Abstract
Objective
Combination fluoropyrimidine-based chemotherapy is the standard first-line treatment for metastatic colorectal cancer (CRC). We performed a propensity score (PS)-based analysis to report our real-world experience with long-term follow-up of this regimen for metastatic CRC.
Methods
In this retrospective study, 170 patients with newly diagnosed metastatic CRC treated with first-line combination chemotherapy between January 2003 and March 2021 were identified. Cox proportional hazards regression analysis and PS-based approaches with the logistic regression model were adopted, and the results were compared.
Results
The hazard ratio for overall survival (OS) in the oxaliplatin- and irinotecan-based groups was 0.79 (95% confidence interval = 0.56–1.11), and the median OS times in these groups were 16.8 and 13.0 months, respectively. The median time to progression (TTP) for these regimens were 9.0 and 8.9 months, respectively. The objective response rates for the oxaliplatin- and irinotecan-based groups were 42.7% and 34.6%, respectively. OS and TTP did not differ between these regimens in all PS matching models.
Conclusions
First-line treatment using fluoropyrimidine-based chemotherapy regimens in combination with oxaliplatin or irinotecan in patients with metastatic CRC provided comparable efficacy and tolerable toxicity profiles in a real-world setting with long-term follow-up.
Introduction
Colorectal cancer (CRC) is the third most commonly diagnosed cancer globally and the second-leading cause of cancer-related mortality. 1 In Thailand, CRC is currently the third most prevalent cancer among men, and it ranks fourth among women. 2 Although the incidence of CRC has been gradually decreasing in many Western countries over the past decade, 3 low- and middle-income countries, including Thailand, have experienced an increasing incidence, primarily because of effective screening programs. It is important to note that screening recommendations can vary depending on specific conditions, individual risk factors, and the guidelines followed in a particular country or region. For instance, individuals with Lynch syndrome, characterized by deficient DNA mismatch repair (MMR) or microsatellite instability-high, 4 typically start colon cancer screening at a younger age than the general population. In 2018, Thailand reported 17,534 cases of newly diagnosed CRC, accounting for 10.3% of all new cancer cases. 5 Furthermore, CRC is the only cancer with an increasing incidence in Thailand over the past two decades. 6 A study reported that 41.8% of patients with CRC presented with metastatic disease at diagnosis. 7 However, another study found that almost half of the patients who initially presented with resectable disease experienced disease recurrence after surgical resection or developed metastatic disease thereafter, typically in the liver or lungs. 8
Patients with unresectable metastatic CRC who receive supportive care alone have been reported to have poor prognoses, including median overall survival (OS) of 5 months. 9 Palliative chemotherapy improves OS and quality of life compared with the best supportive care alone. Combination therapy with leucovorin (LV), 5-fluorouracil (5-FU), and oxaliplatin (FOLFOX regimen) and LV, 5-FU, and irinotecan (FOLFIRI regimen) has become the standard first-line treatment strategy, and these regimens result in median OS of approximately 2 years. 10 Capecitabine, an oral chemotherapy agent metabolized to 5-FU, has displayed benefits as a single agent 11 and in combination with oxaliplatin (CAPOX) 12 or irinotecan (CAPIRI). 13 Adding biological agents, including anti-vascular endothelial growth factor (receptor) (VEGF[R]) 14 or anti-epidermal growth factor receptor (EGFR) agents, 15 enhanced the effect of cytotoxic therapy. However, systemic chemotherapy remains the mainstay of treatment for patients with CRC, especially in Thailand, where access to additional biological agents remains limited.
In clinical trials of first-line chemotherapy, FOLFOX and FOLFIRI were demonstrated to provide comparable OS but different toxicity profiles. 16 , 17 However, most studies were conducted in Western patients, whose results might differ from those in Asian populations. This study provided the real-world experience of first-line combination chemotherapy in patients with metastatic CRC from the Medical Oncology Service of a tertiary-care hospital in Southern Thailand.
Materials and methods
Study participants and procedure
This was a retrospective, single-center study. The reporting of this study conforms to the STROBE guidelines. 18 Patients who received first-line palliative chemotherapy for CRC at the Medical Oncology Service of Prince of Songkla University Hospital (Songkhla, Thailand) were eligible for the study. The inclusion criteria were as follows: 1) histologically confirmed diagnosis of adenocarcinoma arising from the colon or rectum; 2) advanced (metastatic and/or recurrent) disease; 3) no prior chemotherapy excluding adjuvant chemotherapy with a disease-free interval ≥6 months after the completion of chemotherapy; 4) availability of clinical data at the beginning of therapy and follow-up; 5) age ≥18 years; 6) Eastern Cooperative Oncology Group (ECOG) performance status ≤2; 7) and completion of at least one cycle of FOLFOX, FOLFIRI, CAPOX, or CAPIRI with or without biological agents. Patient data were collected from the Electronic Medical Record (EMR) of the Hospital Information Systems (HIS) of Prince of Songkla University Hospital.
This study was reviewed and approved by the Ethics Committee at the Research Center of Prince of Songkla University Hospital (approval number REC.64244144). The requirement for written informed consent was waived because of the retrospective study design. All identifying information was removed from this study to protect patient privacy.
The FOLFOX regimen consisted of oxaliplatin 85 mg/m2 on day 1, and LV 200 mg/m2 was administered as a 2-hour infusion before 5-FU 400 mg/m2 was administered as an intravenous bolus. Thereafter, 5-FU 600 mg/m2 was administered as a 22-hour infusion immediately after a 5-FU bolus. LV and 5-FU administration was repeated on days 1 and 2. The FOLFIRI regimen consisted of irinotecan 180 mg/m2 on day 1, and LV 200 mg/m2 was administered as a 2-hour infusion before 5-FU 400 mg/m2 was administered as an intravenous bolus. Thereafter, 5-FU 600 mg/m2 was administered as a 22-hour infusion immediately after a 5-FU bolus injection. LV and FU administration was repeated on days 1 and 2. The CAPOX regimen consisted of oxaliplatin 130 mg/m2 on day 1 in combination with oral capecitabine 1000 mg/m2 twice daily on days 1 to 14. The CAPIRI regimen consisted of irinotecan 240 mg/m2 on day 1 in combination with oral capecitabine 1000 mg/m2 twice daily on days 1 to 14. The FOLFOX and FOLFIRI regimens were repeated at 2-week intervals until disease progression, death, occurrence of unacceptable toxicity, or indication of patient preference, whereas CAPOX and CAPIRI were repeated at 3-week intervals. Most patients received a maximum of 12 cycles of FOLFOX and FOLFIRI or 8 cycles of CAPOX and CAPIRI, according to the national reimbursement criteria.
After the first-line regimen failed, subsequent therapeutic regimens were considered depending on the patients’ performance status, their preference, and the affordability of the agents.
Measurement
The primary endpoints were OS and time to progression (TTP) in the entire cohort. OS and TTP in propensity score-matched populations, response rates, and toxicity were the secondary endpoints. OS was defined as the time from the first day of first-line chemotherapy to death from any cause. TTP was defined as the time from chemotherapy initiation to objective tumor progression. Treatment response was assessed every 2 to 3 months using chest and/or abdominopelvic computed tomography (CT). Tumor response and progression were defined according to the Response Evaluation Criteria for Solid Tumors. Response rates were provided for all patients (intention-to-treat [ITT] analysis) and assessable patients. Toxicity was graded using the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) version 5. The prognostic factors for OS were also evaluated.
Statistical analysis
The statistical methods used were mainly descriptive. Continuous variables were presented as the mean and standard deviation, median and interquartile range (IQR), or minimum and maximum as appropriate. Categorical variables were presented as frequency distributions and percentages. Differences between the treatment groups were evaluated using Pearson’s chi-square test or Fisher’s exact test for categorical variables and analysis of variance or the Kruskal–Wallis test for continuous variables. OS and TTP were evaluated using the Kaplan–Meier method. A stratified two-sided log-rank test was used to compare the treatment groups, and hazard ratios (HRs) were estimated using a stratified Cox proportional hazards regression model. A subgroup analysis was performed to determine the preferred regimen for each subgroup. The propensity score matching principle was implemented for age, sex, liver metastasis, prior adjuvant chemotherapy, metastasectomy status, and bevacizumab co-administration. Univariate analysis was performed for exploratory purposes to examine the prognostic factors for OS. Covariates included age, sex, previous therapy, ECOG performance status, clinical presentation, disease status at the metastatic stage, primary tumor size and location, number of metastatic organs involved, cancer differentiation grade on histopathology, hemoglobin and albumin levels, neutrophil-to-lymphocyte ratio (NLR), baseline initial carcinoembryonic antigen (CEA) levels, CEA responses, and the use of combination therapy with bevacizumab. All P-values were two-sided, with P < 0.05 indicating statistical significance.
Results
Baseline characteristics
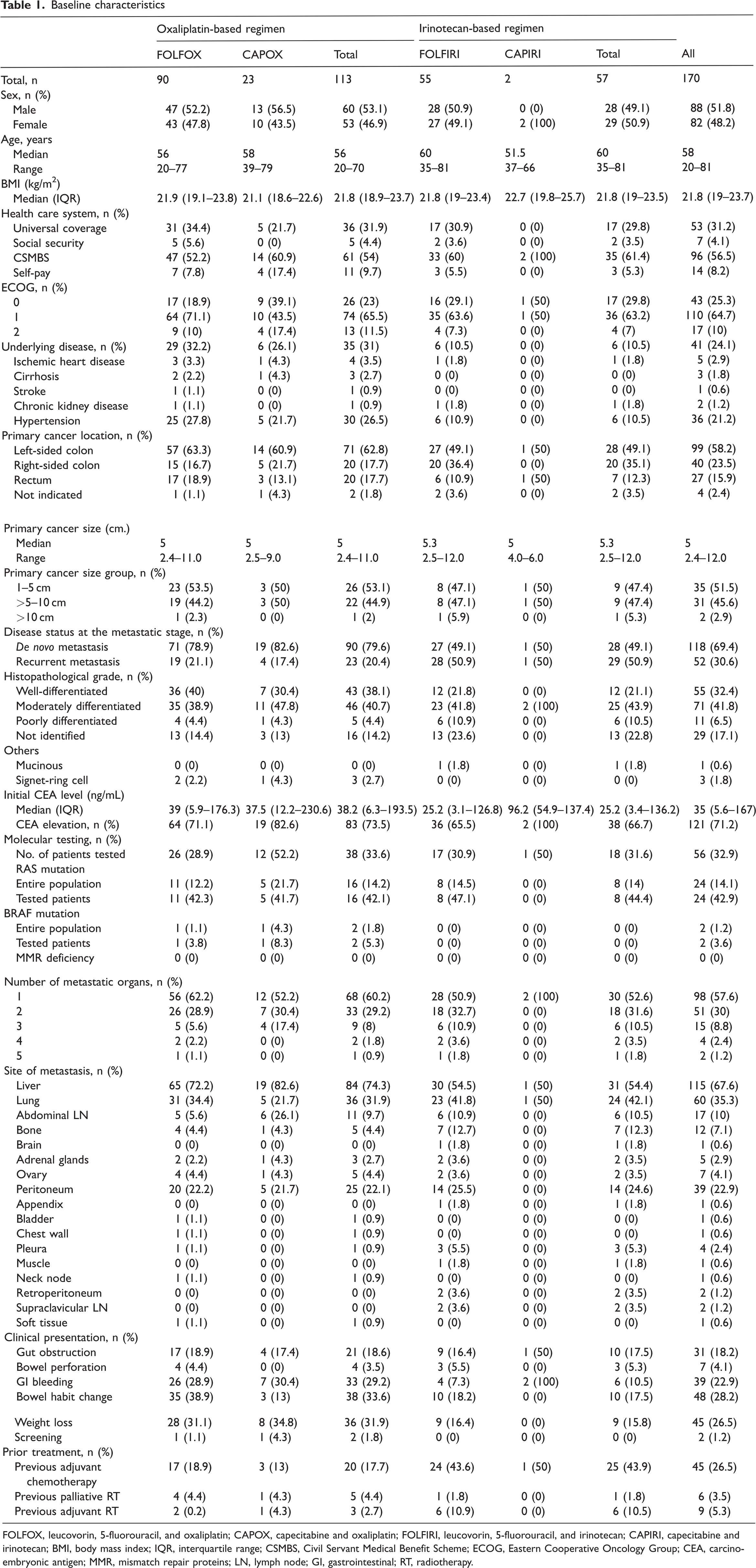
Between January 2003 and March 2021, 239 patients received first-line palliative chemotherapy at the Medical Oncology Service of Prince of Songkla University Hospital. Sixty-nine patients were excluded from this analysis because they received first-line single-agent chemotherapy. In total, 170 consecutive patients (88 men [51.8%]) who received FOLFOX, CAPOX, FOLFIRI, or CAPIRI were included in this study (Figure 1). There were 113 patients in the oxaliplatin-based group (FOLFOX and CAPOX) and 57 patients in the irinotecan-based group (FOLFIRI and CAPIRI). The baseline characteristics of the patients were generally balanced between the two groups, excluding the disease status at the metastatic stage, incidence of liver metastasis, and percentage of patients who received prior adjuvant chemotherapy (Table 1). The median patient age was 58 years, and the median body mass index (BMI) was 21.8 kg/m2. Most patients in both groups had an ECOG performance status of 1. In total, 11% and 7% of patients in the oxaliplatin-based and irinotecan-based groups, respectively, had an ECOG performance status of 2. The primary cancer location was more commonly on the left side of the colon in both groups. The median size of the primary cancer was 5.0 cm. Approximately 70% of the patients presented with de novo metastasis.

Consort diagram for eligibility, treatment, and analysis. The population as of the data cutoff date of October 30, 2021 is presented. PSU, Prince of Songkla University Hospital; FOLFOX, LV, 5-FU, and oxaliplatin; CAPOX, capecitabine and oxaliplatin; FOLFIRI, LV, 5-FU, and irinotecan; CAPIRI, capecitabine and irinotecan; 5-FU, 5-fluorouracil; LV, leucovorin.
Baseline characteristics
FOLFOX, leucovorin, 5-fluorouracil, and oxaliplatin; CAPOX, capecitabine and oxaliplatin; FOLFIRI, leucovorin, 5-fluorouracil, and irinotecan; CAPIRI, capecitabine and irinotecan; BMI, body mass index; IQR, interquartile range; CSMBS, Civil Servant Medical Benefit Scheme; ECOG, Eastern Cooperative Oncology Group; CEA, carcinoembryonic antigen; MMR, mismatch repair proteins; LN, lymph node; GI, gastrointestinal; RT, radiotherapy.
Molecular testing, including RAS mutation, BRAF mutation, and MMR deficiency tests, was performed in 30% of the patients. RAS mutations were found in 42.9% (24/56) of the tested patients. Two patients with BRAF mutations were included in the oxaliplatin-based group. MMR deficiency was not detected.
Sixty percent of the patients had only one metastatic site, and the most common site of metastasis was the liver, followed by the lungs, peritoneum, and abdominal lymph nodes. Liver metastasis was found in 74.3% and 54.4% of the patients in the oxaliplatin- and irinotecan-based groups, respectively. The receipt of prior adjuvant chemotherapy was more common in the irinotecan-based group than in the oxaliplatin-based group (43.9% vs. 17.7%). The adjuvant chemotherapy regimens are listed in Supplementary Table 1.
Treatment pattern
Most patients received combination chemotherapy (71.1%), and approximately two-thirds received the oxaliplatin-based regimen as first-line chemotherapy (66.5%, Table 2). FOLFOX was the most commonly used regimen (52.9%), followed by FOLFIRI (32.4%), CAPOX (13.5%), and CAPIRI (1.2%). Forty percent of the patients received bevacizumab co-administration during first-line treatment.
Treatment patterns.

FOLFOX, leucovorin, 5-fluorouracil, and oxaliplatin; CAPOX, capecitabine and oxaliplatin; FOLFIRI, leucovorin, 5-fluorouracil, and irinotecan; CAPIRI, capecitabine and irinotecan; IQR, interquartile range.
The median numbers of cycles of FOLFOX, CAPOX, FOLFIRI, and CAPIRI were 11, 7, 8, and 10, respectively. The most common cause of treatment discontinuation was treatment completion according to the national reimbursement criteria (39.8% in the oxaliplatin-based group and 35.1% in the irinotecan-based group). Progressive disease was the second most common cause (33.6% in the oxaliplatin-based group and 29.8% in the irinotecan-based group). Adverse events (AEs) leading to treatment discontinuation were observed in 4.4% and 1.8% of the patients in the oxaliplatin- and irinotecan-based groups, respectively. Only 1.8% of the patients received maintenance therapy after a complete course of chemotherapy with no disease progression. Metastasectomy was performed in approximately 20% of the patients in both groups. The most common site of metastasectomy was the liver, followed by the omentum and lungs (Supplementary Table 1).
Half of the patients in whom first-line chemotherapy failed received second-line treatment (Supplementary Table 2), including 54.0% and 49.1% of patients in the oxaliplatin- and irinotecan-based groups, respectively. The irinotecan-based regimen was the most frequently administered regimen in patients who received oxaliplatin-based chemotherapy, and vice versa. Bevacizumab was administered to 20% of the patients who received second-line chemotherapy. Thirty percent of the patients (25/89) who received second-line chemotherapy proceeded to third-line treatment when disease progression developed, and only 20% of those in whom third-line therapy failed received fourth-line treatment. Only five patients received anti-EGFR agents during later-line treatment. One patient with a BRAF mutation received a combination of anti-BRAF, anti-MEK, and anti-EGFR inhibitors as the second-line treatment, which resulted in median OS of 10.8 months. The other patient who had a BRAF mutation did not receive subsequent therapy, and OS in this patient was only 2.5 months.
OS
Overall population
According to the ITT analysis, the median OS times (Figure 2) were 16.8 and 13.0 months in the oxaliplatin- and irinotecan-based groups, respectively (HR = 0.79; 95% confidence interval [CI] = 0.56–1.11; P = 0.175). The 1-, 3-, and 5-year survival rates in the oxaliplatin-based group were 64.7%, 20.5%, and 10.5%, respectively, versus 59.3% (P = 0.327), 18.0% (P = 0.183), and 6.0% (P = 0.169), respectively, in the irinotecan-based group. All patients who survived for 5 years underwent metastasectomy at some point during the course of treatment.

OS and TTP for oxaliplatin- and irinotecan-based regimens in the intention-to-treat and propensity score matching analyses. (a) Median OS among all patients. (b) Median OS among propensity score-matched patients. (c) Median TTP among all patients and (d) Median TTP among propensity score-matched patients. OS, overall survival; TTP, time-to-progression; CI, confidence interval; HR, hazard ratio.
Subgroup analysis of OS between the oxaliplatin- and irinotecan-based groups (Figure 3) revealed no significant difference in benefit across the subgroups. Excluding patients with BMI <18.5 kg/m2 and metastatic organ involvement at two or more sites, the oxaliplatin-based regimen appeared to be more beneficial. However, each subgroup had a small sample size and a wide 95% CI range, suggesting the need for cautious data interpretation. Overall, patients who received second-line treatment had median OS of 19.5 months, versus 8.5 months in patients who did not receive second-line treatment (Supplementary Figure 1).

Subgroup analysis of OS. BMI, body mass index; ECOG, Eastern Cooperative Oncology Group; CEA, carcinoembryonic antigen; CI, confidence interval.
In total, 63 of 113 patients (55.8%) in the oxaliplatin-based group received bevacizumab co-administration as first-line therapy, compared with 27 of 57 patients (47.4%) in the irinotecan-based group. Overall, bevacizumab co-administration during first-line regimen did not improve OS compared with the findings in patients who did not receive this treatment (Supplementary Figure 1). The patients who underwent primary tumor resection with metastasectomy had median OS of 56.9 months, versus 12.7 months for patients who did not undergo metastasectomy (HR = 0.14, 95% CI = 0.07–0.27, P < 0.001; Supplementary Figure 1).
Propensity score matching
After applying the propensity score matching principle with a 2:1 ratio, 82 patients in the oxaliplatin-based group and 41 patients in the irinotecan-based group were assessed. The median OS times (Figure 2) were 17.0 and 16.5 months for patients in the oxaliplatin- and irinotecan-based groups, respectively (HR = 0.94; 95% CI = 0.63–1.4; P = 0.756).
TTP
Overall population
The difference in median TTP between the oxaliplatin- and the irinotecan-based groups was not statistically significant (9.0 months vs. 8.9 months, P = 0.934, Figure 2). The 1-year progression-free survival (PFS) rates in these groups were 28.6% and 29.0%, respectively (P = 0.842). After 2 years, patients who did not experience disease progression or recurrence remained progression-free for at least 5 years. However, the 1–5-year PFS rates did not differ between the oxaliplatin- and irinotecan-based groups.
Propensity score matching
After applying the propensity score matching principle, the median TTP values (Figure 2) were 8.3 and 7.4 months for patients in the oxaliplatin- and irinotecan-based groups, respectively (HR = 0.95; 95% CI = 0.65–1.39; P = 0.8).
Response
In total, 155 patients were considered assessable for response, including 103 patients (91%) in the oxaliplatin-based group and 52 patients (91%) in the irinotecan-based group. Ten patients in the oxaliplatin-based group and five patients in the irinotecan-based group had no data for the response rate evaluation. Furthermore, eight patients were lost to follow-up before the evaluation period, and seven patients were referred to other hospitals before the evaluation.
The response rates for the two treatment groups are presented in Table 3. The objective response rates (ORRs) from the assessable population in the oxaliplatin- and irinotecan-based regimens were 42.7% and 34.6%, respectively (P = 0.424). The clinical benefit rates (CBRs) were 79.6% and 82.7% in the oxaliplatin- and irinotecan-based groups, respectively (P = 0.808). ITT analysis revealed a concordant result with the per-protocol plan. The ORRs in the ITT population for the oxaliplatin- and irinotecan-based regimens were 38.9% and 31.6%, respectively (P = 0.44), whereas the CBRs were 72.6% and 75.4%, respectively (P = 0.829).
Treatment response.

IQR, interquartile range; ORR, objective response rate; CEA, carcinoembryonic antigen.
The complete response (CR) rate was higher in the oxaliplatin-based group (10.7%) than in the irinotecan-based group (5.8%), albeit without significance (P = 0.387). Metastasectomy was performed at a certain point during the treatment course in 78.6% of the patients who achieved CR.
In patients who received bevacizumab co-administration during first-line combination chemotherapy, the ORR increased to 41.7%, compared with 38.6% in patients who did not receive bevacizumab (P = 0.818). Moreover, an actual benefit rate of 5.8% in the ORR (45.8% vs. 40%) was achieved when bevacizumab was added to oxaliplatin-based regimens, whereas no benefit was demonstrated in irinotecan-containing regimens (33.3% vs. 35.7%).
The median durations of response in the oxaliplatin- and irinotecan-based groups were not statistically different (10.3 months vs. 10.6 months). The CEA response after chemotherapy was also evaluated. In total, 9% of the patients who received oxaliplatin-based regimen and 8.7% who received irinotecan-based regimen experienced normalization of CEA levels after treatment. Approximately 70% of the patients from each group experienced a greater than 50% reduction in CEA levels, and they were considered CEA responders.
Toxicity
All patients were assessed for toxicity. The observed toxicities according to CTCAE version 5 are listed in Table 4. Generally, toxicity was mild in both groups. All-grade toxicities were observed in 89.4% and 89.5% of patients who received oxaliplatin- and irinotecan-containing chemotherapy, respectively. Grades 3 to 5 toxicities were observed in approximately half of the patients (54.4%) in the irinotecan-based group and 43.4% of patients in the oxaliplatin-containing group, but the difference was not significant. The rates of all-grade thrombocytopenia (26.5% vs. 5.3%; P = 0.02), febrile neutropenia (3.5% vs. 14.0%; P = 0.022), diarrhea (16.8% vs. 56.1%; P < 0.001), and peripheral neuropathy (48.7% vs. 7.0%; P < 0.001) significantly differed between the oxaliplatin- and irinotecan-containing regimens.
Toxicity.

*P < 0.05.
AST, aspartate aminotransferase; ALT, alanine aminotransferase; ALP, alkaline phosphatase.
In the oxaliplatin-based group, the most common AE was neutropenia (55.8%), followed by leukopenia (54.9%), peripheral neuropathy (48.7%), and thrombocytopenia (26.5%). Only four patients (3.5%) developed grade 3 to 4 peripheral neuropathy leading to discontinuation of treatment. Moreover, one patient discontinued treatment because of grade 3 to 4 episodic febrile neutropenia. The most common toxicity in the irinotecan-containing group was neutropenia (61.4%), followed by leukopenia (59.6%), diarrhea (56.1%), and anemia (33.3%). Among the 21 patients who experienced grade 3 to 4 neutropenia (36.1%), one patient discontinued treatment because of a history of grade 3 to 4 neutropenia. There were two therapy-related deaths from febrile neutropenia, including one in each group.
Prognostic factors
Various parameters were calculated using univariate analysis to define the prognostic factors for OS (Supplementary Table 3). Patients with metastatic organ involvement at two or more sites had worse prognosis than those with one metastatic site. Outcomes were worse among patients with a greater number of metastatic organs. The cancer differentiation grade also influenced the prognosis; specifically, patients with well-differentiated or moderately differentiated carcinoma had better OS than those with poorly differentiated carcinoma. Regarding laboratory parameters, patients with albumin levels ≥3.5 g/dL had a good prognosis, whereas those with NLR > 5 and an elevated initial CEA level experienced poorer prognoses. Metastasectomy was one of the strongest prognostic indicators of favorable prognosis. The CEA response was also a prognostic indicator. Patients who experienced CEA normalization during the treatment had better prognoses than their counterparts. Patients with a CEA response of <40% of the initial value had poorer prognoses. The best-response status from the first-line regimen affected survival. CR was a more favorable prognostic factor than progressive disease. A best response of partial response and stable disease also resulted in better outcomes than progressive disease (Figure 4).
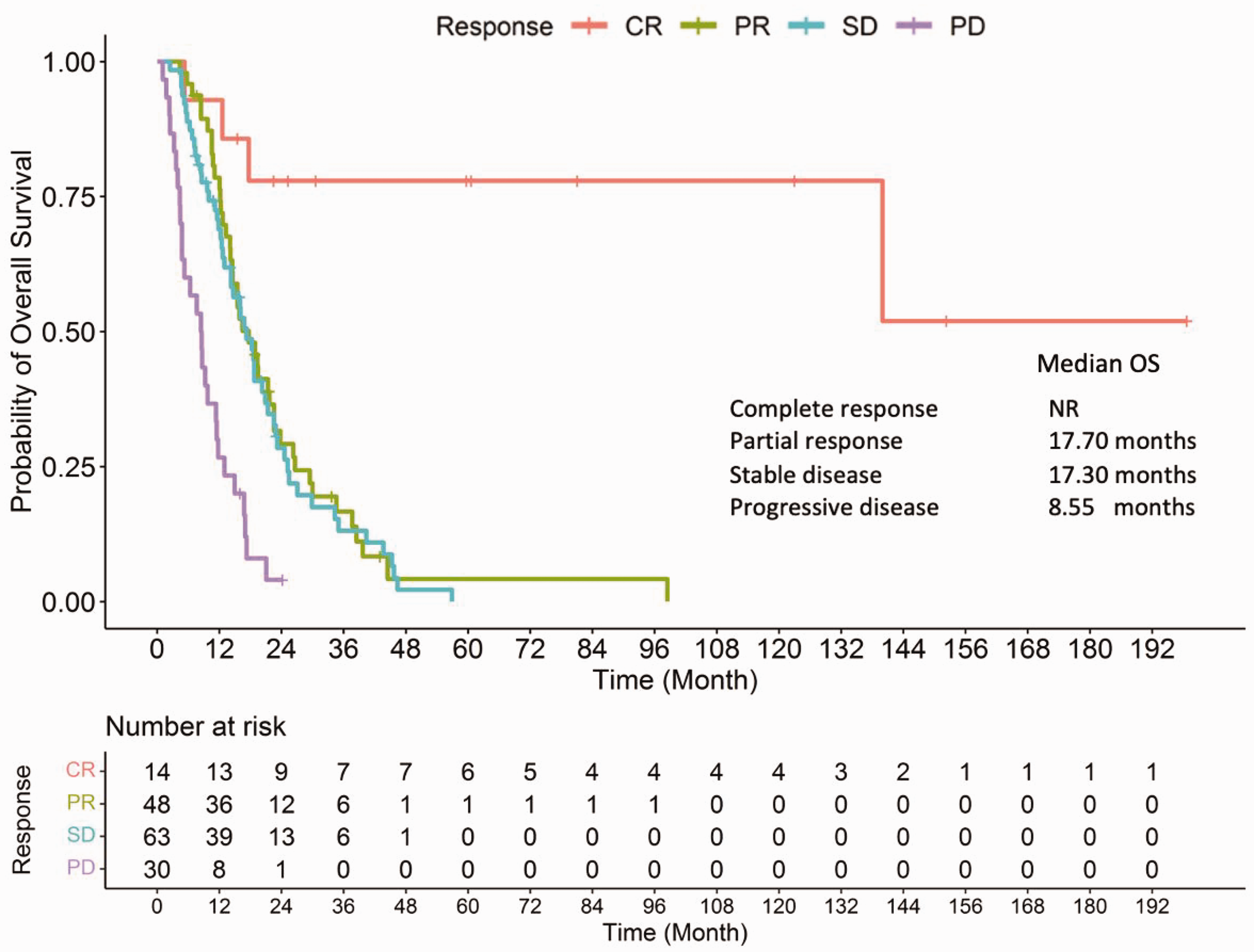
OS according to the best response to first-line treatment. OS, overall survival; CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease; NR, not reached.
Discussion
Combination chemotherapy remains the primary treatment approach for patients with metastatic CRC. Furthermore, the co-administration of biological agents, such as anti-VEGF(R) or anti-EGFR therapies, should be considered to enhance clinical outcomes. MicroRNA (miRNA) has emerged as a potential biomarker for predicting the response to VEGF(R) and EGFR inhibitors. The utilization of miRNA biomarkers can facilitate treatment decisions by aiding in the selection of the most appropriate therapeutic approach for individual patients. 19 However, it is important to note that access to high-cost chemotherapy and/or biological regimens in Thailand is limited, posing a challenge to the widespread implementation and availability of these treatments. 20 , 21 This study demonstrated the characteristics, treatment patterns, and real-world experiences of patients treated with first-line combination chemotherapy for metastatic CRC in a single institution in Southern Thailand. Propensity score matching was applied to decrease the possible bias of the heterogeneous population in each patient group. We found that most patients received standard combination chemotherapy regimens, including FOLFOX4, FOLFIRI, CAPOX, and CAPIRI. Notably, CAPIRI is not widely recommended because of the possibility of severe diarrhea as a common AE. Only two patients from this study received CAPIRI; therefore, the efficacy data of CAPIRI could not be extracted in the current study.
Biomarker testing is recommended as part of standard investigations in CRC because the biomarker results can reveal the most appropriate treatment option for individual patients. In our study, RAS, BRAF, and MMR testing was only performed in 30% of the patients, which is lower than the rates in Western populations, which ranged from 63% to 88%. 22 , 23 Nonetheless, this finding was comparable to the biomarker testing rate of 34% reported in a study in China. 24 KRAS mutation was detected in 42.9% of the tested patients, similar to the results of other reports of Western and Eastern populations.22–24 RAS mutation has been established as a predictive factor to preclude the indication for anti-EGFR inhibitors, 25 but its role as a prognostic indicator remains uncertain. Nuria et al. demonstrated that KRAS mutation was associated with poor prognoses. 25 By contrast, Nazim et al. revealed that KRAS mutation was not a prognostic factor. 26 Our study demonstrated that KRAS mutation was not a prognostic factor; however, this could have been influenced by the small sample size.
Previous studies reported no difference in OS, TTP, and ORR between the FOLFOX and FOLFIRI groups; however, most prospective studies were conducted in Western populations. 12 , 13 A retrospective study in Korea reported median PFS times of 6.2, 7.9, 6.0, and 7.1 months for FOLFOX, CAPOX, FOLFIRI, and CAPIRI, respectively. 27 These results were comparable with those of a large prospective study of Western patients that recorded median TTP of 7.0 months for both FOLFOX and FOLFIRI. 16 Our current study also illustrated that oxaliplatin- and irinotecan-based regimens were active chemotherapy regimens, reaching median TTP values of 9.0 and 8.9 months, respectively.
In this study, differences in the patients’ baseline characteristics between the treatment groups might have confounded the treatment outcomes, namely liver metastasis, the metastasectomy status, prior adjuvant therapy, and bevacizumab co-administration. Hence, propensity score matching was applied to adjust for the effects of these factors. However, after propensity score matching, the OS outcome was not statistically different. Thus, the chemotherapy regimens might have similar efficacy. Regarding OS, our data revealed median OS times of 16.8 and 13.0 months for patients in the oxaliplatin- and irinotecan-based groups, respectively, and 17.0 and 16.5 months, respectively, with all propensity score matching models included. In comparison, studies of Western populations recorded OS times of 14.0 and 15.0 months in the FOLFOX and FOLFIRI groups, respectively. 16 This study confirmed that FOLFOX and FOLFIRI should be used in Thai patients to provide the greatest clinical benefit. The analysis of subgroup populations illustrated that patients with low BMI and multiple metastatic sites benefited more from FOLFOX than from FOLFIRI. However, this exploratory analysis was conducted in a small population with no previous supporting reports. Therefore, additional studies should be conducted to provide clearer evidence.
All patients who achieved 5-year survival underwent metastasectomy at a certain point during treatment. However, the choice of chemotherapy regimen for each patient depends on tumor biology, the patient’s characteristics, and his or her preferences. The toxicity profile of chemotherapy is considered a practical point for identified the optimal regimen for each patient. In this study, neuropathy and thrombocytopenia were common AEs associated with oxaliplatin, similar to previous findings. 16 , 27 Diarrhea was more prevalent in the irinotecan-based group. However, neutropenia was commonly found in the irinotecan-based group, occurring at a higher rate than that previously reported. 16 , 27 Despite this, most AEs were mild in severity. Only six patients (3.5%) discontinued treatment because of toxicity. Furthermore, the efficacy of FOLFOX and FOLFIRI were similar, and the cost-effectiveness profile did not differ between the regimens. 28
Adding bevacizumab to standard first-line combination chemotherapy increased median PFS, OS, and ORR to 9.4 to 10.6 months, 21.3 to 29.0 months, and 55.2%, respectively. 29 , 30 Our study recorded TTP, OS, and ORR of 9 months, 15 months, and 41.7%, respectively, in patients who received bevacizumab. However, this difference could be partly affected by the small proportion of patients with bevacizumab co-administration (40%) during first-line treatment. In Thailand, bevacizumab reimbursement is only approved for second-line treatment in patients categorized in the Civil Servant Medical Benefit Scheme, which covers 20% to 25% of the entire population. Anti-EGFR inhibitors, including panitumumab and cetuximab, have been approved as effective targeted therapies when added to first-line chemotherapy in patients with extended RAS and/or BRAF wild-type metastatic CRC. However, none of the patients in our study received anti-EGFR monoclonal antibodies during first-line treatment owing to the limited criterion for national reimbursement and the affordability of these agents for patients paying out of pocket. Only four patients (2.4%) received anti-EGFR inhibitors during later-line treatment.
Patients who underwent primary tumor resection and metastasectomy at a certain point during treatment experienced significantly prolonged median OS of 56.9 months. Conversely, patients with unresectable disease had median OS of 12.7 months. The 5-year OS rate in patients undergoing resection was 47.7%, which is comparable with previous findings of 25% of 40% in patients who underwent metastasectomy. 31 Patients who received chemotherapy alone rarely survived beyond 3 years (3-year OS = 9.6%), and none lived beyond 5 years. With this finding, a multi-disciplinary approach with co-evaluation by the colorectal and hepato-pancreato-biliary surgery teams should always be included to consider primary tumor resection and metastasectomy in patients with potentially resectable metastatic CRC to maximize the benefit of treatment.
In both the oxaliplatin- and irinotecan-based groups, approximately 50% of patients received second-line treatment. Patients in the oxaliplatin-based group most commonly received irinotecan-based treatment in the second line, and vice versa. The proportion of patients who received subsequent treatment differed from that reported in previous studies in European countries (58%–60%) 22 , 23 and in China (31%). 24 However, the standard subsequent chemotherapy regimens were similar. Our study confirmed that the receipt of second-line treatment is a favorable prognostic factor. OS in patients who received second-line treatment was 11 months. Although bevacizumab co-administration during second-line treatment could be reimbursed in some patients, only 20% of patients received this treatment. Tournigand et al. concluded that the sequence of FOLFOX and FOLFIRI therapy did not affect survival outcomes, 17 in line with our findings. However, later-line treatment was administered to only 14.7% of patients, which was lower than that in a study in Spain (27%). 23 However, the figure was is higher than that in China (10.4%). 24 Different numbers of lines of treatment could lead to differences in OS.
Previous studies provided evidence supporting several factors associated with shorter survival in patients with CRC, including age >70 years, multiple disease sites, peritoneal metastasis, right-sided disease, poorly differentiated histopathology, high NLRs, KRAS mutation, and progressive disease following first-line chemotherapy. 22 , 32 However, the prognosis of older patients might be influenced by preexisting comorbidities and the type of treatment received. Although older patients with T4 CRC are more susceptible to severe postoperative complications, there is no consensus regarding the impact of age on survival outcomes in non-metastatic stage CRC. 33 Conversely, hemoglobin levels of ≥10 g/dL and primary surgery with metastasectomy were reported to be associated with a better prognosis. 22 , 34 Our study incorporated these findings and demonstrated that multiple sites of metastasis (>1), a high NLR (>5), and poorly differentiated histopathology portended a poor prognosis. Conversely, an albumin level of ≥3.5 g/dL and a history of metastasectomy were associated with favorable outcomes. Furthermore, our study revealed that a reduction in the CEA level was indicative of a favorable prognosis, as patients who achieved normalized CEA levels after treatment exhibited better survival outcomes. However, in the case of non-normalized CEA levels, reduced CEA levels were also associated with a good prognosis. Our study revealed that the patients with ≤40% reductions in CEA levels from baseline through the treatment period had worse OS.
The current study had several limitations. First, this was a single-center, retrospective study. There might have been some imbalance in characteristics between the treatment groups. The OS and TTP data might not reflect actual findings compared with those retrieved from prospective studies because OS was calculated from the first day of chemotherapy. Therefore, the duration from diagnosis to treatment initiation can vary in individual patients, and survival outcomes might have been affected in patients whose treatments were delayed. Second, the patients in this study were not representative of all patients with metastatic CRC from our institution, as the cohort mainly included patients who were directly administered chemotherapy by medical oncologists to standardize the chemotherapy regimens and the monitoring schema. Third, the major cause of discontinuation of first-line chemotherapy was the national reimbursement criterion opposed to disease progression. Median TTP and OS might have been longer if most patients had been able to continue chemotherapy until disease progression or the occurrence of unacceptable toxicity. Fourth, biological agents, namely anti-VEGF(R) or anti-EGFR inhibitors, were infrequently co-administered with first-line chemotherapy according to the national reimbursement policy. This might have partly contributed to the short OS in this study.
In conclusion, from our real-world experience with long-term follow-up, first-line combination chemotherapy, including FOLFOX, CAPOX, and FOLFIRI, for patients with metastatic CRC had comparable efficacy in line with previous findings. The opportunity to undergo primary tumor resection and metastasectomy was a key factor in achieving better treatment outcomes in patients with potentially resectable disease. Hence, a multidisciplinary approach is essential for patients with metastatic CRC.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231193583 - Supplemental material for Nineteen-year, real-world experience of first-line combination chemotherapy in patients with metastatic colorectal cancer: a propensity score analysis from southern Thailand
Supplemental material, sj-pdf-1-imr-10.1177_03000605231193583 for Nineteen-year, real-world experience of first-line combination chemotherapy in patients with metastatic colorectal cancer: a propensity score analysis from southern Thailand by Jirapat Wonglhow, Chirawadee Sathitruangsak, Arunee Dechaphunkul and Patrapim Sunpaweravong in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605231193583 - Supplemental material for Nineteen-year, real-world experience of first-line combination chemotherapy in patients with metastatic colorectal cancer: a propensity score analysis from southern Thailand
Supplemental material, sj-pdf-2-imr-10.1177_03000605231193583 for Nineteen-year, real-world experience of first-line combination chemotherapy in patients with metastatic colorectal cancer: a propensity score analysis from southern Thailand by Jirapat Wonglhow, Chirawadee Sathitruangsak, Arunee Dechaphunkul and Patrapim Sunpaweravong in Journal of International Medical Research
Footnotes
Acknowledgments
We thank all patients and families for participating in the study. We additionally thank Pruphetkaew N for assistance with statistical analysis and Samusor S and Songserm M for assistance with data collection. We also would like to thank Editage (![]() ) for the English language review. A fraction of patients in this study received irinotecan through a donation from Berli Jucker Public Company Limited (study protocol number 55-294-14-1-1).
) for the English language review. A fraction of patients in this study received irinotecan through a donation from Berli Jucker Public Company Limited (study protocol number 55-294-14-1-1).
Author contributions
WJ and SP conceived and designed the analysis. WJ collected the data. WJ, SC, DA, and SP contributed to data analysis. WJ performed the analysis. WJ, SC, DA, and SP drafted, critically reviewed, and finally approved the article.
Availability of data and materials
The original contributions presented in the study are included in the article/additional file. Further inquiries can be directed to the corresponding author.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
