Abstract
This report presents the case of false-negative cerebral spinal fluid (CSF) cryptococcal antigen (CrAg) lateral flow assay (LFA) in a HIV-positive 25-year-old male. The patient presented with headache, nausea and vomiting for 5 days and syncope for 1 day. An initial CSF CrAg LFA test was negative, but a 1:4 dilution of the CSF was weakly positive and a 1:8 dilution was positive. A serum cryptococcal antigen test was weakly positive. Cultures of blood and CSF were all positive for Cryptococcus neoformans. The explanation for the false-negative CSF CrAg LFA test is that the antigen concentration was too high causing the postzone phenomenon.
Introduction
Cryptococcosis is a severe opportunistic fungal infection with high morbidity and mortality, caused predominantly by members of the Cryptococcus neoformans and Cryptococcus species complexes.1,2 The usual symptoms include headache, fever, nausea, neck stiffness, abnormal behaviour and photophobia. Early diagnosis and treatment are essential to ensure a favourable outcome. Cryptococcosis is definitively diagnosed from pathogen cultures onto Sabouraud dextrose agar. However, the culture results typically take several days due to its slow growth. 3 This case report describes a false-negative result from a cerebrospinal fluid (CSF) cryptococcal antigen (CrAg) lateral flow assay (LFA) in a patient with disseminated cryptococcal disease, whose blood culture and CSF culture tested positive for C. neoformans. The false-negative result occurred due to the postzone phenomenon. When the antigen concentration is too high, excess antibodies will prevent cross-linking and interfere with antibody–antigen precipitation, leading to false-negative results. 4 This case highlights the causes of false-negative CrAg LFA and suggests that CSF India ink staining and smear Gram staining should be combined with CrAg LFA and culture to increase the sensitivity to detect C. neoformans.
Case report
In April 2022, a 25-year-old male was admitted to the emergency department of Ningbo City First Hospital, Ningbo, Zhejiang Province, China for an intracranial infection. He had a history of HIV for more than 6 years and took irregular anti-AIDS medication. Five days earlier, he had developed a persistent headache associated with nausea and vomiting. One day previously, he was referred to Ninghai First Hospital, Ningbo, Zhejiang Province, China for syncope. Cranial computed tomography and magnetic resonance imaging showed no abnormalities.
The patient was subsequently admitted to the infection division of Ningbo City First Hospital, Ningbo, Zhejiang Province, China for further treatment. 4 On admission, the physical examination showed the following: body temperature, 37.0°C; blood pressure, 129/92 mmHg; heart rate, 60 beats/min; respiratory rate, 14 breaths/min. The laboratory results were as follows: white blood cells, 5.02 × 109/l; neutrophils, 91.2%; haemoglobin, 137 g/l; platelets, 118 × 109/l; C-reactive protein, 1.51 mg/l; procalcitonin, 0.09 ng/ml; urea, 2.63 mmol/l; creatinine, 49 µmol/l; aspartate aminotransferase, 14 U/l; alanine aminotransferase, 14 U/l; partial pressure of oxygen, 152 mmHg; partial pressure of carbon dioxide, 35 mmHg; and potassium ion, 2.7 mmol/l. He was administered 2 g ceftriaxone intravenously (i.v.) every 12 h as empiric treatment pending diagnostic testing.
The blood and CSF specimens were sent for microbiological examinations. The result of CSF CrAg LFA (IMMY, Norman, OK, USA) was negative, but serum cryptococcal antigen was weakly positive. On the presumption that the tic of limbs and obnubilation were due to intracranial infection and low potassium, the patient was started on empiric therapy with Depakine® (1.2 g sodium valproate) i.v. micropump. Supplemental oxygen and intravenous fluids were also initiated. Unfortunately, despite these therapies, the patient’s condition deteriorated over the next few days. The patient was transferred to the intensive care unit (ICU) and 0.2 g fluconazole i.v. once a day was empirically administrated to combat a fungal infection.
After 3 days of culturing, both the blood culture and CSF culture grew tiny white colonies in both Columbia blood and Sabouraud dextrose plates, which were identified as C. neoformans by matrix-assisted laser desorption-ionization time of flight mass spectrometry. The ICU physician in charge was informed by telephone immediately about the positive culture of C. neoformans. However, the patient's parents withdrew treatment and requested discharge after being informed of the risk of the related condition. By telephone follow-up, the patient was reported to have died at home the day after discharge.
Drug susceptibility demonstrated that the strain was susceptible to 5-fluorocytosine (4 µg/ml), amphotericin (0.5 µg/ml), fluconazole (1 µg/ml), itraconazole (0.125 µg/ml) and voriconazole (0.06 µg/ml).5–7 The CSF sample was retrieved from a 4°C refrigerator for re-examination, which found the following: a 1:2 CSF dilution in the CrAg LFA was negative; a 1:4 dilution was weakly positive; and 1:8 and 1:16 dilutions were positive. In addition, large numbers of Cryptococcus were found by microscopy using CSF India ink staining and Gram staining.
Ethics approval was obtained from the Ethics Committee of Ningbo First Hospital (no. 2022RS060). This study was performed in line with the principles of the Declaration of Helsinki. All of the patient’s details were de-identified. The patient provided verbal informed consent for treatment and for publication of this case report. The reporting of this case report conforms to CARE guidelines. 8
Discussion
Cryptococcosis is definitively diagnosed from pathogen cultures in Sabouraud dextrose agar. However, C. neoformans may take several days to culture because of its slow growth. 9 Cryptococcal meningitis can be diagnosed by microscopy of fresh CSF using India ink staining, which has 100% specificity but only 50% sensitivity depending on cell titres and capsule production. 10 In the early stages, India ink staining results may appear normal. The CrAg, a component of the glucuronoxylomannan polysaccharide capsule, which is soluble and detectable in serum or body fluids as a marker of infection, is used for timely and sensitive diagnoses of cryptococcosis.11,12 Therefore, CrAg testing is useful in the diagnosis of both cryptococcal antigenaemia (disseminated disease before meningitis onset) and cryptococcal meningitis.
In this current case, the C. neoformans blood culture and the positive result for the CSF culture revealed that the patient had disseminated cryptococcosis. An initial CSF CrAg LFA test was negative. The CSF CrAg LFA test may have been negative owing to the postzone effect. Therefore, the CSF was diluted and the tests were repeated to resolve the discrepancy. After diluting the CSF, the result was weakly positive. According to the ‘postzone phenomenon’ described in the CrAg LFA instructions, particularly high concentrations (>0.140 mg/ml) of cryptococcal antigens can produce weakly positive results and individual samples can produce negative results.
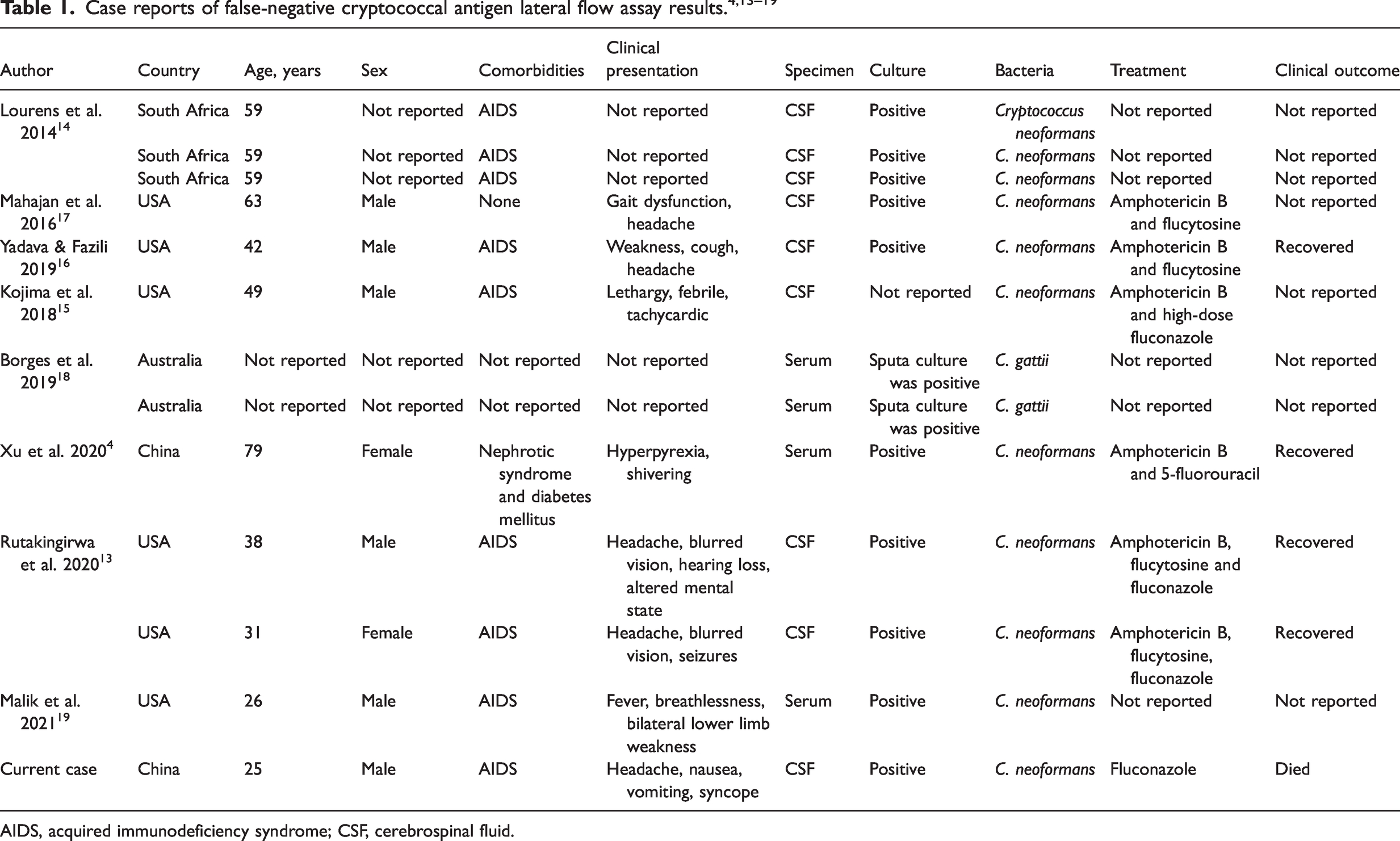
In 2020, a report described three patients with HIV infection, who all tested positive for either reference blood or serum cryptococcal antigen test. 13 CSF CrAg was negative, but two cases turned positive after dilution, so the false-negative results appeared to be caused by the postzone phenomenon. 13 Cryptococcal culture was positive. 13 Reports of false-negative results due to the postzone phenomenon are uncommon. Thirteen cases of false-negative CrAg LFAs have been described in nine studies since 2014 (Table 1), including the current case.4,13–19 These were sporadic reports from four continents; United States, South Africa, Australia and China.4,13–19 The youngest patient was 25 years old and five patients were older than 50 years.4,13–19
AIDS, acquired immunodeficiency syndrome; CSF, cerebrospinal fluid.
Of the 13 cases of false-negative CrAg LFAs, nine had CSF samples analysed; of these, eight were HIV-infected patients (Table 1).4,13–19 However, only the CSF specimens were tested. A retrospective study of patients with culture-confirmed cryptococcal meningitis showed that the CrAg LFA had 100% sensitivity in the serum. 20 Testing serum for CrAg is recommended, especially for patients who cannot undergo an immediate lumbar puncture or when lumbar puncture is contraindicated. The underlying conditions in these 13 patients included nephrotic syndrome, diabetes mellitus and AIDS.4,13–19 In terms of prognosis, one patient died (current case) and four patients recovered.4,13,16 For the remaining patients, there was no description of their outcomes. However, a sample size of 13 patients is too small to perform statistical analysis of immunity and prognosis. Of the 13 patients, 11 were diagnosed as C. neoformans s.l., while the others were diagnosed with C. gattii s.l.
Several reasons for false-negative cryptococcal antigen tests have been proposed. For example, a low level of cryptococcal antigen production by the strain can produce a false-negative result. 21 In contrast, a very high concentration can also produce false-negative findings because of the presence of postzone phenomenon. 22 Furthermore, the presence of non-specific proteins in the CSF may mask the cryptococcal antigen. In addition, the same commercial preparation may have its own rate of false-negativity as the performance varies in different evaluations. 23 Another unusual reason for false-negative results is due to small-colony variants of C. neoformans. In 2006, a report described a patient with a cystopleural shunt. 23 The CSF isolates not only yielded smaller colonies on Sabouraud dextrose agar, but also on Pagano Levin agar and Malassezia agar, all having colony sizes <1 mm after 3 days of incubation. 23 In comparison, the control strain’s colony size was >3 mm. 23 On corn meal agar supplemented with caffeic acid, isolates demonstrated earlier production of brown pigment than the control strain and another non-C. neoformans. 23 Small-colony variants have been demonstrated in various bacteria, including Staphylococcus aureus, coagulase-negative Staphylococci, Pseudomonas aeruginosa and Enterobacteriaceae. 23
The postzone phenomenon is defined as a false-negative test resulting from a high antigen titre, which interferes with the formation of the antigen–antibody complex that is necessary to visualize a positive test. 16 The postzone phenomenon in the CSF of a patient with cryptococcal meningitis was first described in a latex agglutination test in 1980; 22 and after that, several cases were reported.13,16,21,24 Only a few cases of false-negative CrAg LFA in CSF due to the postzone phenomenon have been reported.13,16 These cases, which likely reflect inherent limitations in traditional antigen-capture assays and diagnostic uncertainty in cases of assumed early cryptococcal meningitis, rather than poor diagnostic performance of the CrAg LFA, are relatively common in clinical practice.13,21 Symptomatic meningitis in the context of a positive serum CrAg but negative CSF CrAg should prompt enhanced diagnostic testing to rule out the postzone effect, such as repeating CrAg on diluted CSF. 13
In conclusion, a negative CrAg LFA result does not exclude the diagnosis of cryptococcal infection. In evaluating patients with symptoms consistent with cryptococcal infection and negative CrAg LFA results, retesting the CSF with alternative LFA kits may resolve the diagnostic dilemma and avoid treatment delay. Evaluation of currently available LFA kits, as well as evaluation of new technologies for the detection of cryptococcal antigen, include careful evaluation of samples with suspected low antigen concentrations. 25 When the microbiology laboratory know that the patient is HIV positive and presents with headache and vomiting, but the CrAg LFA is negative, cryptococcal infection should be highly suspected. False-negative results due to the postzone phenomenon can be excluded by performing serial dilutions of the CSF samples. In addition, laboratories should maintain active communication with the physicians in charge of the patient.
Footnotes
Author contributions
All of the authors contributed to the conception and design of the study. Data collection was performed by Lu Xu and Yi-Jun Mo. Data analysis was performed by Xing-Bei Weng. The first draft of the manuscript was written by Lu Xu. All authors read and approved the final manuscript.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
The authors disclose receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Ningbo Natural Science Foundation (no. 2019A610381) and Ningbo Public Welfare Foundation (no. 2019C50087).