Abstract
Objectives
To investigate the floating, structural, and controlled-release characteristics of a floating poly(lactic-co-glycolic acid) (PLGA)-based controlled-release drug delivery system, and determine the feasibility of this drug delivery system for intravesical instillation.
Methods
PLGA was dissolved in dimethylacetamide, then mixed with IR780 and doxorubicin (DOX) to prepare a drug delivery system capable of solidification and flotation on water at room temperature. Preparations of PLGA, PLGA+IR780, PLGA+DOX, and PLGA+IR780+DOX were formulated. Their floating characteristics in vivo and in vitro were investigated, along with their structural and controlled-release characteristics. Preparations of saline, DOX, and PLGA+IR780+DOX were also formulated; the content of DOX in bladder tissue delivered by each preparation was determined by fluorescence microscopy.
Results
PLGA exhibited stable flotation in vivo and in vitro. A honeycomb structure was observed by scanning electron microscopy. When irradiated with a near-infrared laser, IR780 generated heat that vitrified PLGA, allowing controlled release of DOX from the drug delivery system. The PLGA+IR780+DOX preparation achieved the highest content of DOX in bladder tissue.
Conclusions
Our floating PLGA-based controlled-release drug delivery system exhibited a honeycomb stabilized structure and achieved controlled release when irradiated by a near-infrared laser, making it an ideal drug delivery system for intravesical instillation.
Keywords
Introduction
Bladder cancer is among the most common malignancies in humans; non-muscle-invasive bladder cancer (NMIBC) and muscle-invasive bladder cancer constitute approximately 75% and 25% of cases, respectively. Transurethral resection of bladder tumor (TURBT) is considered the gold standard for treatment of NMIBC.1–2 After TURBT for NMIBC, the rates of recurrence are 12% to 67% within 1 year and 24% to 84% within 5 years; 14% to 21% of affected patients may develop muscle-invasive bladder cancer.3–5 The use of intravesical instillation chemotherapy immediately after TURBT can substantially reduce the risk of recurrence. 6
Intravesical instillation chemotherapy mainly destroys circulating tumor cells after TURBT; it also has an ablative effect on residual tumor cells at the resection site and on small overlooked tumours. 7 There is increasing evidence that prolonged instillation chemotherapy is more effective than brief instillation chemotherapy; 8 the drug concentration is reportedly more important than the treatment duration. 9 Microwave-induced hyperthermia, hyperthermic intravesical chemotherapy, and electromotive drug administration can improve drug efficacy.9–11 However, these methods require urinary catheter insertion and removal. To our knowledge, no studies have demonstrated a treatment approach without urinary catheter involvement. Patients with NMIBC require regular intravesical instillation, which includes urinary catheter insertion and removal; this process causes psychological and physical discomfort. Hematuria, urinary tract infection, and severe complications (e.g., urethral strictures) may occur. 12 Thus, there is an urgent need to develop a controllable floating drug delivery system that can be delivered repeatedly in clinical practice, with multiple pulsed releases after a single instillation.
Poly(lactic-co-glycolic acid) (PLGA), a non-toxic biodegradable biomaterial, is widely used to prepare medical stent materials. 13 PLGA can encapsulate various hydrophobic and hydrophilic agents within drug delivery systems.14–16 At a temperature of 40°C, PLGA undergoes a glass transition, which generates a rigid chain structure and reduces medium adsorption. When the temperature returns to <40°C, PLGA regains its original structure. 17 Furthermore, dimethylacetamide (DMAC) demonstrates high thermal stability, hydrolysis resistance, and robust solubility; it is mainly used in synthetic fibers and medical applications.18,19 Finally, IR780 is a fat-soluble molecule that can strongly absorb near-infrared light and dissipate energy via heat production.20,21 According to the results of a pilot experiment, we prepared a floating PLGA-based drug delivery system by dissolving PLGA in DMAC, then mixing it with IR780 and doxorubicin (DOX).
This study explored the floating, structural, and controlled-release characteristics of this floating PLGA-based system, then evaluated its feasibility for intravesical instillation.
Materials and Methods
Key reagents, animals, and ethical approval
PLGA (n(LA):n(GA) = 80:20), DMAC, and IR780 were purchased from Sigma-Aldrich (USA). DOX was acquired from Zhejiang Hisun Pharmaceutical Co., Ltd. (Taizhou, China). Disodium hydrogen phosphate (Na2HPO4), potassium dihydrogen phosphate (KH2PO4), and sodium chloride (NaCl) were obtained from Nanjing Wanqing Chemical Glassware Instrument Co., Ltd. (Nanjing, China).
Female BALB/c mice were purchased from the Institute of Comparative Medicine, Yangzhou University. Ethical approval for the study protocol was obtained from the Ethics Committee of the Second Affiliated Hospital of Soochow University (no. JD-LK-2021-139-01).
Preparation of floating PLGA and artificial urine-based controlled-release drug delivery system
At room temperature, 200 mg of PLGA were dissolved in 1 mL of DMAC, then thoroughly mixed with 1 mg of IR780 and 1 mg of DOX. The mixture was manually stirred to produce a controlled-release drug delivery system. Artificial urine was composed of 0.2 g Na2HPO4, 0.2 g KH2PO4, and 18 g NaCl dissolved in distilled water to a final volume of 1000 mL. 22
Experimental preparations
Four experimental preparations were established: 200 mg of PLGA dissolved in 1 mL of DMAC (PLGA); 200 mg of PLGA dissolved in 1 mL of DMAC, mixed with 1 mg of DOX (PLGA+DOX); 200 mg of PLGA dissolved in 1 mL of DMAC, mixed with 1 mg of IR780 (PLGA+IR780); and 200 mg of PLGA dissolved in 1 mL of DMAC, mixed with 1 mg of IR780 and 1 mg of DOX (PLGA+IR780+DOX).
Flotation experiments and scanning electron microscopy
For the in vitro flotation experiment, the experimental preparations were separately added to distilled water to observe their floating characteristics.
For the in vivo flotation experiment, female BALB/c mice were immobilized and anesthetized via intraperitoneal administration of 3% pentobarbital sodium. Subsequently, they were catheterized. Then, 0.1 mL of the PLGA+IR780+DOX preparation was injected through the catheter, and color Doppler ultrasound was performed. The position of the PLGA+IR780+DOX preparation in the bladder was observed when the mice were in different positions, revealing the floating characteristics of the drug delivery system.
For structural analysis, the experimental preparations were incubated in water, then observed on a 100-µm scale by scanning electron microscopy (Quanta Inspect; FEI, Hillsboro, OR, USA).
DOX standard curve mapping
A fluorescence spectrophotometer was used to measure the amounts of fluorescence released by six different concentrations of DOX, yielding a standard curve. This curve was used to determine the amount of DOX released by the drug delivery system. The concentrations of DOX were 20, 10, 5, 2.5, 1.25, and 0.75 µg/mL; these concentrations were prepared by dissolving 1 mg of DOX hydrochloride powder in 1 mL of distilled water to prepare a liquid of 1 mg/mL, then diluting the original solution as necessary to obtain the indicated concentrations.
Experimental drug release in water and artificial urine
The PLGA+IR780+DOX drug delivery system was divided into experimental and control groups (1 mL each). In the control group, the system was floated in the water and DOX was naturally released without infrared light excitation. In the experimental group, the system was excited by near-infrared laser irradiation for 10 seconds at 30 minutes, 60 minutes, and 90 minutes after addition to water; the amount of DOX released into the water was determined by measurement of fluorescence intensity. The same assessment was also performed in artificial urine.
Evaluation of DOX in bladder tissue
Three experimental preparations were formulated: saline, DOX, and PLGA+IR780+DOX. The DOX and PLGA+IR780+DOX preparations both contained 100 mg of DOX. Using a catheter, each preparation was injected into a mouse bladder. The PLGA+IR780+DOX preparation was irradiated with near-infrared laser for 10 seconds at 30 minutes, 60 minutes, and 90 minutes after injection; the other preparations were not irradiated. After 120 minutes, mice were sacrificed using a protocol approved by the State Council of the People’s Republic of China. Intact bladder tissue was harvested, and the fluorescence intensity of DOX released from each preparation was measured in bladder tissue.
Results
Flotation and structural characteristics
The four preparations exhibited stable flotation in artificial urine. Additionally, IR780 and DOX dissolved in water exhibited green and red color, respectively. As shown in Figure 1, the aqueous solutions of PLGA+DOX, PLGA+IR780, and PLGA+IR780+DOX were light red, green, and reddish-green, respectively. Thus, the systems exhibited stable flotation in water, while slowly releasing the drug.

Preparations suspended in water. (a) PLGA, (b) PLGA+DOX, (c) LGA+IR780, and (d) PLGA+IR780+DOX. PLGA, poly(lactic-co-glycolic acid); DOX, doxorubicin.
Color Doppler ultrasound revealed that the PLGA+IR780+DOX preparation stably floated in the upper part of the bladder (Figure 2).

Preparation of PLGA+IR780+DOX floating in mouse bladder. PLGA, poly(lactic-co-glycolic acid); DOX, doxorubicin.
After incubation in water and subsequent solidification, each experimental preparation was observed by scanning electron microscopy (scale bar: 100 μm), revealing an interior honeycomb structure that allowed flotation in vitro (Figure 3).
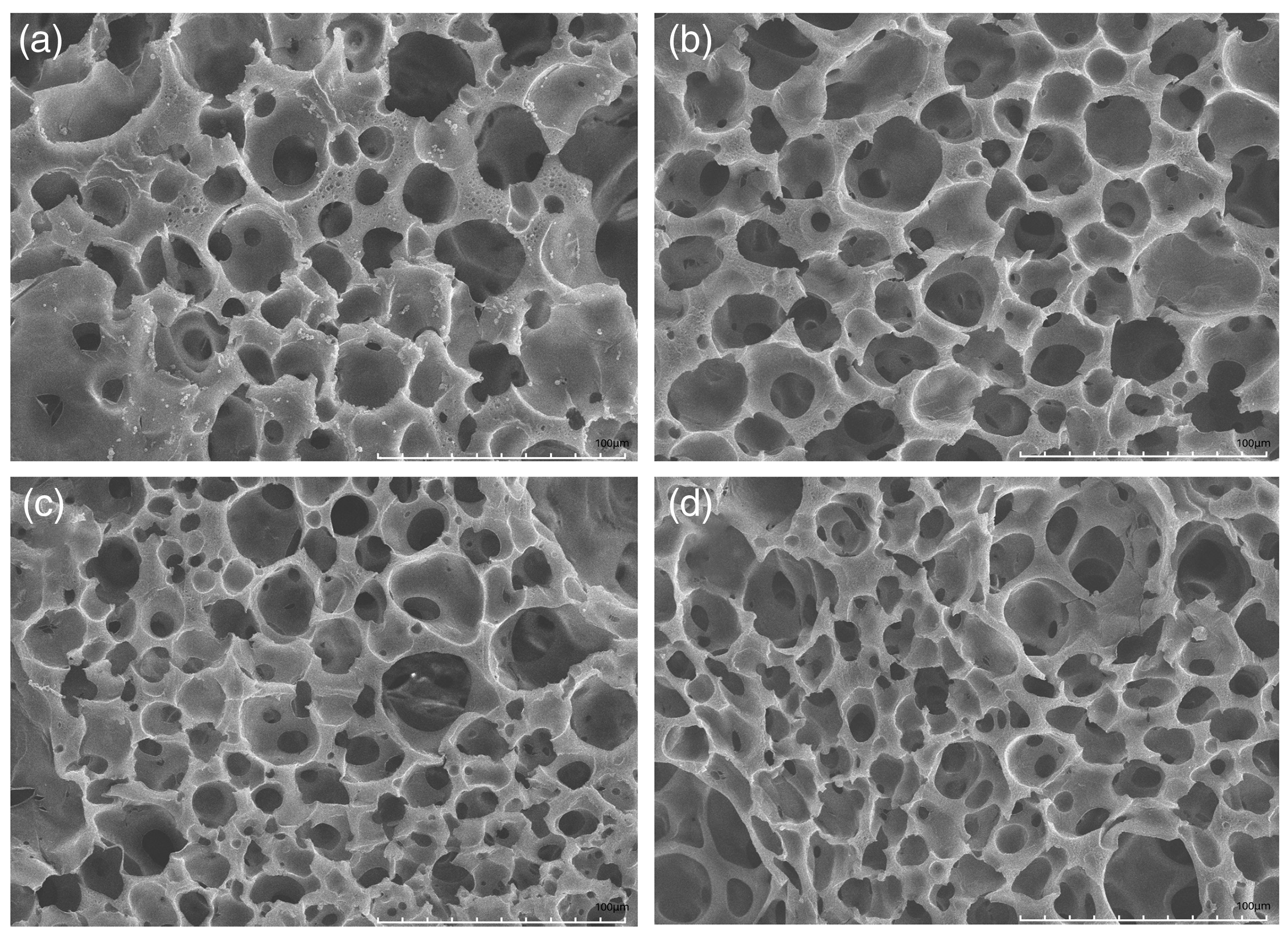
Microstructural analysis via scanning electron microscopy. (a) PLGA, (b) PLGA+DOX, (c) PLGA+IR780, and (d) PLGA+IR780+DOX. PLGA, poly(lactic-co-glycolic acid); DOX, doxorubicin.
Experimental release of DOX in water, artificial urine, and bladder tissue
The fluorescence intensity was proportional to the amount of DOX in water, confirming that the amount of DOX can be determined on the basis of fluorescence intensity (Figure 4). In water, DOX release was slow, reaching approximately 15% to 20% after 120 minutes. After 10 seconds of irradiation with near-infrared laser light, IR780 in the experimental group generated heat that triggered PLGA to undergo glass transition; this released approximately 20% to 25% of DOX during each instance of irradiation, and the amount of release reached approximately 65% to 70% after 120 minutes (Figure 5a). In artificial urine, DOX release was also slow. After irradiation with near-infrared laser light for 10 seconds, DOX was rapidly released through the mechanism observed in water (Figure 5b). In bladder tissue, DOX release from the PLGA +IR780 + DOX preparation was significantly greater than DOX release from the saline or DOX preparations (Figure 6).

Graphical depiction of relationship between optical density (OD) and adriamycin (ADR).
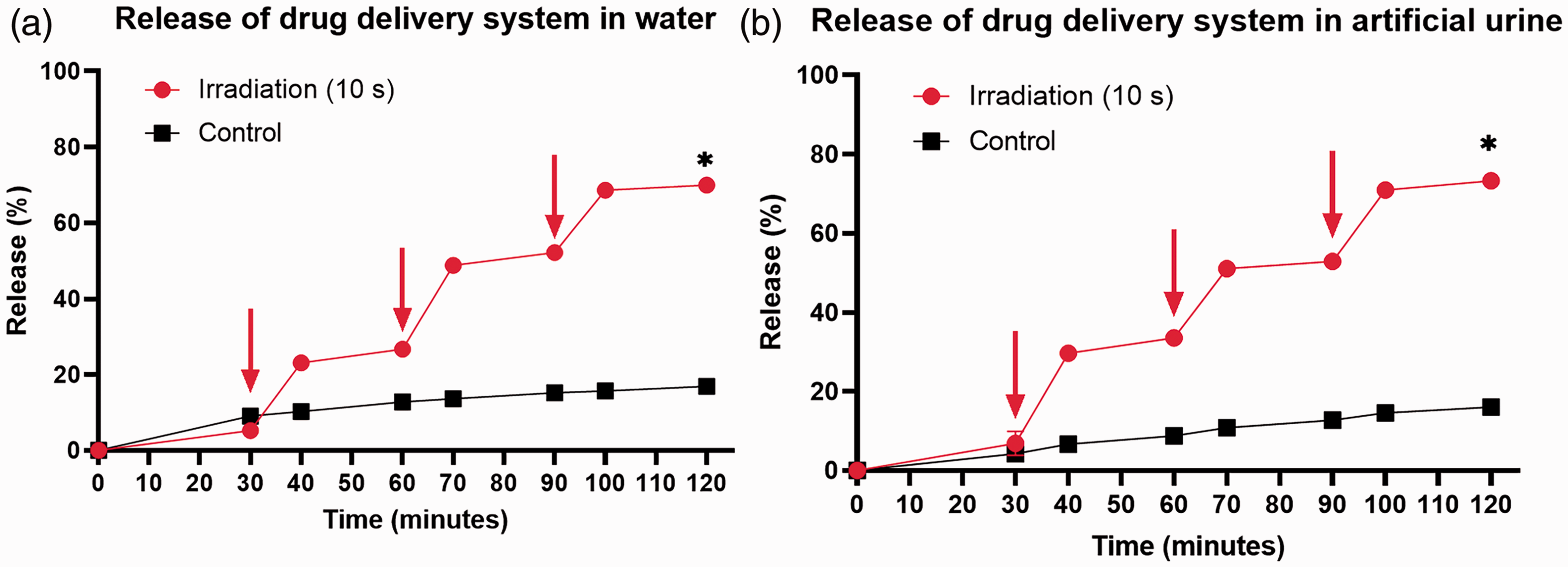
Experimental drug release in water (a) and artificial urine (b).

Experimental drug release in bladder tissue. (a) saline, (b) DOX, (c) PLGA+IR780+DOX.
Discussion
The gold standard for treatment of NMIBC is TURBT, followed by routine intravesical instillation chemotherapy involving urinary catheter insertion and removal. 1 There is a need to minimize the frequency of urinary catheter insertion and removal, which constitutes a physical and psychological burden for patients.
In recent years, various new materials have been generated through the integration of clinical medicine and materials science.23–25 Bladder perfusion defects are the focus of efforts to generate carriers with improved efficiency. Some researchers have used polymeric hydrogels (e.g., thermosensitive hydrogel composite systems) as drug carriers to increase the duration of drug retention in the bladder.26,27 However, these drug carrier systems can only extend the duration of release; they cannot allow controlled release. Moreover, nanoparticles are popular in treatments for bladder perfusion defects28,29 because they can increase local drug penetration and reduce side effects. Despite their capacity for local penetration, nanoparticles cannot be suspended and allow long-term pulsed release in the bladder; they also cannot reduce the frequency of bladder perfusion. Here, we designed a floating PLGA-based controlled-release drug delivery system, as an alternative to repeated use of a urinary catheter. We assessed the feasibility of our system through a series of experimental studies.
In pilot experiments, we explored the system composition and excitation conditions. We selected PLGA, which exhibited flotation and degradation properties, for dissolving in DMAC; this mixture was combined with IR780 and DOX to form a drug delivery system that could be excited by a near-infrared laser light for 10 s. As an alternative to the repeated insertion and removal of a urinary catheter, the drug delivery system was required to demonstrate robust flotation, structural, and controlled-release characteristics. In vitro and in vivo experiments revealed that all preparations and systems exhibited stable flotation in water and mouse bladder, indicating that the drug delivery system exhibited low density and poor solubility in water or urine. Thus, when placed in the human body, the drug release and dissolution of some substances lead to localization at the top of the bladder in various postures, preventing urethral obstruction that would hinder urination. Scanning electron microscopy revealed that each preparation had a honeycomb structure, confirming that the system is structurally stable and does not undergo structural changes because of drug release or partial drug dissolution; these characteristics suggest long-term stability in vivo. The system will not exhibit uncontrolled release related to structural variations after placement in the body.
To explore controlled-release characteristics, we measured the concentration of DOX based on its fluorescence intensity. Experimental release from the floating PLGA-based controlled-release drug delivery system showed, while irradiated by near-infrared laser light in water or artificial urine, PLGA underwent glass transition because of heat generated by IR780. The resulting pulsed release of DOX rapidly increased the local drug concentration, leading to successful controlled release after a single instillation.
Finally, we determined the efficacy of this system by measuring the amount of DOX in the bladder tissue, thereby confirming its feasibility. The results showed that our system can increase the amount of drug in bladder tissue and has the potential to improve efficacy by controlling the repeated release of chemotherapeutic drugs to exert effects equivalent to multiple instillations; this may eventually help to reduce the frequency of urinary catheter insertion and removal. Overall, we provide a novel strategy for improving intravesical instillation after NMIBC, which can reduce complications and remove physiological and psychological burdens from patients.
In conclusion, we generated a floating PLGA-based controlled-release drug delivery system with a honeycomb stabilized structure. It demonstrated controlled release when irradiated by a near-infrared laser, making it an ideal drug delivery system for intravesical instillation. However, this study was limited to in vitro and in vivo (animal-based) analyses; further research is required to confirm the clinical applicability of this system.
Footnotes
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Suzhou Youth Science and Technology Project of Rejuvenation Health through Science and Education (KJXW2019014).
Author contributions
KF, YZ, and JH made major contributions to the completion of the experiment and writing of the manuscript; TS, JN, and XL analyzed and interpreted the data; HZ designed and participated in the entire experiment. All authors read and approved the final manuscript.
