Abstract
Objectives
This study assessed the antifibrotic effects of canagliflozin, with or without irbesartan, on renal injury in Dahl salt-sensitive (SS) rats fed a high-salt (HS) diet.
Methods
After the preconditioning stage, Dahl SS rats (n = 47) were divided into five experimental groups as follows: low-salt (LS, n = 7), HS (n = 10), HS with canagliflozin (n = 10), HS with irbesartan (n = 10), and HS with canagliflozin and irbesartan (n = 10).
Results
The HS diet increased systolic blood pressure (SBP), renal fibrosis, fibrotic protein expression, and transforming growth factor-β1 (TGF-β1)/Smad2/3 pathway protein expression compared with the findings in the LS group. Irbesartan reduced SBP and slowed the loss of renal function. Canagliflozin significantly reduced body weight and renal fibrosis and suppressed the TGF-β1/Smad2/3 pathway. The combined therapy exerted better renoprotective effects on all outcome parameters.
Conclusions
These results indicate that canagliflozin and irbesartan exert different effects on renal injury in SS hypertensive rats, and the combined regimen could have stronger effects than either monotherapy.
Keywords
Introduction
Among the host of factors leading to essential hypertension, high dietary salt intake, which results in salt-sensitive hypertension (SSHT), is a key environmental factor. 1 Although restricted salt intake is beneficial, people continue to consume excessive salt in some areas, especially in North China. 2 Hypertension with chronic kidney disease (CKD) is caused by various factors, among which salt sensitivity and increased activity of the renin–angiotensin–aldosterone and sympathetic nervous systems might be more important. 3 Therefore, we must pay close attention to kidney damage in SSHT.
Individuals with SSHT have a higher plasma volume, greater total peripheral resistance, and increased blood pressure, mainly through the retention of water and sodium attributable to high-salt (HS) diet intake. 4 Renal fibrosis is a characteristic kidney pathogenesis of SSHT. 5 Although the mechanisms behind the proposed activation pathways of transforming growth factor-β (TGF-β) are not completely understood, TGF-β1 is widely regarded as a key regulator of fibrosis in the kidney and other tissues. 6 Among the TGF-β signaling pathways, TGF-β/Smad signaling plays an important role in renal fibrosis. 7 Smad2 and Smad3 are the two major receptor-activated factors that promote TGF-β1–mediated tissue fibrosis. 8 Dahl salt-sensitive (SS) rats develop SSHT and chronic ischemic nephropathy on an HS diet, which has been demonstrated to induce proteinuria, glomerulosclerosis, interstitial fibrosis, and eventually a progressive loss of renal function. 5
Sodium-glucose cotransporter 2 (SGLT2) inhibitors represent a new category of antidiabetic drugs 9 that inhibit glucose reabsorption in renal proximal tubules, leading to increased urinary glucose excretion and lower plasma glucose levels. 10 Multicenter clinical trials demonstrated that SGLT2 inhibitors can improve renal function and reduce the risk of major kidney events in patients with type 2 diabetes (T2D). 11 Moreover, SGLT2 inhibitors are promising as first-line therapies for nondiabetic CKD. 12 However, the effect of SGLT2 inhibitors on renal fibrosis in patients with SSHT has not been well studied. Because angiotensin II receptor blockers (ARBs) are the basic treatments for CKD, our study explored the effects of canagliflozin and irbesartan on renal injury in Dahl SS rats on an HS diet.
Materials and methods
Experimental protocol
All animal experiments in this research were performed in compliance with the Guide for the Care and Use of Animal Ethics Committee of Hebei Medical University and approved by the Ethics Committee of Hebei General Hospital (No. 2022178). Seven-week-old male Dahl SS rats (n = 47) were purchased from Beijing Vital River Laboratory Animal Technology Company (Beijing, China). The rats were housed in a temperature- (22 ± 2°C), light- (12-hour/12-hour light/dark cycle), and humidity-controlled (60% ± 10%) room. All rats had free access to standard chow and distilled water. All animals were acclimatized for 1 week and maintained on a low-salt (LS, 0.3% NaCl) diet at the animal facility of the Clinical Research Center. After pretreatment, all rats were randomly divided into the following five groups: LS group, (LS, n = 7), LS diet with vehicle treatment (5% hydroxyethylcellulose, 30 mg/kg/day); HS group (n = 10), HS diet (8% NaCl) with vehicle treatment; HS with canagliflozin (Xian Janssen Pharmaceutical Ltd., Xi'an, China) group (HS + CANA, n = 10), HS diet (8% NaCl) with canagliflozin treatment (30 mg/kg/day); HS with irbesartan (Sanofi (Hangzhou) Pharmaceutical Co., Ltd., Hangzhou, China) group (HS + IRB, n = 10), HS diet (8% NaCl) with irbesartan treatment (30 mg/kg/day); and HS with canagliflozin and irbesartan group (HS + CANA + IRB, n = 10), HS diet (8% NaCl) with canagliflozin and irbesartan treatment (both 30 mg/kg/day). To achieve better oral administration, all drugs were dissolved in 5% hydroxyethylcellulose (10 mg/mL, MedChemExpress, Monmouth Junction, NJ, USA). Food and water intake were measured daily. Body weight (BW) and systolic blood pressure (SBP) were measured weekly. Random blood glucose (RBG) and fasting blood glucose (FBG) levels were measured in the middle and at the end of the experiment. After fasting for approximately 4 hours, all rats were anesthetized with sodium pentobarbital (50 mg/kg body weight, i.p.). Whole blood was obtained by the abdominal aorta, and kidneys were isolated.
Plasma and urine collection and analysis
Before the end of the experiment, five rats randomly selected from each group were housed individually in metabolic cages to collect 24-hour urine under food and water consumption. Blood samples were collected from the abdominal aorta, and serum was separated by centrifugation at 1006.2 × g for 10 minutes at 4°C and stored at −80°C. Urinary and plasma biochemical indices were determined using an automatic biochemical analyzer (DS-261, SINNOWA Medical Science and Technology Co., Ltd., China).
Blood pressure measurement
SBP was measured by tail-cuff plethysmography (BP-2000; Visitech Systems, Inc., Apex, NC, USA) according to the manufacturer’s instructions. Measurements were obtained for each conscious rat every 2 weeks until sacrifice. Before each measurement, the rats were prewarmed at 36°C for 15 to 20 minutes to obtain reliable results. The average of six pressure readings was calculated for each measurement. SBP was always measured at the same time of day (10 am to 12 am) by the same experimenter.
Histologic evaluation
One-half of the left kidney from each rat was fixed in 4% paraformaldehyde for 24 hours. After dehydration, the tissues were embedded in paraffin, and 5-µm-thick sections were stained with hematoxylin and eosin (H&E) for morphological analysis and with Masson staining for the evaluation of fibrosis. All sections were analyzed by light microscopy (Nikon, Tokyo, Japan). The renal fibrosis area fractions were quantified using ImageJ 8.0 (US National Institutes of Health, Bethesda, MD, USA) by calculating the mean optical density (IODsum/Areasum) of the fibrotic areas with positive staining. Ten randomly and nonoverlapping fields (×200 magnification) were chosen for each section.
Immunofluorescence (IF) staining was performed to detect the expression of renal TGF-β1 in rats. After being sealed, the kidney tissue sections were incubated with anti-TGF-β1 antibody (sc-130348; Santa Cruz Biotechnology, Santa Cruz, CA, USA; 1:500) in 1× phosphate-buffered saline (PBS) overnight at 4°C. The slides were then blocked with 10% goat nonimmune serum at 37°C for 1 hour. After returning to room temperature, the samples were washed with PBS and incubated with fluorescein-labeled secondary antibody (Zen BioScience, Chengdu, China) for 1 hour at 37°C. Finally, the sections were counterstained with 4′,6-diamidino-2-phenylindole (1 mg/mL) for nuclear staining and visualized using a fluorescence microscope (Zeiss, Munich, Germany). The fluorescence intensity of the positive area was measured using ImageJ 8.0. For each experimental group, five random fields were selected to calculate the mean fluorescence activity.
Western blotting (WB)
The protein concentrations of renal samples were determined using a bicinchoninic acid protein assay kit (Thermo Fisher Scientific, Waltham, MA, USA). Protein samples (50 µg) were separated by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred onto a polyvinylidene difluoride membrane. The membranes were blocked with 5% skimmed milk for 2 hours at room temperature and then incubated with primary antibodies overnight at 4°C as follows: anti-collagen I (ab270993; Abcam, Cambridge, UK; 1:1000), anti-α-smooth muscle actin (α-SMA; ab7817; Abcam; 1:3000), TGF-β1 (ab179695; Abcam; 1:1000), anti-Smad2/3 (ab202445; Abcam; 1:1000), anti-pSmad2/3 (ab254407; Abcam; 1:1000), anti-Smad7 (sc-265846; Santa Cruz Biotechnology; 1:1000), and anti-glyceraldehyde-3-phosphate dehydrogenase (GAPDH; ab181603; Abcam; 1:10,000 dilution). The membranes were subsequently washed and then incubated with an appropriate horseradish peroxidase (HRP)-conjugated secondary antibody (ab6721; Abcam) diluted 1:5000 for 1 hour at room temperature. After exposure to Hyperfilm enhanced chemiluminescence (Thermo Fisher Scientific), the antigen–antibody complexes were detected using Odyssey XF (LI-COR, Lincoln, NE, USA). The signal intensities of target bands were analyzed using ImageJ 8.0. The optical density of the corresponding internal reference GAPDH was used for correction.
Statistical analysis
Statistical analysis was performed using IBM SPSS 23.0 software (IBM Corp., Armonk, NY, USA). All data are expressed as the mean ± standard error of the mean. The statistical significance of differences between the groups was determined using one-way or repeated-measures analysis of variance combined with the Newman–Keuls post hoc test. For all tests, P < 0.05 was considered statistically significant.
Results
Changes in physiological data
We first confirmed the differences in BW and other physiological parameters among the five groups during the 12-week experiment (Figure 1). After treatment for 4 weeks, BW was lower in the HS group than in the LS group and lower in the HS + CANA and HS + CANA + IRB groups than in the HS + IRB group. After treatment for 4 weeks, mean food intake (MFI) was higher in the HS + CANA and HS + CANA + IRB groups than in the HS + IRB group. After treatment for 6 weeks, mean water intake (MWI) was higher in the HS group than in the LS group and higher in the HS + CANA and HS + CANA + IRB groups than in the HS + IRB group. After treatment for 5 weeks, SBP was lower in the LS, HS + IRB, and HS + IRB + CANA groups than in the HS group. At 13 weeks of age, FBG levels were lower in the HS + CANA and HS + CANA + IRB groups than in the LS and HS groups (all P < 0.05; Figure 2a). No significant differences in RBG levels were found among the groups (Figure 2b).

Time courses of body weight, mean food and water intake, and SBP in rats in the five experimental groups. (a–d) Changes in body weight, mean food intake, mean water intake, and SBP, respectively. All data are presented as the mean ± standard error of the mean (n = 7, 10, 10, 10, and 10 rats in the LS, HS, HS + CANA, HS + IRB, and HS + CANA + IRB groups, respectively). The red horizontal line represents the time points at which the data were statistically different among the groups.*P < 0.05 vs. LS, **P < 0.05 vs. HS, &P < 0.05 vs. HS + CANA, &&P < 0.05 vs. HS + IRB. SBP, systolic blood pressure; LS, low-salt; HS, high-salt; CANA, canagliflozin; IRB, irbesartan.

Fasting blood glucose and random blood glucose levels in rats in the five experimental groups at 13 weeks of age. *P < 0.05 vs. LS, **P < 0.05 vs. HS. LS, low-salt; HS, high-salt; CANA, canagliflozin; IRB, irbesartan.
Physiological and biochemical data at the end of the experiment
At the end of the experiment, we examined the effects of different treatments on physiological and biochemical data among the five groups (Table 1). Compared with the LS group, BW was lower in the HS, HS + CANA, and HS + CANA + IRB groups (all P < 0.05). Compared with the HS + IRB group, the HS and HS +CANA groups had lower BW (both P < 0.05). BW was lower in the HS + CANA + IRB group than in the HS + IRB group (P < 0.05). FBG levels were higher in the HS +CANA and HS + CANA + IRB groups than in the LS and HS groups (all P < 0.05). SBP was higher in the HS group than in the LS, HS+IRB, and HS + CANA + IRB groups (all P < 0.05). SBP was lower in the HS + CANA + IRB group than in the HS + CANA group (P < 0.05). Compared with the LS group, MFI was lower in the HS and HS + CANA groups (both P < 0.05). MFI was higher in the HS + CANA + IRB group than in the HS, HS + CANA, and HS + IRB groups (all P < 0.05). Compared with the LS group, the other four groups had higher MWI (all P < 0.05). Heart rate (HR) was lower in the HS + CANA + IRB group than in the HS group (P < 0.05). Compared with the LS and HS groups, urine volume was significantly higher in the HS+CANA group (P < 0.05), whereas urine volume was decreased by irbesartan in the HS + CANA + IRB group (P < 0.05).
Physiological and biochemical parameters of the rats at the end of the 12-week experiment.
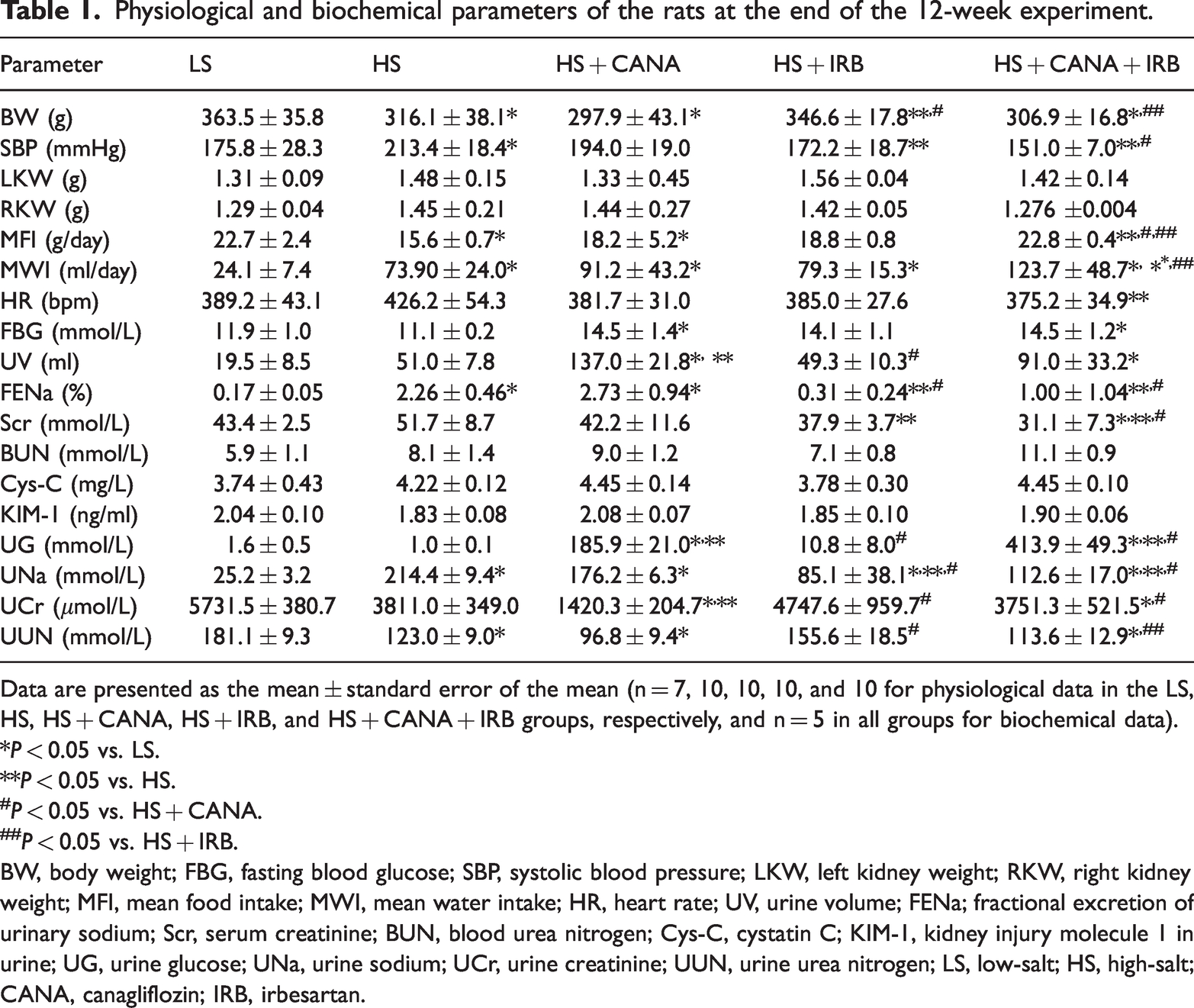
Data are presented as the mean ± standard error of the mean (n = 7, 10, 10, 10, and 10 for physiological data in the LS, HS, HS + CANA, HS + IRB, and HS + CANA + IRB groups, respectively, and n = 5 in all groups for biochemical data).
*P < 0.05 vs. LS.
**P < 0.05 vs. HS.
P < 0.05 vs. HS + CANA.
P < 0.05 vs. HS + IRB.
BW, body weight; FBG, fasting blood glucose; SBP, systolic blood pressure; LKW, left kidney weight; RKW, right kidney weight; MFI, mean food intake; MWI, mean water intake; HR, heart rate; UV, urine volume; FENa; fractional excretion of urinary sodium; Scr, serum creatinine; BUN, blood urea nitrogen; Cys-C, cystatin C; KIM-1, kidney injury molecule 1 in urine; UG, urine glucose; UNa, urine sodium; UCr, urine creatinine; UUN, urine urea nitrogen; LS, low-salt; HS, high-salt; CANA, canagliflozin; IRB, irbesartan.
To assess markers of renal injury in the rats, we next measured several important indices of kidney function (Table 1). Compared with the LS group, 24-hour urinary total protein (24-hour UTP) was significantly increased in the HS group (P < 0.05; Figure 3). After treatment, 24-hour UTP was significantly lower in the HS+IRB, HS+CANA, and HS+CANA+IRB groups (all P < 0.05; Figure 3). There were significant decreases in serum creatinine (Scr) levels and the fractional excretion of urinary sodium (FENa) in the HS + IRB and HS + CANA +I RB groups (all P < 0.05), whereas the decreases in the levels of these indices in the HS + CANA were not statistically significant. There were no significant differences in blood urea nitrogen (BUN), blood cystatin C (Cys-C), or urinary kidney injury molecule 1 (KIM-1) levels among the five groups.

Twenty-four-hour UTP in the different groups. **P < 0.05 vs. HS
Renal histological changes
We then performed histochemical staining, including H&E, Masson, and TGF-β1 staining, to evaluate the pathological changes in the kidneys of rats in the five groups. H&E staining demonstrated that compared with the LS group, morphological abnormalities of glomeruli, dilation of tubules, and epithelial cell edema were observed in the HS group, and these changes were markedly attenuated in the HS + CANA, HS + IRB, and HS + CANA + IRB groups (Figure 4a). The improvement of morphologic lesions was more obvious in the HS + CANA + IRB group. Masson staining (Figure 4b) and IF staining (Figure 4c) were analyzed by ImageJ 8.0. As illustrated by Masson staining (Figure 4d), interstitial fibrosis was more evident in the HS group than in the LS group. Meanwhile, collagen deposition was markedly alleviated in the HS + CANA, HS + IRB and HS + CANA + IRB groups. The combined treatment had a more beneficial effect. We used IF staining to analyze the expression of TGF-β1 in the kidneys of rats (Figure 4c). The results revealed that the expression changes in TGF-β1 were similar to the changes in collagen deposition in rat kidney tissue in the five groups (Figure 4e). Taken together, these results indicate that canagliflozin and/or irbesartan can improve renal fibrosis caused by an HS diet in Dahl SS rats, possibly through the TGF-β1/Smad2/3 pathway.

Renal histological damage in the different groups. (a) H&E staining (original magnification ×200). (b) Masson staining (original magnification ×200). (c) Representative IF images presenting the induction of TGF-β1 protein expression in the renal cortex (original magnification ×200). Slides were counterstained with 4′,6-diamidino-2-phenylindole for nuclear visualization. (d) The percent Masson-positive area (blue) relative to the entire area of Masson-stained specimens in the different groups (one-way analysis of variance, *P < 0.05 vs. LS, **P < 0.05 vs. HS, &P < 0.05 vs. HS + CANA, &&P < 0.05 vs. HS + IRB). (e) The percent TGF-β1–positive area (red) relative to the entire area of IF-stained specimens in the different groups (one-way analysis, *P < 0.05 vs. LS, **P < 0.05 vs. HS, &P < 0.05 vs. HS + CANA, &&P < 0.05 vs. HS + IRB). The values are expressed as the mean ± standard error of the mean.
Expression of fibrotic proteins and TGF-β1/Smad2/3 pathway proteins
To further confirm the effects of canagliflozin and irbesartan on renal fibrosis and the TGF-β1/Smad2/3 pathway, WB was performed. Renal fibrosis is characterized by the production of collagen and the activation of α-SMA. Subsequently, the expression of collagen I and α-SMA was determined by WB. The results indicated that the HS diet significantly increased the protein expression of collagen I (P < 0.05; Figure 5a–b) and α-SMA (P < 0.05; Figure 5a, c) in the kidneys of Dahl SS rats, whereas these proteins were downregulated in the HS + CANA and HS +CANA + IRB group (both P < 0.05). However, these fibrotic proteins were not significantly downregulated in the HS + IRB group. Compared with the LS group, TGF-β1 (P < 0.05; Figure 5d), Smad2/3 (P < 0.05; Figure 5e), and p-Smad2/3 expression (P < 0.05; Figure 5f) was significantly higher in the HS group, whereas these changes were reversed by canagliflozin and irbesartan (all P < 0.05). Similarly, additive effects were observed in the combined treatment group (all P < 0.05). In addition, compared with the LS group, the expression of Smad7 was significantly decreased in the HS group (P < 0.05; Figure 5g), whereas Smad7 was dramatically upregulated in the HS + CANA, HS + IRB, and HS + CANA + IRB groups (all P < 0.05). Taken together, these findings suggest that canagliflozin better alleviates renal fibrosis caused by an HS diet in Dahl SS rats than irbesartan, possibly through inhibiting the TGF-β1/Smad2/3 pathway.

Changes in renal fibrotic factors and TGF-β/Smad pathway proteins in the five groups. (a) The protein expression of fibronectin, collagen I, α-SMA, TGF-β1, Smad2/3, p-Smad2/3, Smad7, and GAPDH in Continued.the five groups were measured by WB. The relative expression of collagen I (b), α-SMA (c), TGF-β1 (d), Smad2/3 (e), p-Smad2/3 (f), and Smad7 (g) normalized to GAPDH expression was quantified by densitometry. Data are presented as the mean ± standard error of the mean of three animals in each group (one-way analysis of variance, followed by the Newman–Keuls post hoc test). *P < 0.05 vs. LS; **P < 0.05 vs. HS; &P < 0.05 vs. HS + CANA; &&P < 0.05 vs. HS + IRB. α-SMA, α-smooth muscle actin; TGF-β1, transforming growth factor-β1; GAPDH, glyceraldehyde-3-phosphate dehydrogenase; LS, low-salt; HS, high-salt; CANA, canagliflozin; IRB, irbesartan.
Discussion
The beneficial renal effects of SGLT2 inhibitors have been observed in patients with nondiabetic nephropathy. 13 However, distinct from clinical trials, it is surprising that the results of preclinical studies in rodents with nondiabetic renal injury have thus far been rather inconsistent, which is partially attributable to the different SGLT2 inhibitors used. ARBs are widely used to treat hypertension and hypertension-related heart and kidney damage because of their ability to alleviate tissue fibrosis 14 and improve endothelial function. 15 Preclinical findings supported a role of HS intake in increased renal TGF-β1 production, which possibly resulted from an increased plasma volume and cardiac output in response to an HS diet. 16 The results of this study demonstrated that SGLT2 inhibitors and ARBs have nephroprotective effects in models of SSHT, and greater benefits could be obtained by using the treatments in combination.
In this study, canagliflozin and irbesartan, both alone and in combination, induced various effects on BW, MFI, MWI, SBP, and HR in Dahl SS rats fed an HS diet. The HS diet reduced BW in Dahl SS rats, and canagliflozin aggravated the weight loss caused by HS diet consumption. Conversely, irbesartan had no such effects. The reduction in BW induced by SGLT2 inhibitors was consistent with the findings of a previous clinical study, 17 and the effect appeared to be related to SGLT2 inhibitor-induced reductions in glucose levels, thereby enhancing fat utilization. 18 Consistent with most previous studies, 19 canagliflozin significantly increased MFI in the present study, whereas irbesartan did not significantly affect MFI. Our data, together with previously published findings, 20 clearly illustrated that an HS diet significantly increases MWI in Dahl SS rats, and canagliflozin can further improve MWI in such animals. However, the lack of effects of irbesartan on the HS-induced increase in water intake indicated that the change in water intake is independent of AT1-receptor stimulation. 21 The urine volume of Dahl SS rats fed an HS diet was significantly increased by canagliflozin, whereas irbesartan reversed the increase in urine output. These results might be explained by the diuretic action of canagliflozin being enhanced by angiotensin II. 20
However, the effects of SLGT2 inhibitors on HS-induced hypertension in rats remain controversial.22,23 In the present study, our data revealed a nonsignificant decrease in SBP following canagliflozin treatment despite its extremely pronounced diuresis activity. Irbesartan significantly decreased SBP in HS diet-fed Dahl SS rats without increasing urine volume. The combination of canagliflozin and irbesartan more strongly reduced SBP than either drug alone. The nonsignificant decrease in SBP induced by canagliflozin might have partly resulted from the compensatory activation of sympathetic tone and renin release by HS diet consumption and natriuresis. 23 Furthermore, although the noninvasive tail-cuff methodology is well accepted for blood pressure monitoring in rats, this method also has some uncertainties because of the need for habituation to restrain and other influences on tail circulation. 24
Contrary to previous studies, 25 our data revealed that canagliflozin treatment significantly increased blood glucose levels in Dahl SS rats fed an HS diet for 12 weeks, which might be explained by the reduction in blood volume through natriuresis. The development of kidney dysfunction in HS diet-fed Dahl SS rats, including increased albumin, Scr, and FENa levels, was prevented by irbesartan and the combined treatment but not by canagliflozin alone. The effects of SGLT2 inhibitors on renal function in our experimental cohorts were consistent with the results of previous clinical 26 and animal 27 studies. Similar to an experiment in Dahl SS diabetic rats, 27 maximal renoprotection was achieved when canagliflozin was combined with irbesartan, the underlying mechanism of which warrants further consideration. However, inconsistent with previous studies in nondiabetic rats, 28 increases in the levels of BUN and other renal injury markers (Cys-C and KIM-1) in the present study were not prevented in the different treatment groups.
There have been few reports on the renoprotective effects of SGLT2 inhibitors in SSHT, and the molecular mechanism of their antifibrotic effects remains unknown. In our experimental conditions, renal fibrosis as determined by Masson staining was suppressed by irbesartan alone or in combination with canagliflozin, in line with the improved functional and histological parameters. Moreover, canagliflozin administration prevented the development of renal fibrosis without affecting blood pressure or renal function. 23 Indeed, maximal renoprotection from morphologic changes and renal fibrosis was achieved by the combined therapy, consistent with previous findings in diabetic Dahl SS rats. 29
Of further interest, we explored the effects of canagliflozin and irbesartan on TGF-β1/Smad2/3 signaling, which plays a key role in the development of renal fibrosis. In the present study, kidney TGF-β1 expression, as determined by IF staining and WB, was higher in the HS group than in the LS group, and this upregulation was accompanied by increased expression of fibronectin, collagen I, α-SMA, Smad2/3, and p-Smad2/3. However, both canagliflozin and irbesartan reduced the expression of TGF-β1 and other profibrotic factors with a positive correlation with renal fibrosis. Furthermore, we found that the combined therapy had an additive effect on the suppression of profibrotic factors and TGF-β1/Smad2/3 signaling, which was more susceptible to canagliflozin than irbesartan in the kidneys. These results suggest that canagliflozin directly decreased renal fibrosis and TGF-β1/Smad2/3 activation in SS hypertensive kidneys partially independent of its antihypertensive effects. Taken together, we speculate that SGLT2 inhibitors exert a better antifibrotic effect on renal fibrosis in SSHT, and this effect might be related to the suppression of TGF-β1/Smad2/3 signaling.
To our knowledge, this is the first study to compare the renoprotective effects of canagliflozin and irbesartan, both alone and in combination, in experimental SSHT. These findings provide novel data supporting that these two medications exert different effects on physiological indices, kidney function, renal fibrosis, and TGF-β1/Smad2/3 signaling. The data indicated that canagliflozin exerts a stronger antifibrotic effect, possibly by inhibiting the TGF-β1/Smad2/3 pathway. However, in almost all aspects of renoprotection, the combined therapy was consistently superior to either monotherapy.
Limitations of the study
This study had several limitations. First, this was an in vivo study, and the results were not verified in vitro. Second, the mechanism underlying the inhibitory effect on hypertensive renal injury was not fully elucidated. Nevertheless, our research can be considered a starting point for further studies investigating the difference in renoprotection between SGLT2 inhibitors and ARBs in SSHT.
Conclusion
The current results indicate that canagliflozin and irbesartan exhibit different renoprotective effects in SSHT models. Irbesartan displayed a better antihypertensive effect, whereas the antifibrotic effect of canagliflozin was more remarkable, possibly involving inhibition of the TGF-β1/Smad2/3 pathway. Consistently, additional molecular and histological benefits were offered when canagliflozin and irbesartan were co-administered, but the nephroprotective cellular mechanisms of the combined therapy remain to be further explored. Taken together, our study suggests that selective SGLT2 inhibitors can be used in combination with ARBs to further prevent the progression of nondiabetic nephropathy, especially in SSHT.
Footnotes
Author contributions
Conception and design: Jianlong Zhai, Zhongli Wang, and Tingting Zhang
Administrative support: Yifang Guo
Provision of study materials or patients: Lili He, Sai Ma, Qingjuan Zuo, and Guorui Zhang
Collection and assembly of data: Jianlong Zhai, Zhongli Wang, Tingting Zhang, and Xinyu Wang
Data analysis and interpretation: Jianlong Zhai, Zhongli Wang, Tingting Zhang, and Yifang Guo
Manuscript writing: All authors
Final approval of manuscript: All authors
Declaration of conflicting interests
The authors have no relevant financial or non-financial interests to disclose.
Data availability statement
The original data presented in the study are included in the article, and further inquiries can be directed to the corresponding author.
Funding
This work was supported by grants from the 2019 Hebei Science and Technology Project (No. 19277787D) and the 2019 Hebei Innovation Capability Promotion Project (No. 199776249D).
