Abstract
Primary pulmonary venous malformation is rare and usually presents as single or multiple round masses or nodules. Here, we present the first report of a case of venous malformation presenting as Mauritia arabica-like bronchial wall thickness that was initially misdiagnosed as bronchiectasis. A Chinese man in his late 20s presented complaining of hemoptysis for 10 days. Computed tomography demonstrated bronchiectasis and M. arabica-like bronchial wall thickening in the left lower lobe. He was unresponsive to medical treatment for bronchiectasis and underwent thoracoscopic left lower lobectomy. Histopathological examination revealed venous malformation around the bronchial walls with no bronchiectasis. Venous malformation should be considered in the differential diagnosis of bronchiectasis, especially in patients with the following triad of signs: no response to antibiotics, M. arabica-like bronchial wall thickness, and normal accompanying arteries.
Keywords
Introduction
According to the International Society for the Study of Vascular Anomalies (ISSVA) classification, vascular anomalies are categorized as vascular tumors and vascular malformations. 1 Vascular tumors include proliferative changes in endothelial cells and vascular malformations are structural vascular abnormalities without proliferation of endothelial cells. Venous malformations include common venous malformation and specific types, such as familial venous malformation, cutaneo-mucosal venous malformation, blue rubber bleb nevus syndrome, glomuvenous malformation, cerebral cavernous malformation, familial intraosseous vascular malformation, verrucous venous malformation, and other unclassified. 1 Venous hemangioma was the previous name for venous malformation.
The estimated prevalence of venous malformations is 0.8% to 1% in the general population. 2 Venous malformations are mostly associated with congenital dysplasia. They can develop in various organs, especially the skin and brain. 2 Primary pulmonary venous malformations are rare and often present as round masses or nodules in the pulmonary parenchyma or pleura. 3 Patients may be asymptomatic or present with recurrent coughing and hemoptysis. 2 Here, we report the first known case of pulmonary venous malformation that presented as hemoptysis with Mauritia arabica-like bronchial wall thickening and was initially misdiagnosed as bronchiectasis.
Case Report
The reporting of this study conforms to the CARE guidelines. 4 A Chinese man in his late 20s came to our hospital on 10 July 2021 complaining of a 10-day history of hemoptysis. The patient had experienced hemoptysis two to three times a day with a small amount of fresh blood, cough, production of white sputum, and mild chest tightness. He did not have fever or night sweats. He had experienced a 5-kg loss of body weight during the previous month. He had never previously had a health check-up. He had no history of hemoptysis or comorbidities and was a non-smoker. There was no family history of hemoptysis, malignancy, or other condition of note. Reduced breath sounds were detected on auscultation of the left lower lung. No other specific signs were observed during physical examination.
His white blood cell count, C-reactive protein level, erythrocyte sedimentation rate, and procalcitonin level were within normal limits. Sputum smear and culture were negative for bacteria and fungi. Renal and liver function tests were normal. Antinuclear antibodies, anti-dsDNA antibody, anti-neutrophil cytoplasmic antibodies, antiphospholipid antibodies, and anti-cyclic citrullinated peptide antibodies were all negative. A computed tomography (CT) scan of the chest revealed cylindrical bronchial dilation, M. arabica-like thickened bronchial walls, multiple patchy shadows and nodules, and a large patchy ground-glass shadow in the left lower lobe, indicating bronchiectasis and alveolar hemorrhage (Figure 1). Enhanced CT of the chest showed partial enhancement of the bronchial walls (Figure 2).

A computed tomography image of the thorax (axial view) demonstrating bronchiectasis, alveolar hemorrhage, and Mauritia arabica-like thickening of the bronchial walls (green arrows) in the left lower lobe.
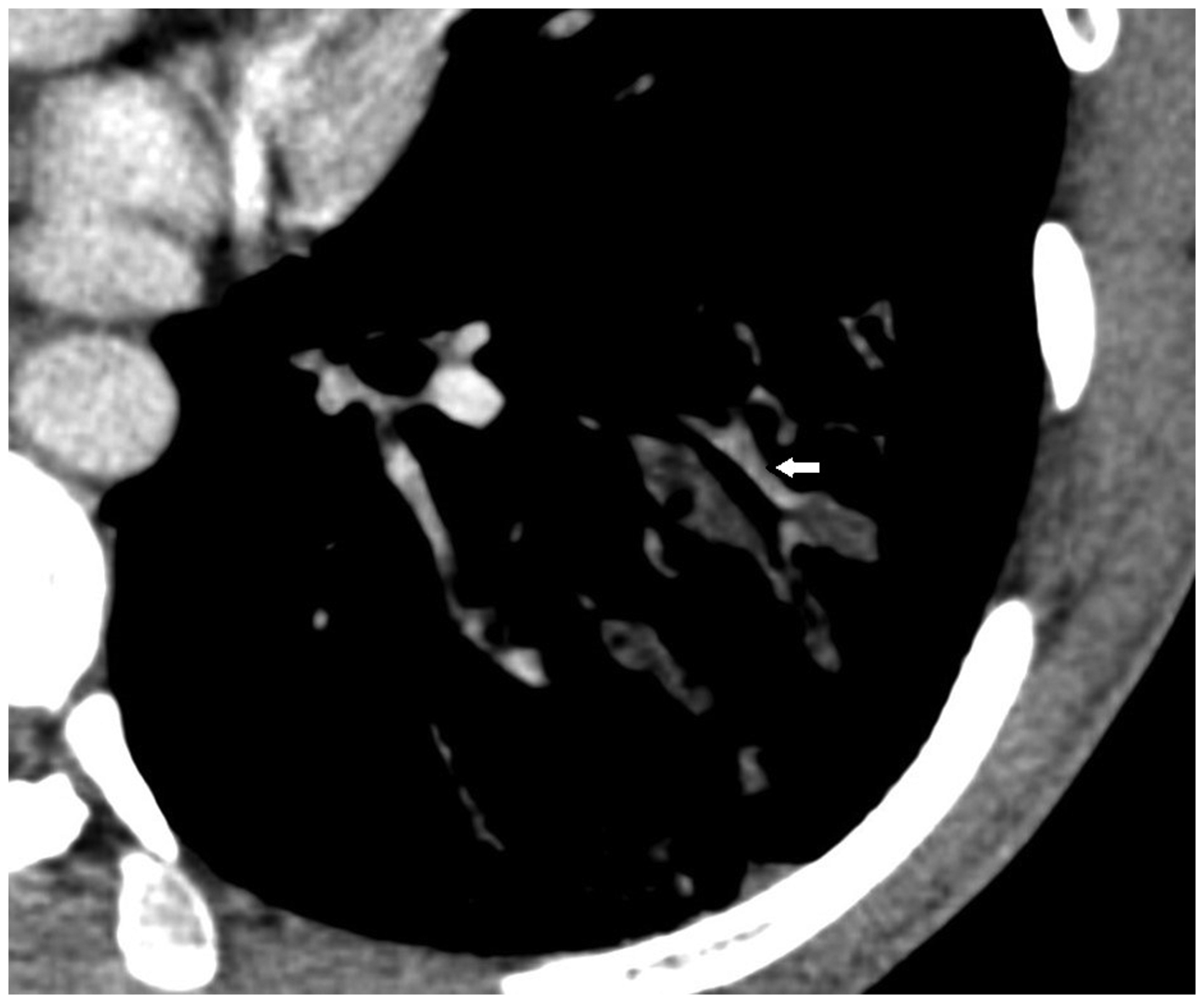
Enhanced computed tomography scan of the thorax demonstrating partial enhancement of the bronchial walls (white arrow).
Based on the diagnosis of bronchiectasis and the antibiotic treatment the patient had received at a previous hospital, piperacillin and tazobactam were prescribed. Pituitrin was administered as hemostatic therapy. However, the hemoptysis was not relieved, and levofloxacin was added to cover an atypical pathogen 4 days later. A second CT scan of the chest performed after 10 days of treatment showed no resolution of the lesions. Bronchoscopy showed persistent bloody discharge in the posterior segment of the left lower lobe. Endobronchial ultrasound detected low echo lesions with a clear high echo boundary at the medial-anterior basal segment, lateral basal segment, and posterior basal segment (Figure 3). The antibiotics were changed to imipenem, cilastatin, and doxycycline. However, after 7 days of escalating antibiotic therapy, the hemoptysis was still not relieved. A transbronchial biopsy and brushing of the left lower lobe demonstrated infiltration of neutrophils and lymphocytes. Bronchoalveolar lavage fluid smears and cultures were negative for bacteria and fungi.

Findings on bronchoscopy. (a) The carina. (b) The left main bronchus contains bloody discharge. (c) The left lower lobe bronchus also contains bloody discharge. (d) The posterior segment in the left lower lobe shows persistent hemoptysis. (e) The left basal segment and (f) Endobronchial ultrasound view of the lateral basal segment showing a low echo lesion with a clear high echo boundary.
Antibiotic therapy was withdrawn, and a multidisciplinary consultation was held with a respiratory physician, vascular interventional physician, and a thoracic surgeon. Considering the small amount of hemoptysis, it was hard to identify the culprit vessel during interventional angiography. Therefore, the patient underwent thoracoscopic lobectomy of the left lower lobe after providing written informed consent. Histopathological examination revealed remote hemorrhage of the bronchial wall and many irregular cyst-like structures filled with red blood cells around the bronchial walls. The inner walls of the cysts contained flattened epithelium (Figure 4). The irregular cyst-like structures were negatively immunostaining with CD34 and positively immunostaining with D2-40, which suggested venous malformations. The patient recovered and was discharged after surgery. At the telephone follow-up 1 year later, the patient reported that he had recovered well, had no hemoptysis, and that his weight had returned to the level before the onset of his symptoms.

Histological view of the venous malformation showing remote hemorrhage of the bronchial wall and large numbers of irregular cyst-like structures filled with red blood cells around the bronchial walls. The inner walls of the cysts have a flattened epithelium. There is no obvious manifestation of bronchiectasis (hematoxylin-eosin staining; magnification, 12.6×).
Discussion
We searched the PubMed database using the terms (hemangioma OR venous malformation) AND (lung OR pulmonary) without limitation on publication time. To the best of our knowledge, this is the first report of multiple venous malformations presenting initially as M arabica-like bronchial wall thickening and mimicking bronchiectasis.
Pulmonary venous malformations can be confused with pulmonary cavernous hemangiomas. Pulmonary cavernous hemangioma refers to closely situated tangles of vessels or widely dilated vascular channels that have focal connective tissue in their walls. This type of vessel malformation does not have the characteristics of an artery. 5 According to the ISSVA classification, hemangioma is categorized as a vascular tumor and “pulmonary cavernous hemangioma” as a type of pulmonary venous malformation. 1 Although the term pulmonary cavernous hemangioma is no longer used, the pulmonary venous malformation in our case had some of the characteristics of pulmonary cavernous hemangioma.
When they occur in the lung, primary pulmonary venous malformations usually present as single or multiple masses or nodules,6,7 but there is marked variation in symptoms. Patients may be asymptomatic or may present with coughing, hemoptysis, chest pain, pleural effusion, and/or dyspnea. 3 There could be a single lesion in the lung or multiple lesions accompanied by extrapulmonary lesions in the mediastinum, pleura, chest wall, pericardium, thymus, or liver. 3 Primary pulmonary venous malformations may be misdiagnosed as carcinoma or metastases. 3
In this case, the dilated venous spaces around the bronchial walls and hemorrhage within the bronchial walls mimicked thickening and enlargement of the bronchus and are the typical signs of bronchiectasis on CT. However, there were some characteristics suggesting a diagnosis other than bronchiectasis. First, the patient was unresponsive to treatment for bronchiectasis. Second, according to the CT scan, the bronchial walls showed irregular pointed oval-like thickenings similar to the ventral side of the M. arabica (Linnaeus) shell, with partial enhancement. M. arabica is a mollusk with a lengthwise opening in the center of the ventral side of its shell. Both sides of this opening are lined with saw-tooth projections. The bronchial wall thickness seen on the CT images was very similar to the view of the ventral side of the M. arabica shell. Third, compared with the normal side of the lung, the arteries along the bronchial walls were not enlarged or twisted as in bronchiectasis.
The venous malformations in this case were located around the bronchus along the bronchial vascular bundle. The lesions were partially enhancing and drained into the pulmonary veins without obstruction of the left inferior pulmonary vein. Diseases that include thickening of the interstitium along the bronchial vascular bundle, such as lymphangitis carcinomatosa, 8 lymphoma, 9 and sarcoidosis, 10 should be included in the list of differential diagnoses. Drainage into the pulmonary veins may be the essential feature that secures the diagnosis. However, it may be difficult to determine the relationship between the lesions and the pulmonary vein initially. In this case, we became aware of the drainage route after the pathological diagnosis was made. Pulmonary venous malformation should be distinguished from pulmonary aneurysm. While a pulmonary venous malformation drains into the pulmonary vein, a pulmonary aneurysm is connected to the pulmonary artery.
Our experience in this case suggests that a venous malformation should be considered in the differential diagnosis of bronchiectasis, especially in patients with the following triad: unresponsiveness to antibiotics, M. arabica-like bronchial wall thickness, and normal accompanying arteries. However, further cases need to be accumulated to confirm our findings.
Research Data
Research Data for Venous malformation presenting as Mauritia arabica-like bronchial wall thickness: a case report
Research Data for Venous malformation presenting as Mauritia arabica-like bronchial wall thickness: a case report by Miaochan Lao, Ping Gao, Yanhui Liu, Lixu Yan and Xinglin Gao in Journal of International Medical Research
Footnotes
Acknowledgements
The authors are grateful to Hui Liu from the Department of Radiology, Guangdong Provincial People's Hospital, Guangdong Academy of Medical Sciences for radiological interpretation and to Pingping Chen from the Department of Pulmonary and Critical Care Medicine, Guangdong Provincial People's Hospital, Guangdong Academy of Medical Sciences for interpretation of the endobronchial ultrasound images.
Author contributions
Miaochan Lao and Ping Gao made a substantial contribution to writing the manuscript. Yanhui Liu and Lixu Yan interpreted the pathological data. Xinglin Gao analyzed the patient data. All authors have read and approved the final manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
This case report was approved by the Guangdong Provincial People’s Hospital Research Ethics Committee (approval number: KY-Q-2022-399-01). Verbal informed consent was obtained from the patient for publication of this article. All patient details have been de-identified.
Funding
This work was supported by the Guangzhou Science and Technology Planning Project [grant number 202102080611] and the Medical Scientific Research Foundation of Guangdong Province [grant number B2022157].
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
