Abstract
The potential utility of nifekalant, a new Class III antiarrhythmic drug, to offer long-term protection against ventricular arrhythmia has been investigated in this case report. A 44-year-old male patient with dilated cardiomyopathy complicated with heart failure and persistent ventricular tachycardia was treated with nifekalant. The patient was treated with nifekalant for 31 days, which effectively terminated ventricular tachycardia and maintained sinus rhythm, with no clinical adverse reactions. After heart transplantation, postoperative follow-up showed good cardiac function and no arrhythmia. On the basis of nifekalant’s working mechanism, there is a good chance that it can cure ventricular arrhythmia on a long-term basis.
Keywords
Introduction
On 1 July 2019, a patient with dilated cardiomyopathy complicated with heart failure and persistent ventricular tachycardia presented at our hospital. On the basis of his symptoms, oral administration of metoprolol 12.5 mg twice daily was reduced to 6.25 mg twice daily due to low blood pressure (80/45 mmHg) and cool hands and feet, which was difficult to tolerate. The patient’s blood pressure remained low, so the medication was stopped. Dopamine and norepinephrine were added to maintain blood pressure. An intravenous injection of amiodarone 300 mg was administered at a rate of 1000 μg/minute, and the QT interval (QTc) was 525 ms. No reduction of ventricular arrhythmias was observed under electrocardiogram (ECG) monitoring. Ventricular arrhythmias did not decrease under ECG monitoring after intravenous injection of 100 mg of lidocaine at a rate of 100 mg/hour. Therefore, commonly used antiarrhythmic drugs such as amiodarone were clinically ineffective. 1 Nifekalant, a new Class III antiarrhythmic drug, can terminate ventricular tachycardia, but there is insufficient evidence and clinical experience to show the safety and efficacy of long-term nifekalant use.
Case report
A 44-year-old male patient first presented at our hospital on 7 April 2019 with exercise-induced chest tightness, shortness of breath, and palpitations for 2 months. The patient had no family medical history of heart disease and was in good psychological condition. Echocardiography revealed significant dilation of the left ventricle and a left ventricular ejection fraction of 34% (Figure 1a). A dynamic ECG showed 14,816 single ventricular premature beats (SVPB) within 24 hours using a 24-hour Holter monitor. Among these beats, 507 were paired, and ventricular tachycardia occurred 48 times, with ventricular arrhythmia accounting for 16.8% of the total heartbeats (Figure 2). Ventricular premature beats and ventricular tachycardia have several forms. Cardiac magnetic resonance revealed myocardial fibrosis from the basal segment to the middle segment of the anterior septum and anterior wall (Figure 3). Additionally, coronary angiography was performed, and coronary heart disease was excluded (Figure 4). Subsequent genetic testing revealed two rare gene mutations that were consistent with dilated cardiomyopathy (TNNI3K: c.62G>A, VPS13A: c.7323dupT, and TNNI3K: c.62G>A). This is such a rare occurrence that it has never been reported before. The patient was diagnosed with dilated cardiomyopathy and ventricular arrhythmia (including recurrent SVPB, burst tachycardia, and stage-C heart failure ). After medical treatment, the clinical symptoms were relieved.
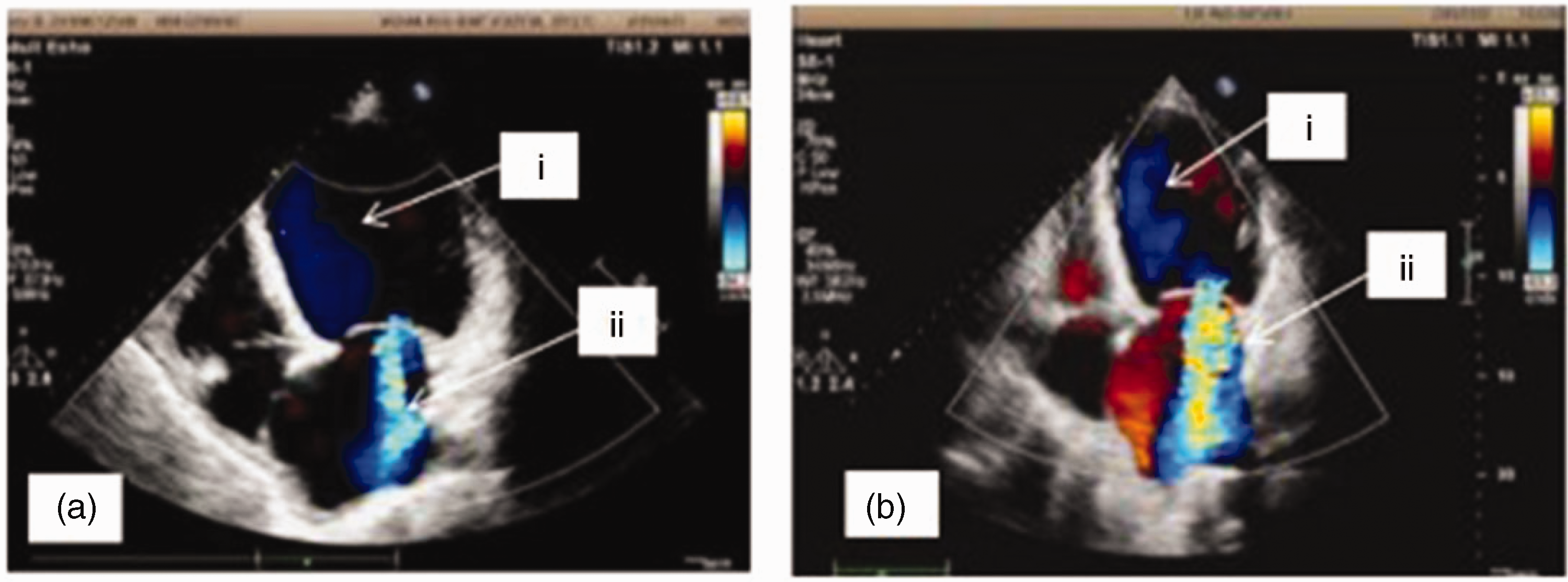
Echocardiography at two time points. (a) i. LVEDD and ESD were 74 and 62 mm, respectively; ii. Moderately severe central mitral regurgitation. (b) i. LVEDD and ESD were 78 and 66 mm, respectively; ii. Severe central mitral regurgitation.

Twelve-lead ECG image.
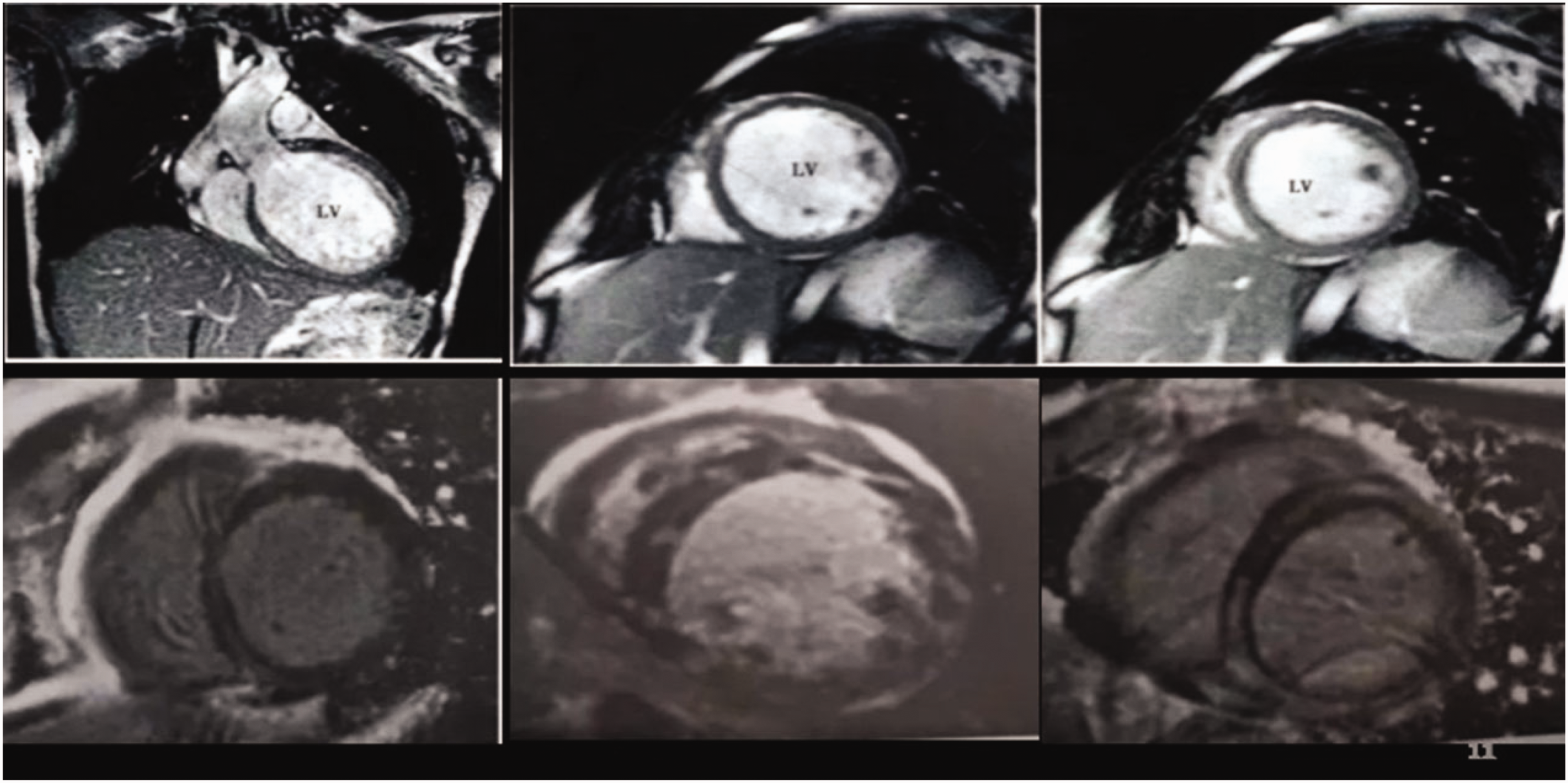
Cardiac MRI image.

Angiography image. Frequent pleomorphic ventricular tachycardia, and coronary angiography did not reveal coronary stenosis.
He was hospitalized again 2.5 months later from 1 July to 8 August 2019 with recurrent heart failure due to dilated cardiomyopathy and ventricular arrhythmia. Laboratory tests showed N-terminal pro-B-type natriuretic peptide levels of 4365.00 pg/mL (reference range, 0–125 pg/mL) and cTIn levels of 0.032 ng/mL (reference range, 0–0.03 ng/mL). Considering his safety, radiofrequency ablation or implantable cardioverter defibrillator implantation was recommended, but he refused these suggestions. Cardiopulmonary exercise testing showed a maximum peak oxygen consumption of 9.5 mL · kg−1 min−1. He had definite indications for cardiac transplantation after extensive evaluation. While waiting for heart transplantation, he had recurrent electrical (or arrhythmic) storms (Figure 5). Drugs such as β-blockers, lidocaine, and amiodarone tablets were ineffective. Ventricular tachycardia recurred and was classified as New York Heart Association class IV. Echocardiography was performed again. The arrhythmia was significantly improved after treatment with nifekalant, but reexamination of the cardiac ultrasound showed no improvement in the left ventricular ejection fraction, which remained at approximately 22% (Figure 1b). Twenty-four-hour Holter monitoring revealed 76 instances of paroxysmal atrial fibrillation and non-sustained ventricular tachycardia. Therefore, nifekalant was used to preserve tachycardia and sinus rhythm. During treatment, nifekalant was replaced with amiodarone to further prevent arrhythmia because of the low cost of nifekalant and the specified duration in the surgical instructions. Thus, nifekalant was used, and the other antiarrhythmic drugs were discontinued. A normal nifekalant dose (0.3 mg/kg) was injected intravenously, followed by continuous intravenous administration at a rate of 0.2–0.4 mg/kg/hour. The patient was administered oral furosemide (20 mg once daily) and spironolactone (20 mg twice daily) to maintain his body weight, rivaroxaban (15 mg once daily) as anticoagulant therapy, and potassium chloride to maintain serum potassium levels at 5.0–5.5 mmol/L. No drugs interacted with nifekalant. However, pulseless ventricular tachycardia was observed in the patient again 29 hours after stopping nifekalant, and thus, it was used again for cardioversion of ventricular tachycardia and to maintain serum potassium levels at 5.0–5.0 mmol/L and serum magnesium levels at 1.0–1.2 mmol/L and to correct QTc to 445–506 ms. The ventricular arrhythmia was effectively controlled, and dynamic electrocardiogram results revealed that the ventricular arrhythmia was significantly relieved, without the occurrence of torsade de pointes ventricular tachycardia. After nifekalant treatment for 31 days, a heart donor was found and the patient successfully underwent the cardiac transplantation procedure.
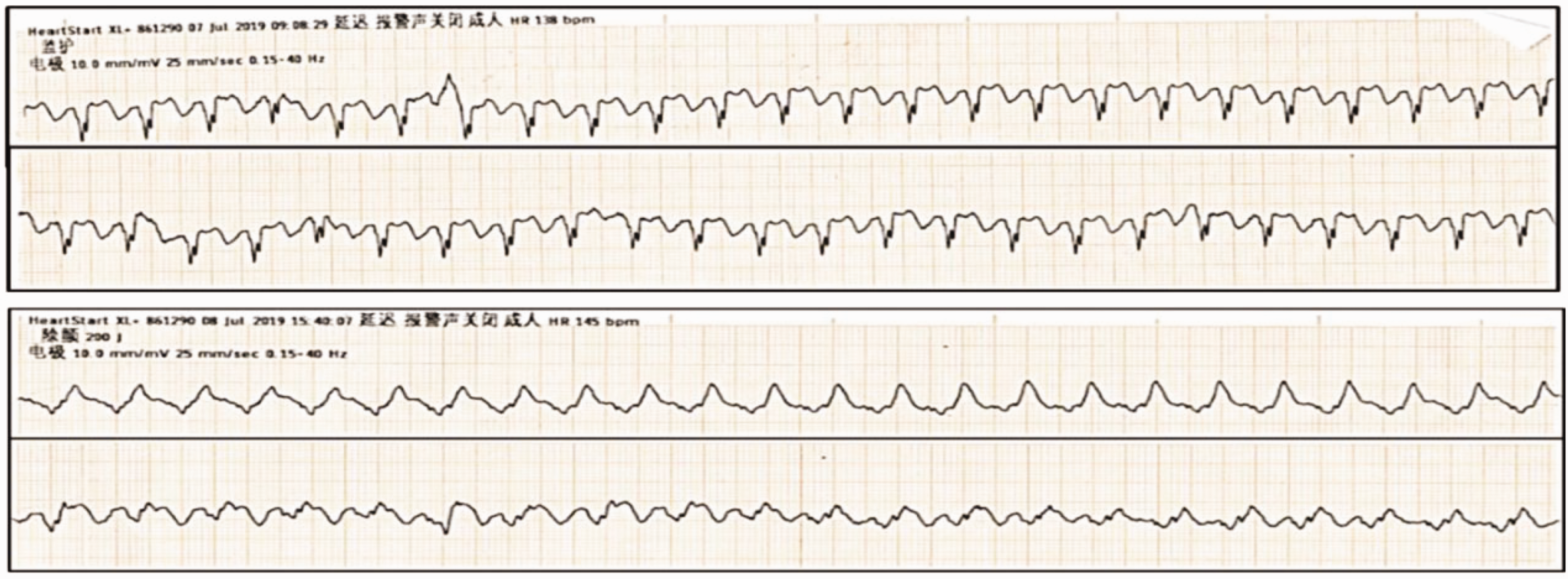
Recurrent ventricular arrhythmia during treatment.
The reporting of this study conforms to CARE guidelines. 2 The patient provided written informed consent to publish his case.
Discussion
A patient with dilated cardiomyopathy and left ventricular enlargement had recurrent SVPB and ventricular tachycardia. Ventricular tachycardia such as ventricular fibrillation was found to be the main cause of cardiac sudden death in dilated cardiomyopathy patients except in those with heart failure. 3 For these patients, common antiarrhythmic drugs have usually been an ineffective treatment, but nifekalant is a new and highly recommended Class III antiarrhythmic drug that blocks cardiac delayed rectifier potassium currents without affecting the inward sodium and calcium currents or β-adrenergic activity. This drug is usually used for treating ventricular arrhythmia, and it has achieved significant positive effects. In addition to effective termination, nifekalant was highly effective in preventing arrhythmia recurrence during long-term maintenance.4,5 Patients with clinical heart failure often have ventricular arrhythmia, but sudden death is much more difficult to prevent and treat than heart failure. Thus, for such patients, clinical treatment requires an effective and highly controllable drug that does not reduce myocardial contractility or blood vessel dilation to lower blood pressure. Commonly used antiarrhythmic drugs include lidocaine and β-blockers have some negative inotropic effects, whereas nifekalant does not show these effects. 6 Amiodarone showed no obvious effects on this patient, but his ventricular arrhythmia was effectively controlled after taking nifekalant. The patient’s ventricular arrhythmia also returned after discontinuing nifekalant, demonstrating its effectiveness. Nifekalant is considered for long-term use because as a pure potassium channel blocker, 7 it could block the delayed rectifier potassium current quickly without blocking sodium and calcium channels. Furthermore, it prolongs the atrial and ventricular effective refractory period. Nifekalant was reported to rapidly extend the QT period and slow the heart rate. 8 However, its heart conduction effect is not obvious. Nifekalant does not block adrenergic β receptors, reduce the myocardial contraction force, dilate blood vessels, or reduce vascular resistance. It has little effect on blood pressure and is suitable for the treatment of cardiogenic shock combined with ventricular arrhythmia. The novelty of our study lies in the duration of nifekalant use to treat ventricular arrhythmias with dilated cardiomyopathy, which is 31 days. This treatment duration is unprecedented because the prescribing information indicates that the maximum nifekalant treatment duration is 14 days, and the longest period of use reported in the literature is 19 days. Additionally, the drug’s safety was shown because torsade de pointes ventricular tachycardia did not occur during medication use in our patient.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221133704 - Supplemental material for Long-term nifekalant use in a patient with dilated cardiomyopathy and recurrent ventricular tachycardia
Supplemental material, sj-pdf-1-imr-10.1177_03000605221133704 for Long-term nifekalant use in a patient with dilated cardiomyopathy and recurrent ventricular tachycardia by Hongyan Xiao, Qitong Chen, Liang Tao in Journal of International Medical Research
Footnotes
Author contributions
Drs Xiao, Chen, and Tao contributed equally to the article. Hongyan Xiao conceived and coordinated the study, designed, performed, and analyzed the experiments, and wrote the paper. Liang Tao and Hongyan Xiao revised the paper. Qitong Chen performed data collection and data analysis. All authors reviewed the results and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethics statement
The patient provided consent for treatment. Ethical approval was not required because this manuscript only reports a case. The patient provided written informed consent for the use of his data for scientific purposes.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Wuhan Health and Family Planning Commission of Wuhan Municipality Scientific Research Project [grant number: WX13C48].
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
