsinki. All study participants gave their written informed consent. All patient details have been de-identified. The reporting of this study conforms to STROBE guidelines. 11
Biopsies and cell culture
A specially made nasal scraping spoon was used to collect NEC from the turbinates in patients undergoing nasal septal correction (Figure 1). The special wide spoon tip can accommodate more cells. Its overall length is approximately 15.5 cm, which means that it is convenient to hold and can reach the surface of the middle turbinate. When scraping the cells, the operator only needs to gently scrape the surface mucosa of the middle turbinate 3–4 times to obtain a sufficient number of living cells. The operator does not need to apply too much force during the procedure and the aim is to not cause any bleeding.

A customized nasal scraping spoon made of metal materials with a total length of 15.5 cm. It can be used repeatedly after disinfection and sterilization.
After anaesthesia, the nasal mucosa was contracted with gauze containing 1% adrenaline and lignocaine, and the mucus was cleaned. The nasal mucosa of the middle turbinate was scraped with a nasal scraping spoon. At this time, the cells in the nasal scraping spoon contain primary NEC. The scraping spoon with NEC was immediately transferred to a sterile non-enzymatic Eppendorf tube containing BEpiCM medium (ScienCell™ Research Laboratories, Carlsbad, CA, USA) comprising 1% Bronchial Epithelial Cell Growth Supplement (ScienCell™ Research Laboratories) and 1% penicillin/streptomycin solution (P/S) (ScienCell™ Research Laboratories). The scraping spoon containing the mucosa was stirred in the medium. The cells were centrifuged at 1000 g for 5 min at 4 °C using an X1R centrifuge (Thermo Fisher Scientific, Rockford, IL, USA) and the supernatant was discarded. BEpiCM medium was first added to transfer the cells to a 15 ml centrifuge tube and then more was added to resuspend the cells according to the number of cells. The cells were inoculated in a six-well plate (Nest, Wuxi, China) coated with 3.33 mg/ml rat tail tendon collagen type I (Corning, New York, NY, USA). Cells were seeded at 37 °C in a humidified atmosphere of 5% CO2. BEpiCM medium and rat tail collagen only support the adherent growth of NEC. The medium was replaced every 2–3 days. The cells were photographed within 10 days of the start of cell culture. These cells were considered primary NEC. The cultured NEC can be passaged for four to five generations and these are known as the passaged cells. When passaged, the cells need to be stripped from the petri dish with 0.25% trypsin to obtain a single cell suspension and then transferred into a new petri dish for further culture. Generally, the cells were passaged every 5 to 7 days.
Cell counts were performed with cell counting chamber slides (Thermo Fisher Scientific) using an automated Countess II system (Thermo Fisher Scientific) according to the manufacturer's instructions. 2 Cell counts were reported as the range of minimum and maximum cell counts/sample.
Immunofluorescence
Cytokeratin 18 (CK-18) antibody staining by immunofluorescence was used to determine whether the cultured cells were NEC. CK-18 is the marker for secretory epithelial cells, such as NEC.12,13 Briefly, 80%–90% of the cells were removed from the incubator, the medium was discarded and 4% paraformaldehyde was added, and the cells were fixed at room temperature for 30 min. After washing, the cells were permeabilized with 0.3% Triton X-100 in 0.01 M phosphate-buffered saline (PBS; pH 7.2–7.4) for 15 min. The CK-18 antibody (1:50 dilution; Abclonal, Wuhan, China) was diluted with 0.01 M PBS (pH 7.2–7.4) containing 4% bovine serum albumin. The cells were washed again and incubated with the diluted CK-18 antibody at 4 °C overnight. After incubation with the primary antibody, the cells were washed three times with 0.01 M PBS (pH 7.2–7.4) for 10 min each time. Cy3-labeled goat anti-rabbit IgG(H + L) (Beyotime, Shanghai, China) was diluted with QuickBlock™ secondary antibody (Beyotime) for immunofluorescence in the ratio of 1: 500. The cells were incubated with the diluted secondary antibody at room temperature for 1 h in the dark. After incubation with the secondary antibody, the cells were washed three times with 0.01 M PBS (pH 7.2–7.4) for 10 min each time. 4′,6-diamidino-2-phenylindole was added to the dish, followed by incubation for 10 min at room temperature in the dark. After cleaning the plate and sealing it with an antifade mounting medium (Beyotime), the cells were observed under a microscope. In order to exclude the possibility of cell autofluorescence, a control group without primary antibody was used. To check cell viability, cells were stained with Ki67 (1 µg/ml dilution; Abcam®, Cambridge, MA, USA) using the same technique as described above. Cell viability is reported as the percentage of positive cells compared with total cells.
Results
This prospective, observational study collected samples from the turbinates of 19 patients (10 males and 9 females) aged 26–58 years. During the postoperative period, no complications were observed, such as postoperative bleeding and pain. The whole sampling process lasted approximately 5 min.
On day 1 of cell culture, the cells were attached to the plates. There was a mixed population of cells, including epithelial cells, fibroblasts and a few blood cells. Due to the particularity of the cell culture medium, the other types of cells, being non-adherent, remained suspended and were washed away on day 3 during routine cell washing and medium changes. The primary NEC appeared small, round and bright under an inverted phase-contrast microscope (Figure 2a). More than 60% of the cultures formed a complete monolayer between days 7 and 10, while the remaining cultures showed only small patches of epithelial cells. Photomicrographs of the cells showed a morphology typical of epithelial cells (Figures 2b & 2c). None of the cultures showed any fungal and/or bacterial contamination during cultivation. Cell samples ranging from 8.31 × 105 to 2.04 × 106 cells per sample were obtained.

Photomicrographs showing the morphology of primary nasal epithelial cells that were collected from the nasal cavity using the nasal scraping spoon as observed under an inverted phase-contrast microscope. The nasal epithelial cells appear small, round and bright on day 1 (scale bar 200 µm) (a). The nasal epithelial cells were polygonal, paving stone-like on day 7 (scale bar 200 µm; b) (scale bar 100 µm; c).
The immunofluorescence results of the primary and passaged NEC cells showed that all cells stained positive for CK-18, proving that the cultures consisted of 100% pure epithelial cells (Figure 3). The results showed that approximately 80% – 90% of the primary and passaged NEC nuclei were stained with Ki67 (Figure 4).
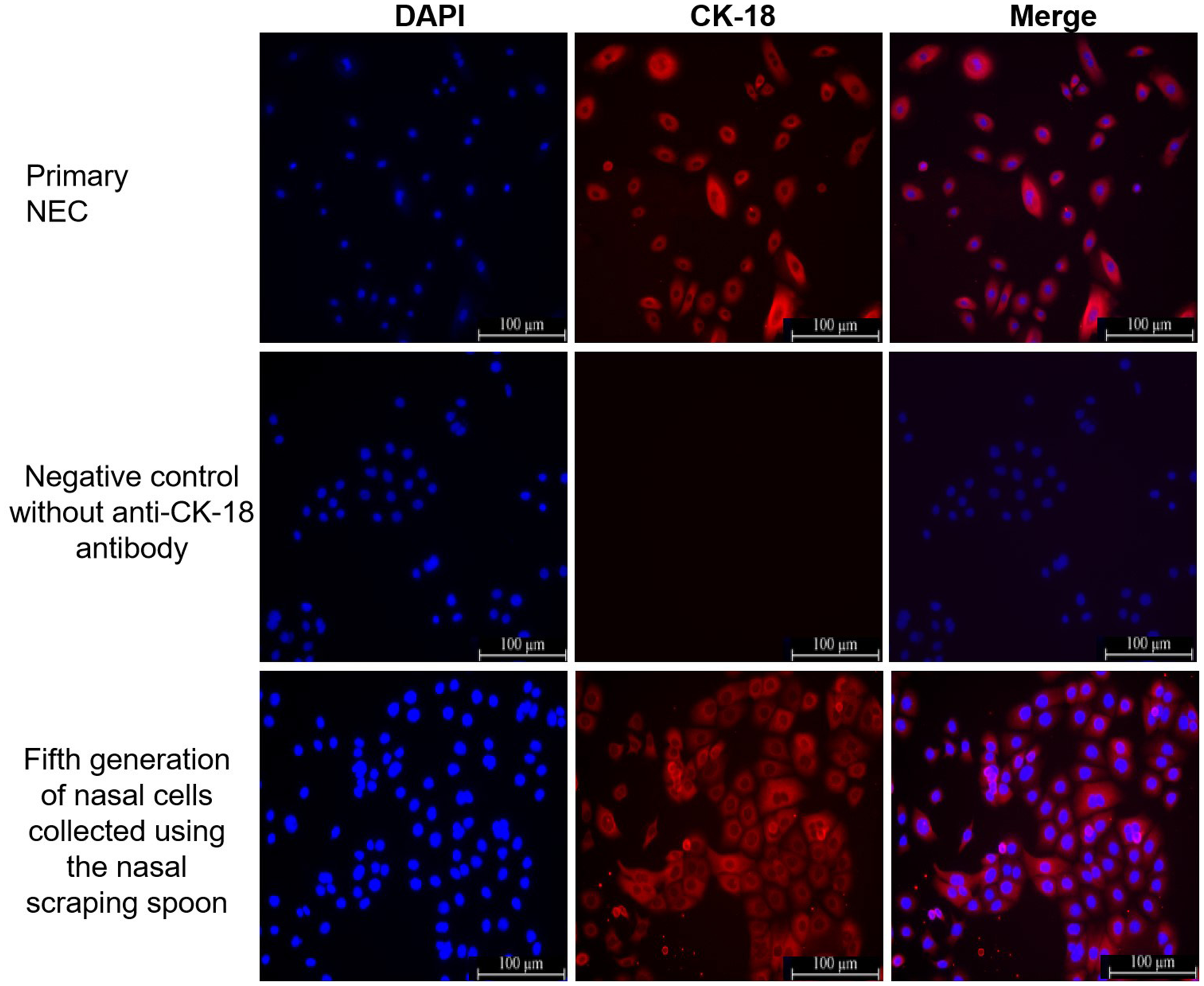
The localization of cytokeratin 18 (CK-18) staining was observed via immunofluorescence staining of primary nasal epithelial cells (NEC) and the fifth generation of cells that were collected from the nasal cavity using the nasal scraping spoon. Nuclei were stained using 4′,6-diamidino-2-phenylindole (DAPI; blue). Cell membranes were stained with CK-18 (red). Antibody-free negative controls were used to exclude the possibility of the cell autofluorescence. Scale bar 100 µm. The colour version of this figure is available at: http://imr.sagepub.com.

Immunofluorescence images of Ki67 staining of primary nasal epithelial cells (NEC) and the fifth generation of cells that were collected from the nasal cavity using the nasal scraping spoon to show cell viability. Viable cells are stained red. Nuclei were stained using 4′,6-diamidino-2-phenylindole (DAPI; blue). Scale bar 100 µm. The colour version of this figure is available at: http://imr.sagepub.com.
Discussion
This prospective, observational study collected biopsies from the mid-part of the inferior turbinate of patients requiring nasal endoscopic surgery under general anaesthesia. No complications or injuries were experienced by the patients during and after surgery. Cells showed a typical epithelial cell phenotype when examined and photographed under a light microscope between days 7 and 10. All cell cultures showed epithelial cell lineage by exhibiting positive staining for CK-18. Approximately 80%–90% of cells showed proliferative activity by staining with Ki67.
There is good evidence that NEC may be a suitable surrogate for the study of certain aspects of the function of bronchial airway epithelial cells (AEC), since bronchial AECs are difficult to obtain from children and healthy adults. 14 Bronchial epithelial cells are usually obtained by bronchoscopy, which is an invasive procedure. 15 For NEC, the use of a nasal brush under local anaesthesia yields a certain number of epithelial cells but also causes discomfort to patients. 10
Nasal brush biopsies are a relatively simple source of epithelial cells compared with the more aggressive bronchial brushing (bronchoscopic) method. However, the number of cells obtained through this method can be insufficient. This current study harvested cells from patients who required nasal endoscopic surgery under general anaesthesia without additional damage and psychological barriers. Previous studies have shown that the clinically used concentrations of adrenaline and lidocaine do not affect the CBF of epithelial cells collected using the nasal brush.16,17 The nasal scraping spoon used in this current study is metallic, inexpensive, sterilizable and reusable; and allows the collection of enough high-purity cells. The fifth generation of nasal cells collected using the nasal scraping spoon had an immunofluorescent appearance that was consistent with cultured primary NEC and all cells were positive for CK-18 staining (Figure 3).
Compared with immortalized cell lines, primary cell culture is more physiologically similar to in vivo cells, but there are also huge challenges, including limited sample accessibility, difficulty in cell separation and contamination. In the present study, none of the cultures showed fungal and/or bacterial contamination. A previous study showed a 100% success rate for viable cultures derived from healthy controls. 18 However, due to bacterial contamination, only 75% of the samples obtained from CF patients grew successfully. 18 Bronchial and nasal brush biopsies obtained from patients with chronic obstructive pulmonary disease (COPD) showed an 80–90% success rate. 19
There are some controversies on whether intranasal brushing can be used to culture primary cells associated with nasal sinus disease. 20 A previous study showed that NEC from patients with COPD cannot substitute for in vitro bronchial epithelial cells in airway inflammation studies. 19 Another study reported that despite differences in inflammatory mediator levels, nasal cells could be used as surrogates for bronchial cells in studies of lower airway inflammation, because they show a similar response to cytokine stimulation. 15 An examination of the feasibility of intranasal brushing for the culture of NEC in patients with allergic rhinitis reported that the collection of NEC using intranasal brushing may be an alternative method for epithelial cell culture in patients with AR as they maintain the unique allergic characteristics. 21 We are exploring new possibilities for increasing the growth rate of primary epithelial cells in nasal polyps and intend to demonstrate feasibility at the molecular level.
This current study had two limitations. First, as there is no conventional uniform standard to obtain NEC, this novel method of cell collection was only compared in the discussion section with the methods mentioned in the references. Secondly, the study did not involve more in-depth research, such as the examination of ciliary ultrastructure.
In conclusion, the specific advantage of this novel nasal scraping spoon is to make the sampling process smooth, obtain a sufficient number of living cells and minimize psychological and physical trauma. Thus, the nasal scraping spoon may be a suitable and reliable substitute tool. Furthermore, this current study has confirmed that the method is effective for recovering viable NEC to constitute primary cultures and suggest that this may help beginners master the skills of primary epithelial cell culture.
Footnotes
Acknowledgements
The authors would like to thank all participants involved in this research.
Author contributions
Hangjin Li analysed the data and wrote the manuscript. Junting Yin, Yiqin Wang and Hui Qu undertook the cell experiments and collected the data. Shen Yu assisted in the performance of the study. Jizhe Wang was the principal investigator and supervised the study. Hui Qu and Jizhe Wang helped revise the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
This research was supported by the National Natural Science Foundation of China (Grant No. 12172082).
