Abstract
Temporal lobe epilepsy is the most common focal epilepsy syndrome and has a broad spectrum of presentations. Nevertheless, isolated vestibular symptoms without other symptoms typical of temporal lobe seizures are relatively rare. Here, we report one female patient who suffered from chronic refractory vertigo and had inappropriate pharmacotherapy for several years. Eventually, epileptic vertigo and dizziness (ictal vertigo) were accurately diagnosed by detailed history taking and serial examinations assisted by sphenoid electroencephalography. Awareness of this unique syndrome is important in the diagnosis of patients with epileptic vertigo and dizziness.
Keywords
Introduction
Epilepsy is a condition defined by recurrent seizures caused by an imbalance between excitatory and inhibitory control over certain brain cells in cortical areas. Temporal lobe epilepsy (TLE) is the most common type of focal epilepsy and exhibits a broad spectrum of symptoms that vary depending on whether it is localized in the mesial or neocortical regions. Common clinical manifestations include abnormal sensation (e.g., déjà vu), unexplainable fear and anxiety, auditory and visual hallucinations, and amnesia.1,2 Under relatively rare conditions, TLE can also present with vestibular symptoms such as vertigo or dizziness without obvious convulsion or disturbance of consciousness.2,3 A comprehensive systematic review of epileptic vertigo or dizziness (EVD) identified 84 studies that collectively described 11,354 patients experiencing vertigo or dizziness in relation to their seizures. Among these patients, some experienced vertigo or dizziness solely because of seizures (EVD-only). Among the total patient population, only 1055 individuals were suspected to have EVD, and a mere 0.8% of these patients received a confirmed diagnosis of isolated EVD. Failure to identify this disorder early may result in a delay in treatment. Here, we report one case of unilateral non-lesional TLE presenting as chronic and intractable ictal vertigo to enhance the awareness and improve the medical care of this unique syndrome.
Case report
We have de-identified all patient details, and we obtained verbal patient consent for treatment and publication of this study.
A female patient in her 30 s, who was right-handed, had been suffering from intermittent dizziness and vertigo for years. Each episode typically lasted approximately 5 s, but sometimes episodes would last 3 to 10 minutes, and she was fully aware during these episodes. Most of the time these dizziness and vertigo episodes were mild, although sometimes they could be of moderate severity, which temporarily impaired her ongoing activities. For some episodes, she would describe the dizziness and vertigo as a sensation of being in a sinking boat, feeling intensely imbalanced. These episodes would occur approximately three to ten times a month. More rarely, episodes described as an experience of having a “poor understanding of what people say” also occurred. The episodes were not provoked by posture change, including head turning, and the patient had no persistent tinnitus or hearing deficit.
The patient had sought medical attention at both ear, nose, and throat and neurological clinics on multiple occasions, including ear, nose, and throat evaluation with otoscopy examination and a Hallpike test, but no definitive diagnosis was made. Despite undergoing numerous medication trials, the patient experienced no improvement in her condition. Her physical and neurological examinations yielded essentially normal results. Additionally, during earlier investigations, there were indications of questionable epileptiform discharges on electroencephalography (EEG) in the left temporal area. Based on the paroxysmal clinical features and abnormal EEG findings, epileptic vertigo was suspected. She was prescribed several different anti-seizure medications (ASMs), and the episodes subsided substantially to approximately once or twice per month. Consequently, the patient was referred to our specialized facility, which focuses on epilepsy management, for a more comprehensive investigation.
This patient had normal cognitive performance. Her brain magnetic resonance images were essentially normal, which included the following sequences: localizer 3-plane scout, axial (T2 fs, fast spin-echo (FSE)), T2 (fluid attenuated inversion recovery (FLAIR), diffusion-weighted imaging ALL b1000), three-dimensional (3D) axial time of flight spoiled gradient recalled echo, O-COR T2 FSE, 3D (axial T1 BRAVO, axial SWAN, Fast), with contrast (axial T1 FS FLAIR, coronal T1 FLAIR, sagittal T1 FLAIR, 3D axial T1 BRAVO). To gain a better understanding of the paroxysmal nature of these findings and to investigate the suspected seizure disorder by focusing on the temporal lobe, we planned a more in-depth investigation. This involved scrutinizing both the mesial and lateral aspects of the temporal areas through an interictal sphenoid EEG examination. 4 The patient's interictal sphenoid EEG recording showed intermittent epileptiform discharges, presenting as paroxysmal spike-and-wave complexes over the left temporal area, with maximal activity over the mesial (Sp1) and mid-temporal (T3) areas with regional involvement and essentially normal background activity (Figure 1). During EEG monitoring, the patient's interictal epileptiform discharges were spread over the left temporal area, with maximal activity over the T3 and F7 areas with regional involvement. No contralateral discharges occurred during the monitoring. While the patient was having an attack of habitual vertigo, one episode with a short run (8 s) of quasi-rhythmic focal delta activity (1.5 Hz, medium to high amplitude) occurred in the T3, F7, and T5 areas. The ictal epileptiform discharges continued to show focal, sharp, and delta activity (1.5 Hz, medium to high amplitude) in the left temporal area, with initial mild accentuation in amplitude and a gradual decrease in frequency (to 1 Hz) and in amplitude. The Laplacian montage showed prominent sharp-and-slow delta waves (1.5 Hz) at T3 with regional involvement at T5 and less prominent involvement at F7 (Figure 2). During the episode, the patient remained aware, although she experienced an episode of strong vertigo, describing it as the sensation of swinging in a floating boat. Based on the multiple, episodic, non-positional attacks of vertigo with brief duration, which correlated with ictal EEG abnormalities, a diagnosis of left non-lesional TLE was established. Oral lacosamide (150 mg twice per day), lamotrigine (100 mg twice per day), levetiracetam (1000 mg twice per day), and perampanel (4 mg once at bedtime) were prescribed for seizure control. The frequency of the episodes decreased to one to two episodes per 3 months, with each episode lasting only a few seconds and with consciousness remaining clear.

Electroencephalography with sphenoidal electrodes in a common average referential montage showed paroxysmal spike-and-wave complexes over the left temporal area. Maximal activity was observed over the mesial (Sp1) and mid-temporal (T3) areas with regional involvement and essentially normal background activity.
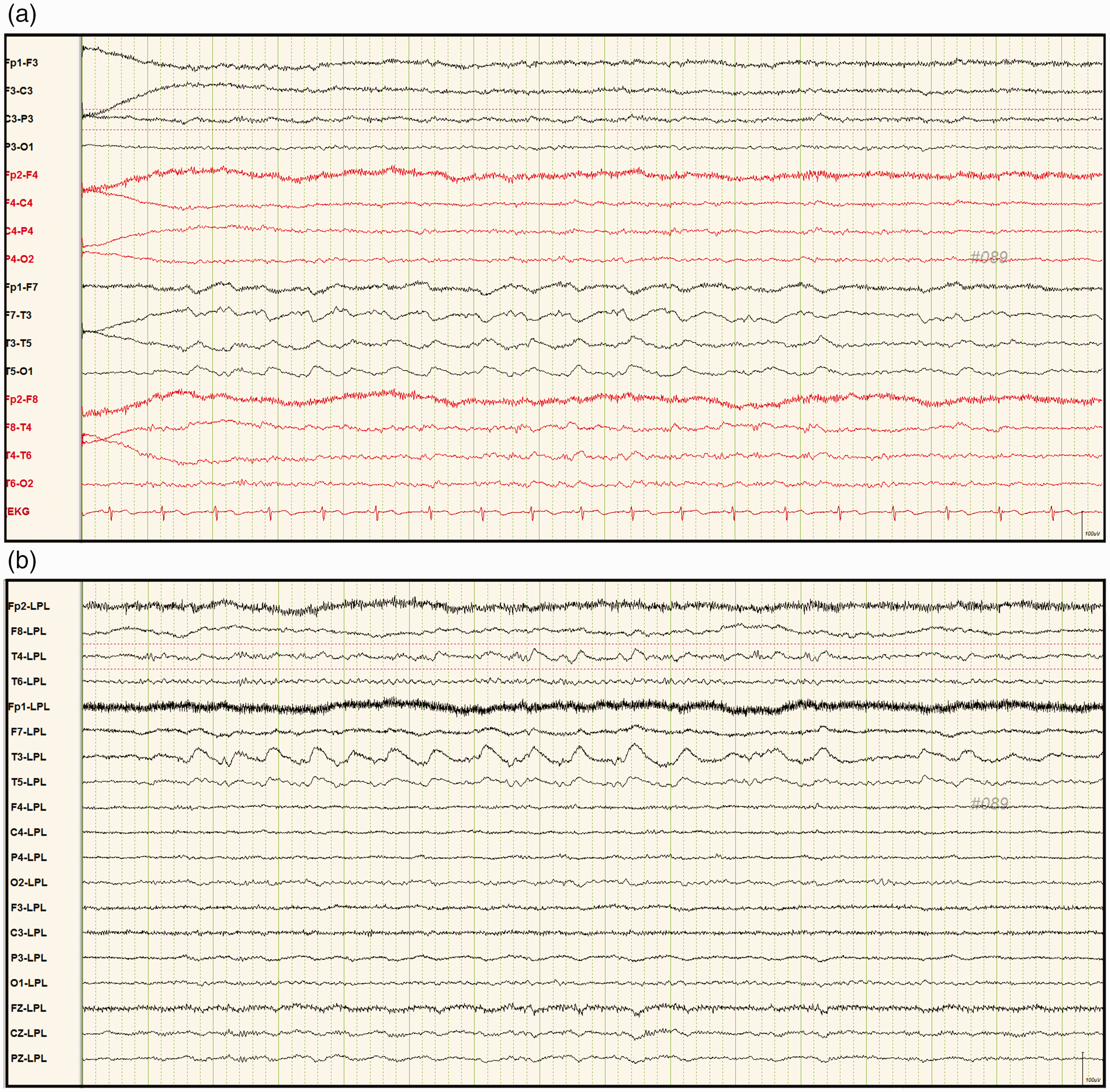
(a) Electroencephalography during longitudinal bipolar montage monitoring while the patient was having an attack of habitual vertigo showed one episode of a short run (8 s) of quasi-rhythmic focal delta activity (1.5 Hz, medium to high amplitude) in T3, F7, and T5 and (b) The Laplacian montage showed prominent sharp-and-slow delta waves (1.5 Hz) at T3 with regional involvement at T5 and less prominent involvement at F7.
The reporting of this study conforms to the CARE guidelines. 5
Discussion
Seizure and vertigo are both common symptoms and diagnoses encountered in the general population. Although it is not uncommon for epileptic seizures to be accompanied by vertigo or dizziness, these symptoms mainly appear in the post-ictal state or as adverse effects of ASMs. Nevertheless, isolated vertigo/dizziness are uncommon in epilepsy and are usually suspected to be related to ear or cardiogenic problems or even to metabolic disturbances. Recent systematic reviews2,3,6–8 have reported that the concept of ictal vertigo or EVD is a type of partial seizure that lacks prominent alteration of consciousness, visible generalized convulsions, automatism, or focal twitching. Nevertheless, there are numerous differential diagnoses related to chronic vertigo, including disorders in many specialty medical fields such as otolaryngology (benign paroxysmal peripheral vertigo, Meniere’s disease), neurology (partial seizure, transient ischemic attack, vertebrobasilar insufficiency, episodic ataxia, basilar migraine), cardiology (arrhythmias), and even psychiatry (generalized anxiety disorder). Because of the chronic course of paroxysmal vertigo accompanied with the ineffectiveness of multiple anti-vertiginous agents and lack of other medical problems in our patient, EVD was considered in the differential diagnosis.
Active epileptiform discharges originating from the superior temporal gyrus or temporo–parieto–occipital junction, the parietal lobe, and the frontal lobe are reported to present as EVD in some studies.2,6–8 A retrospective analysis of video-EEG of 40 patients with vertiginous seizure revealed 20 epileptiform discharges from the lateral temporal lobe and eight from the mesial temporal lobe at seizure onset. 9 The other epileptiform discharges included four from the frontal lobe, one from the parietal lobe, and seven from the occipital lobe. In general, the most common site is the temporal area, especially the lateral temporal area and temporo–parieto–occipital junction, or the vestibular cortex.2,6–8,10 Epileptic vertigo is usually a feature of the non-dominant temporal lobe, although it can have a left temporal origin. 6 It is relatively uncommon to encounter interictal involvement of the mesial temporal area presenting as EVD, as in our case.
Certain ASMs such as carbamazepine, phenytoin, valproic acid, and vigabatrin have been linked to hearing-related side effects. Prolonged use of ASMs such as carbamazepine, phenytoin, valproate, lamotrigine, gabapentin, vigabatrin, and oxcarbazepine may lead to symptoms such as tinnitus, phonophobia, sensorineural hearing loss, dizziness, ataxia, and disequilibrium. 11 However, the patient consistently experienced paroxysmal vertigo, which was confirmed through video-EEG monitoring. This strengthened our confidence in diagnosing the patient with epileptic vertigo. Additionally, the medications prescribed to the patient rarely cause hearing-related adverse effects, and these medications significantly reduced the frequency of her vertigo episodes, both subjectively and objectively. Considering its potential for good efficacy, carbamazepine could also be considered as a further treatment option, with careful monitoring.
In the current patient, the ictal epileptiform activity most likely originated from the mesial and mid-temporal areas, as supported by interictal and ictal EEG. According to previous studies,2,6,7 the most important diagnostic approach for EVD is interictal EEG, with most of these studies using scalp recording and a few using intracranial recording. Although long-term invasive intracranial recording, such as intracranial EEG, offers more details and accurate information for diagnosis, it might not be suitable in a routine outpatient setting in clinical practice. In our case, sphenoid EEG revealed paroxysmal spike-and-wave complexes over the left temporal area and confirmed the diagnosis of epileptic origin.
It is probable that propagation occurred between the mesial and mid-temporal areas and the neighboring areas, including the lateral temporal area and possibly the superior temporal area, which is likely to be at least partly responsible for the EVD in our case. One case series study showed a patient with right mesial temporal lobe seizures who experienced vertigo or dizziness during stimulation of the right superior temporal area 9 and another patient with right ictal mesial temporal onset who showed vertigo during stimulation of the same area but had pathology consisting of focal cortical dysplasia with hippocampal sclerosis. 9 Of note, in a study of propagation patterns in mesial temporal epilepsy, most of the mesial temporal seizures observed showed propagation limited to the ipsilateral hemisphere at the onset of discharge or a graduated sequential propagation. 12 The former is approximately compatible with our case.
In conclusion, EVD is a rare clinical manifestation and a medical challenge, especially under a chronic, refractory course, without a definite diagnosis. We demonstrated that a sphenoid EEG study could be a useful and convenient tool to establish the diagnosis when temporal lobe-related isolated EVD is clinically suspected. Further studies with larger samples of patients with non-lesional TLE and sphenoid EEG recording as well as further investigations of the underlying physio-pathological and pharmacological relationship are warranted.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231187801 - Supplemental material for Unilateral non-lesional temporal lobe epilepsy presenting as isolated ictal vertigo: a case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605231187801 for Unilateral non-lesional temporal lobe epilepsy presenting as isolated ictal vertigo: a case report by Chih-Hung Tsai, Tsang-Shan Chen, Ming-Chi Lai and Chin-Wei Huang in Journal of International Medical Research
Footnotes
Acknowledgements
The authors gratefully extend their appreciation to the staff in the Department of Neurology for their valuable comments on this case.
Author contributions
CH Tsai, TS Chen, and CW Huang clinically evaluated the case. All authors participated in analyzing the case. CW Huang conducted the clinical data interpretation and obtained funding. CH Tsai prepared the first draft of the protocol paper, with contributions and editing of subsequent drafts by TS Chen, MC Lai, and CW Huang. All authors critically reviewed the manuscript and approved the submitted version.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interest
The authors declare no conflicts of interest.
Ethics statement
Because this was a single-case descriptive study, institutional review board approval was not required.
Funding
This work was supported in part by a grant from the Ministry of Science and Technology, Taiwan (MOST-111-2314-B-006 -103 -MY2). This case report was presented at the Taiwan Neurological Society Annual Meeting in 2019.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
