Abstract
Objective
The long non-coding RNA Growth-arrest-specific transcript 5 (GAS5) has been extensively linked with the ability of cancer cells to resist chemotherapeutic interventions. This prospective study aimed to investigate the role of GAS5 in oral squamous cell carcinoma (OSCC), which has been poorly characterized to date.
Methods
GAS5 and miR-196a expression levels were detected by quantitative real-time PCR analysis. Cisplatin (DDP) sensitivity and apoptosis levels were determined using Cell Counting Kit 8 and flow cytometry, respectively. Luciferase reporter and RNA immunoprecipitation assays were performed to confirm target miRNAs of GAS5.
Results
We found that GAS5 was expressed at low levels in DDP-resistant OSCC cell lines and tissues, and that GAS5 levels were intricately linked to the survival rates of OSCC patients. GAS5 overexpression led to the recovery of DDP sensitivity in CAL27/DDP cells. Additionally, in both DDP-resistant and -sensitive lines, GAS5 showed a cytoplasmic distribution and downregulated miR-196a in OSCC tissues. Exogenous transfection of miR-196a alleviated the effects of GAS5 on DDP sensitivity, confirming this as the mechanism of chemoresistance.
Conclusions
These findings may provide new targets for the treatment of chemotherapy-resistant OSCC.
Keywords
Introduction
Oral squamous cell carcinoma (OSCC) is an aggressive cancer of the digestive system that exhibits a high prevalence and morbidity.1,2 Despite the advancements in OSCC treatments, only modest improvements in patient outcomes have been made; thus new treatments for this malignancy are urgently required. The current arsenal for OSCC of drugs has suffered from chemoresistance and tumor relapse, both of which lead to a loss of treatment efficacy.3,4 The molecular mechanisms and prognostic factors that govern chemoresistance in OSCC remain poorly characterized, and further studies are required in this area.
RNAs that lack coding function (non-coding RNAs) regulate gene expression in a range of cell types. Such RNAs that exceed 200 nucleotides in length are termed long non-coding (lncRNAs). Dysregulation of lncRNAs in cancer cells has been implicated in tumor growth and formation.5,6 Depending on the tumor-type, lncRNAs can promote cancer formation or can act as inhibitors of metastatic processes. Many lncRNAs contribute to tumor prognosis, cancer diagnosis, 7 and drug resistance.8–10 However, the contribution of lncRNAs to OSCC formation has remained relatively poorly defined.
One such exemplar of a lncRNA that is linked to cancer formation is Growth-arrest-specific transcript 5 (GAS5), 11 which is a lncRNA that is suppressed in several cancer types in which it drives tumorigenesis including lung, gastric, and ovarian cancers.12–14 Low levels of GAS5 expression also occur in chemotherapy-resistant cancer cells,15–17 but the underlying mechanism of GAS5 in chemoresistance remains poorly characterized. OSCC tends to develop chemoresistance to the current front-line chemotherapeutic agent, cisplatin (DDP). Knowledge of this resistance at the molecular level could improve OSCC therapy and contribute to improving patient survival and prognosis.
Herein, we aimed to demonstrate the functional role of GAS5 in DDP resistance and uncover its underlying mechanism of action in OSCC.
Methods
Sample collection and cell maintenance
OSCC tumor and matched normal tissue samples were obtained from OSCC patients treated at China-Japan Friendship Hospital between July 2017 and May 2019. For all patients, consent to participate was provided in written form. All patient details have been de-identified. This study was approved by the ethics committee of China-Japan Friendship Hospital (2017-28). The reporting of this prospective study conforms to STROBE guidelines. 18
CAL27 (OSCC) and normal NOK cells were purchased from the Shanghai Model Cell Bank (Shanghai, China) and cultured in RPMI medium under standard culture conditions. DDP-resistant OSCC cells (CAL27/DDP) were established by exposing CAL27 cells to increasing doses of DDP in a stepwise manner.
Cell transfection
SiRNAs were designed against GAS5 (si-GAS5), scrambled controls (si-con), miR-196a, and anti-miR-196a by GenePharma (Suzhou, China). GAS5 was sub-cloned into pcDNA-3.1 (oe-GAS5) for overexpression studies. CAL27 and CAL27/DDP cells were transfected for 48 hours using Lipofectamine2000 (Thermo Fisher Scientific, Waltham, MA, USA) according to the manufacture’s protocol, and then subjected to functional experiments. Quantitative real-time PCR (qPCR) was used to assess transfection efficiency.
qPCR analysis
Cells and tissues were lysed in TRIzol (Invitrogen, Waltham, MA, USA) for RNA extraction. cDNA was synthesized using M-MLV Reverse Transcriptase, and qPCR was performed on an ABI Prism 7900 using VeriQuest Fast SYBR Green qPCR Master Mix (Thermo Fisher Scientific). Primer sequences were as follows: GAS5 forward, 5′-AGCTGGAAGTTGAAATGG-3′, GAS5 reverse, 5′-CAAGCCGACTCTCCATACC-3′ and miR-196a forward, 5′‑ACCTGCGTAGGTAGTTTCATGT‑3′, miR-196a reverse, 5′‑CGTCAGAAGGAATGATGCACAG‑3′. The 2−ΔΔCt method was used for relative gene expression analysis. Values were normalized to U6 or GAPDH.
Chemoresistance assays
To assess DDP sensitivity, cells in 96-well plates were treated with 0.5, 1, 2, 4, 8, 16, 32 μM DDP for 48 hours. Cells were then assessed for viability throughout treatment using Cell Counting Kit 8 (CCK-8) reagent. Absorbance values at 450 nm were detected using a microplate reader and normalized to no-drug controls.
Apoptosis assay
To assess DDP-induced cell death, cells were treated with DDP for 48 hours and stained with a commercial propidium iodide and Annexin V staining kit (KeyGEN Biotech, Nanjing, China). Apoptosis rates were assessed by flow cytometry. 19
Nuclear/cytoplasmic fractionations
Subcellular fractionations were separated using PARIS Kits (Life Technologies, Carlsbad, CA, USA). GAS5 levels in each fraction were assessed by qPCR.
Luciferase reporter assays
Wild type (with predicted miR-196a binding sites) and mutant GAS5 luciferase reporter vectors (GAS5-WT and GAS5-MUT, respectively) were obtained from GenePharm and transfected into cells together with miR-con or miR-196a. After transfection for 48 hours, dual luciferase assays were performed using commercially available kits (Promega, Madison ,WI, USA).
RNA immunoprecipitation (RIP) interaction assays
The Magna RIP RNA-Binding IP Kit (Millipore, Burlington, MA, USA) was employed for binding assays. Cells were lysed with RIPA buffer and treated with magnetic beads bound to anti-Ago2 or anti-IgG antibodies (Millipore). Samples were treated with proteinase K, RNA was isolated, and miR-196a and GAS5 expression were assessed by qPCR.
Statistical analysis
For statistical comparisons, data were analyzed using SPSS software (IBM, Armonk, NY, USA). Data are presented as the mean ± SD. The Student’s t test or a one-way analysis of variance were used for data comparisons. P < 0.05 was deemed statistically significant.
Results
GAS5 expression in OSCC
To assess the role of GAS5 in OSCC, we first detected GAS5 expression in tumor tissues of OSCC patients (n = 32) and compared its expression with normal tissues (n = 32). We observed a significant downregulation of GAS5 in OSCC tumor tissues (Figure 1a). GAS5 was most notably downregulated in the OSCC tissues of DDP-resistant patients compared with the OSCC tissues of DDP-sensitive patients (Figure 1b). Similarly, the same trends in GAS5 levels were discovered in DDP-resistant OSCC CAL27/DDP cells (Figure 1c). Additionally, Kaplan–Meier survival analysis indicated that low GAS5 expression was linked with shorter survival in OSCC patients (Figure 1d). Together, these data demonstrate that low GAS5 expression is strongly associated with DDP resistance and implicate GAS5 in the cellular processes that drive tumor formation in OSCC.

GAS5 is downregulated in cisplatin (DDP)-resistant oral squamous cell carcinoma (OSCC) tissues and cells. Quantitative real-time PCR analysis indicated lower levels of GAS5 expression in OSCC tumor tissues (n = 32) vs. matched normal tissues (n = 32) (a), in DDP-sensitive vs. DDP-resistant OSCC tissues (b), and in DDP-resistant OSCC CAL27/DDP cells vs. their parental CAL27 cells or the normal cell line NOK (c) and (d) Overall survival was evaluated through Kaplan–Meier analysis between the low and high GAS5 expression groups among our clinical samples. *P < 0.05, **P < 0.01, ***P < 0.001.
GAS5 overexpression alleviated OSCC drug-resistance
We next compared the IC50 of DDP using CCK-8 assays in CAL27 and CAL27/DDP cells. CAL27/DDP cells were largely insensitive to DDP (Figure 2a). The exogenous expression of si-GAS5 in CAL27 cells reduced GAS5 expression, while oe-GAS5 increased GAS5 levels in CAL27/DDP cells (Figure 2b and 2c). Silencing GAS5 decreased the potency of DDP in CAL27 cells (Figure 2d). In contrast, the exogenous expression of GAS5 enhanced the sensitivity of CAL27/DDP cells to DDP (Figure 2e). Silencing GAS5 also reduced apoptosis rates in response to DDP (Figure 2f). GAS5 overexpression enhanced the DDP-induced apoptosis rate of CAL27/DDP cells (Figure 2g), thus overcoming DDP resistance. These data strongly implicate GAS5 in DDP resistance in OSCC tumors.

GAS5 overexpression restored cisplatin (DDP) sensitivity in oral squamous cell carcinoma (OSCC) cells. (a–b) Cell viability was determined by Cell Counting Kit 8 (CCK-8) assays in CAL27/DDP and CAL27 cells exposed to different concentrations of DDP (0.5, 1, 2, 4, 8, 16, and 32 μM) for 48 hours. (c–d) Quantitative real-time PCR analysis was performed in CAL27 cells transfected with si-GAS5 or si-con. CAL27/DDP cells were transfected with vector or oe-GAS5. (e–f) CAL27 cells were transfected with si-GAS5 or si-con. CAL27/DDP cells were transfected with vector or oe-GAS5 and treated with various concentrations of DDP (0.5, 1, 2, 4, 8, 16, and 32 μM) for 48 hours. Cell viability was determined through CCK-8 assays and (g–h) Apoptosis rates were determined by flow cytometry in si-GAS5- or si-con-transfected CAL27 cells. Vector or oe-GAS5 were transfected into CAL27/DDP cells, which were then treated with DDP. *P < 0.05.
GAS5 sponges miR-196a in OSCC cells
To study the molecular mechanisms of GAS5 related its role in DDP resistance, we predicted the downstream miRNAs of GAS5 using StarBase (http://starbase.sysu.edu.cn/) and miRanda (www.microrna.org) software. Figure 3a shows that GAS5 harbors miR-196a-binding sites. As determined by subcellular location assays, GAS5 was found to be expressed in the cytoplasm of CAL27 and CAL27/DDP cells (Figure 3b and 3c). RIP and dual luciferase reporter assays were used to assess the interaction between GAS5 and miR-196a. In both CAL27 and CAL27/DDP cells, upregulation of miR-196a dramatically reduced the activity of GAS5-WT but did not influence GAS5-MUT vectors (Figure 3d and 3e). RIP assays revealed that GAS5 and miR-196a were abundant in the anti-Ago2 group compared with the anti-IgG group (Figure 3f and 3g). Upregulating GAS5 also reduced miR-196a expression in CAL27 and CAL27/DDP cells. miR-196a expression was unaffected by overexpressing GAS5-MUT (Figure 3h and 3i) and was at high levels in drug-resistant OSCC tissues compared with tissues from drug-sensitive patients (Figure 3j). Moreover, GAS5 and miR-196a expression levels showed a negative correlation in OSCC tissues (Figure 3k). Together, these data show that GAS5 functions directly as a miR-196a sponge.

GAS5 acts as a miR-196a sponge in oral squamous cell carcinoma (OSCC) cells. (a) The GAS5 sequence containing a miR-196a-binding site as predicted by miRanda (www.microrna.org). (b–c) Subcellular fractionation assays showing the cytoplasmic distribution of GAS5 in OSCC cells. (d–e) Luciferase reporter assays and (f–g) RNA immunoprecipitation (RIP) assays were performed to verify the interaction between GAS5 and miR-196a. (h–i) Quantitative real-time (qPCR) analysis of miR-196a expression in CAL27 and CAL27/DDP cells transfected with si-con, si-GAS5, vector, oe-GAS5, or oe-GAS5-MUT. (j) qPCR analysis of miR-196a levels in OSCC tissues of cisplatin (DDP)-resistant patients compared with those of DDP-sensitive patients and (k) Correlation analysis of GAS5 and miR-196a expression in OSCC tissues. *P < 0.05, **P < 0.01, ***P < 0.001.
miR-196a enhanced drug-resistance in OSCC cells
We next transfected cells with miR-196a or anti-miR-196a and confirmed their expected effects on miR-196a expression through qPCR (Figure 4a and 4b). In CAL27 cells, miR-196a overexpression enhanced DDP resistance, while its inhibition imparted enhanced DDP sensitivity in CAL27/DDP cells (Figure 4c and 4d). Upregulating miR-196a also led to a loss of DDP-induced apoptosis in CAL27 cells (Figure 4e). In contrast, inhibiting miR-196a led to higher levels of DDP-induced apoptosis in CAL27/DDP cells (Figure 4f). These data implicate miR-196a as a key mediator of DDP resistance during OSCC progression.
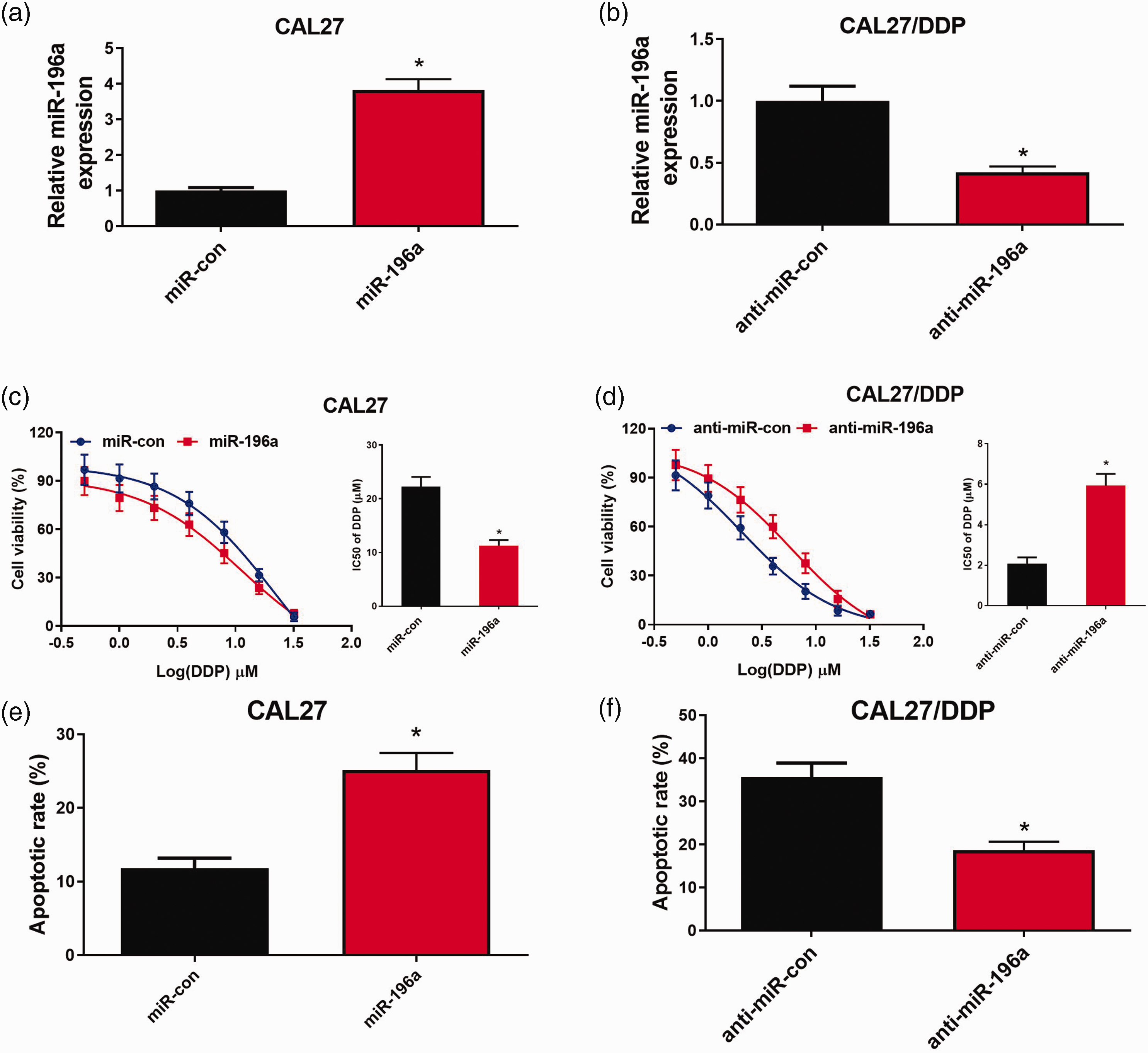
Inhibiting miR-196a enhanced the cisplatin (DDP) sensitivity of oral squamous cell carcinoma (OSCC) cells. CAL27 cells were transfected with miR-con or miR-196a. CAL27/DDP cells were transfected with anti-miR-con or anti-miR-196a, followed by the assessment of miR-196a expression by quantitative real-time PCR analysis. (a–b) The IC50 of DDP by MTT assays. (c–d) Apoptosis assays as assessed by flow cytometry (e–f). *P < 0.05.
GAS5 directly targets miR-196a
We next investigated the molecular mechanism(s) of GAS5 by expressing si-GAS5 ± anti-miR-196a in CAL27 cells and exogenously expressing oe-GAS5 ± miR-196a in CAL27/DDP cells. The effects of silencing GAS5 were alleviated by anti-miR-196a in CAL27 cells (Figure 5a). Consistent with these findings, miR-196a mimics overcame the effects of GAS5 in CAL27/DDP cells (Figure 5b). CCK-8 viability assays showed that the reduced DDP sensitivity of CAL27 cells following GAS5 silencing could be recovered by expressing miR-196a (Figure 5c). DDP sensitivity was also enhanced by miR-196a in CAL27/DDP cells overexpressing GAS5 (Figure 5d). Anti-miR-196a expression in CAL27 cells also prevented the apoptotic effects of silencing GAS5 (Figure 5e), while miR-196a prevented the pro-apoptotic effects of GAS5 in CAL27/DDP cells (Figure 5f). Thus, GAS5 overexpression enhanced DDP sensitivity in OSCC cells through its targeting of miR-196a.

GAS5 overcame cisplatin (DDP) resistance in oral squamous cell carcinoma (OSCC) cells by suppressing of miR-196a expression. CAL27 cells were transfected with si-con, si-GAS5, or si-GAS5 ± anti-miR-196a. CAL27/DDP cells were transfected with vector, oe-GAS5, or oe-GAS5 ± miR-196a, followed by the determination of miR-196a expression by quantitative real-time PCR analysis. (a–b) The IC50 of DDP by MTT assays. (c–d) Apoptosis assays as assessed by flow cytometry (e–f). *P < 0.05.
Discussion
LncRNAs have been strongly linked to chemoresistance. OSCC is treated with chemotherapeutic agents, but resistance to these drugs remains a major clinical challenge. Herein, we investigated the role of GAS5 in OSCC, which had remained largely uncharacterized. In this study, we found that GAS5 is expressed to low levels in DDP-resistant OSCC cells and tissues, and its expression contributes to the survival of OSCC patients. Overexpressing GAS5 restored DDP sensitivity in CAL27/DDP cells and downregulated miR-196a in OSCC tissues. The transfection of exogenous miR-196a alleviated the effects of GAS5 on DDP sensitivity, confirming this to be a key contributor of chemoresistance. Conversely, silencing GAS5 enhanced the drug-sensitivity of DDP-resistant OSCC through its targeting of miR-196a, as evidenced by a decline in cell viability and an increase in cell apoptosis. These data provide valuable information for the development of more effective therapeutics, which is indicated that the promise of targeting GAS5 to restore chemosensitivity in OSCC. This is particularly important given the emergence of lncRNAs as powerhouses in tumor therapy and chemoresistance. 20
New and more effective therapeutic strategies are required to circumvent chemoresistance. Here, we showed that the suppression of GAS5 in drug-resistant OSCC can predict survival. GAS5 overexpression could promote the recovery of DDP resistance in OSCC cells, highlighting this as a novel therapeutic angle. This aligns with other studies that have shown an association between GAS5 and DDP resistance, a notable example being resistant breast cancers. Restoring GAS5 expression could enhance DDP sensitivity through its ability to target miR-221-3p to regulate dickkopf 2 (DKK2) expression and activity. 21 In hepatocellular carcinoma (HCC), GAS5 sponges miR-21 and increases PTEN expression, eventually overcoming HCC resistance to DDP. 22 GAS5 enhancement also reduces resistance to doxorubicin in DDP-resistant bladder transitional cell carcinoma T24/DOX cells by enhancing DDP-induced apoptosis and inhibiting the anti-apoptotic protein Bcl-2. 23
Although there is a link between lncRNAs and chemoresistance, relatively few lncRNAs have been mechanistically characterized. Recently, lncRNAs have been documented to take part in tumor progression by acting as competing endogenous RNAs.24,25 Remarkably, it was found that the GAS5 sequence contains miR-196a-binding sites as predicted by StarBase and miRanda. More significantly, miR-196a mediates chemo resistance. MiR-196a-5p overexpression, which is induced by UCA1, activates the transcription factor CREB by binding to its promoter. This contributes to chemoresistance to cisplatin/gemcitabine via increasing cell proliferation and inhibiting apoptosis in bladder cancer cells. 26 Suppressing miR-196a could alleviate cisplatin resistance in A549/DDP cells by reducing the expression of drug resistance proteins including multidrug resistance 1 (MDR1) and multidrug resistance protein 1 (MRP1). 27 Additionally, miR-196a has been shown to be overexpressed in DDP-resistant breast cancer cells. 28 However, the mechanisms through which miR-196a participates in DDP resistance in OSCC remain undefined. Herein, we report the upregulation of miR-196a in OSCC tissues as well as in DDP-resistant OSCC tissues and cells. MiR-196a could confer DDP resistance in OSCC cells. Additionally, overexpressing miR-196a reversed the effect of GAS5 upregulation on the sensitivity of OSCC cells to DDP, fitting the established notion of miR-196a as a principal regulator in GAS5-mediated DDP sensitivity in OSCC. However, there are some limitations to this study. First, further study is required to investigate the downstream target genes and signaling pathways regulated by the GAS5/miR-196a axis in OSCC. Additionally, in vivo animal experiments should be performed to confirm our in vitro results.
Conclusion
We showed that GAS5 has low expression in DDP-resistant OSCC cell lines and tissues and is intricately linked to the survival rates of OSCC patients. Overexpressing GAS5 could restore DDP sensitivity in CAL27/DDP cells and downregulated miR-196a in OSCC tissues. Transfecting exogenous miR-196a alleviated the effects of GAS5 on DDP sensitivity, confirming its importance in chemoresistance. These findings highlight GAS5 to be a promising therapeutic target for future DDP-related interventions.
