Abstract
Objective
Cytokines and enzymes in follicular fluid (FF) may have a crucial role in fertility. This study aimed to analyse the results of in vitro fertilization/embryo transfer (IVF/ET) and compare them with matrix metalloproteinase 9 (MMP-9) and cytokine concentrations in FF.
Methods
This case–control study included 20 randomly selected patients with a positive pregnancy (PPG) test and 20 with a negative pregnancy (NPG) test after IVF/ET. In FF obtained during oocyte retrieval, the concentrations of soluble forms of MMP-9, interleukin 1 (IL-1) alpha, IL-1 beta, interferon gamma, IL-2, IL-4, IL-6, IL-8, and IL-10 were measured. Their effect on the characteristics of follicles, embryos, and the efficiency of IVF and ET were analysed.
Results
High-sensitivity IL-4 concentrations were significantly higher in patients with a PPG test than in those with an NPG test (3.15 ± 3.23 vs 1.91 ± 0.35 pg/mL). The number of top-quality embryos achieved was significantly higher in patients with a PPG test than in those with an NPG test (2.6 ± 1.39 vs 1.75 ± 1.21), and they were negatively correlated with IL-8 concentrations in FF.
Conclusion
Further research on the role of IL-4 and IL-8 in FF is required to establish any clinical benefit of determining their concentrations in FF of infertile women.
Introduction
The immune system has an important role to play in the proper development of follicles. Cytokines and enzymes present in the follicular fluid (FF) may have a crucial role in fertility. The analysis of FF requires the collection of FF via oocyte retrieval. Therefore, knowledge regarding the cytokine composition of FF in human studies is usually gained by the analysis of fluid collected during various types of gynaecological procedures mainly associated with the treatment of infertility (current or expected, e.g., owing to treatment of other serious diseases). However, knowledge of the interactions between FF cytokines and their effect on the result of fertility treatment is still limited. 1
Metalloproteinases are present in the human reproductive system and play a role in different aspects of fertility. Matrix metalloproteinase 9 (MMP-9) is a member of the gelatinase group and can degrade gelatine and collagen. MMP-9 is involved in cell migration, tissue remodelling, cell proliferation, and destruction of the extracellular matrix. 2
This study aimed to determine the concentrations of MMP-9 and certain proinflammatory and adaptive immunity cytokines, namely interleukin 1 alpha (IL-1α), IL-1 beta (ß), interferon gamma (INF-γ), IL-2, IL-4, IL-6, and IL-10 in FF of women who were prepared for the in vitro fertilization (IVF) procedure, and to examine the association between their underlying fertility problem and immunological profile. The secondary aim was to establish if any of the analysed cytokine or MMP-9 concentrations can predict successful IVF outcomes.
Methods
Patients
This case–control study consisted of patients who underwent IVF in the Fertility Clinic in Bydgoszcz, Poland between May 2021 and September 2021. Forty randomly selected patients were included in the study. Twenty patients had a positive pregnancy (PPG) test after the IVF procedure (positive pregnancy test group) and 20 had a negative pregnancy (NPG) test (negative pregnancy test group). All patients suffered from infertility, which was defined as the inability to achieve pregnancy after 1 year of regular intercourse. Patients were admitted to the clinic for an IVF procedure, for which they were qualified according to recommendations of the Polish Society of Reproductive Medicine and Embryology and the Polish Society of Gynecologists and Obstetricians. 3 Other inclusion criteria were an age of 18 to 45 years and the ability to understand and agree to the principles of the study.
All patients underwent a detailed medical interview, physical examination, and gynaecological assessment of the primary reason for their infertility. We excluded uncooperative patients, pregnant or breastfeeding women, and patients with any acute condition, such as undergoing viral or bacterial infection, past or undergoing oncological treatment, rheumatological disorders, or taking medication that could have an influence on the results of the study, especially systemic steroids, chemotherapy, biological treatment, and immunotherapy.
The size of the study population was established before conducting the study and was based on previous studies on the cytokine profile of FF in women who underwent IVF. Another factor that was taken into account was the limited financial support for the research.
The reporting of this study conforms to the STROBE guidelines. 4 All patients’ medical records and personal information were de-identified to protect the patient’s privacy.
Gynaecological assessment
All patients underwent a detailed medical interview, which included the course of fertility treatment, a physical examination, and gynaecological and cytological examinations. Ultrasound (GE Voluson S10 Expert; Chicago, IL, USA) assessment of the reproductive system was performed to accurately assess antral follicles. Vaginal biocenosis and the pH were determined.
All patients had blood sampled for the assessment of concentrations of anti-Müllerian hormone, follicle-stimulating hormone, prolactin, oestradiol, and progesterone, morphology, electrolytes, activated partial thromboplastin time, blood type, hepatitis B core antibody, hepatitis B surface antigen, anti-hepatitis C virus, anti-human immunodeficiency virus 1–2, the Venereal Disease Research Laboratory test, and Chlamydia trachomatis (PCR). We also evaluated the concentrations of immunoglobulin (Ig) G and IgM antibodies to toxoplasmosis, rubella, and cytomegalovirus.
The male partners had a sperm quality examination and blood sampling for the assessment of blood type and hepatitis B core antibody, hepatitis B surface antigen, anti-hepatitis C virus, anti-human immunodeficiency virus 1–2, the Venereal Disease Research Laboratory test, IgM, and IgG cytomegalovirus.
Patients’ hormonal stimulation, oocyte retrieval, embryo quality assessment, and IVF procedure
Hormonal stimulation of the patients and the procedure leading to IVF were carried out by following the currently applicable standards of the Polish Society of Reproductive Medicine and Embryology and the European Society of Human Reproduction and Embryology. Ovarian hormonal stimulation was conducted in accordance with a short protocol in one menstrual cycle, with the administration of gonadotropin. Six days later, a gonadotrophin-releasing hormone antagonist was administered. Ultrasound assessment of follicles up to the diameter of 18 mm was performed when recombinant human chorionic gonadotrophin ([hCG], Ovitrelle, 250 mcg; Merck, Darmstadt, Germany) was administered. Oocyte retrieval was performed transvaginally 34 to 36 hours after hCG injection. The procedure was performed using an 18 G needle Oocyte Recovery Set, 330 mm (Wallace, Trumbull, CT, USA). FF was obtained to sterile tubes (Falcon, New York, NY, USA) and then transferred to Petri dishes (90 × 15 mm; Falcon) at 37°C. Using a stereoscopic microscope, cumulus–oocyte complexes (COCs) were found and transferred to an organ culture plate (Falcon) with MHM-C (Irvine Scientific, Santa Ana, CA, USA) medium. Using two needles combined with tuberculin syringes (1 mL-U40, KD-JECT III; KD Medical GmbH, Berlin, Germany), the cumulus was cut and oocytes were placed in an incubator (K-systems, Birkeroed, Denmark) at 37°C in CSC-C media droplets (IrvineScientific) coated with oil (Vitrolife, Västra Frölunda, Sweden) on IFV 35-mm (Nunc, Roskilde, Denmark) plates. Hyaluronidase (IrvineScientfic) and stripper tips of 135 μm (ORIGIO® Micropipettes; Origo, Trumbull, CT, USA) were used to decolonize oocytes. The purified oocytes were fertilized with sperm on 50 × 9-mm (Falcon) plates in MHM-C drops coated with oil using micropipettes (ICSI Micropipettes, holding micropipettes, ORIGIO® Micropipettes; Origo). Fertilization was performed using an Olympus IX inverted microscope (Tokyo, Japan) with Hoffman contrast equipped with Integra Ti micromanipulation equipment (Research Instruments, Falmouth, UK). FF samples from each patient were taken on the day of oocyte retrieval and stored at −80°C until immunological assessment.
Oocyte fertilization occurred between 39 and 41 hours after pick-up. Assessment of fertilization was performed at 17 ± 1 hours. Embryo assessment on the third day, according to Gardner and Schoolcraft criteria, occurred at 68 ± 1 hours after fertilization, and assessment on the fifth day occurred at 116 ± 1 hours after fertilization. 5 The quality of the embryos was assessed by an embryologist on the third and fifth days after fertilization. As performed in previous studies, the embryos were assessed as top-quality or non-top-quality. 6 Depending on the patient’s individual decision and the assessed chances of succeeding, one or two embryos were chosen for transfer to the uterus on the fifth day after fertilization. During embryo transfer (ET), the endometrium needed to be at least 7 mm thick. The transfer was performed using a SureView Embryo Replacement Catheter (WLC CE123; Wallace), which was placed 1.7 to 2 cm into the uterus with guidance by ultrasound.
Clinical confirmation of pregnancy was based on blood serum ß-hCG concentrations between the 10th and the 15th days after the transfer.
Immunoassay
FF obtained during oocyte retrieval in stimulated cycles was stored at a temperature of −80°C until assessment. The concentrations of IL-1α, IL-1ß, IL-2, IL-4, IL-6, IL-8, IL-10, IFN-γ, and MMP-9 in FF were measured by commercially available enzyme-linked immunosorbent assay kits (Diaclone, Medix Biochemica Group; Quantikine™ R&D Systems; Kauniainen, Finland). According to the manufacturer’s instructions, these tests are intended for quantitative in vitro measurement in human serum, plasma, tissue homogenates, cell lysates, cell culture supernatant, and other biological fluids. All laboratory procedures were performed by following the accompanying manufacturer’s instructions. A high-sensitivity method was used for IL-2, IL-4, IL-6, and IL-10.
The absorbance value was determined on a spectrophotometer (Infinite® F50; Tecan, Switzerland). The Magellan™ reader control and data analysis software (Männedorf, Switzerland) was used to plot the calibration curve and read the concentrations of the test samples.
IL-1α and IL-1ß (Human IL-1α ELISA Kit 101A-33 and Human IL-1β ELISA Kit 101B-59, respectively; Diaclone, Medix Biochemica Group) assays used the quantitative sandwich enzyme immunoassay technique. The sensitivity or the minimum detectable dose of human IL-1α was 10.0 pg/mL. The maximum measurable dose of human IL-1α was 1000.0 pg/mL. The sensitivity or the minimum detectable dose of human IL-1ß was 6.5 pg/mL. The maximum measurable dose of human IL-1ß was 500 pg/mL. There was no cross-reactivity or interference between human IL-1ß or IL-1ß and analogues.
IL-2, IL-4, IL-6, IL-10, and IFN-γ (Human IL-2 HS ELISA Kit 1HS2-21, Human IL-4 HS ELISA Kit 1HS4-21, Human IL-6 HS ELISA Kit 1HS6-76, Human IL-10 HS ELISA Kit 1HS10-23, and Human IFN-γ HS ELISA Kit 1HSIFG-33, respectively; Diaclone, Medix Biochemica Group) assays used the quantitative high sensitivity sandwich enzyme immunoassay technique. The sensitivity or the minimum detectable dose of human IL-2 was 0.97 pg/mL. The maximum measurable dose of human IL-2 was 60.0 pg/mL. The sensitivity or the minimum detectable dose of human IL-4 was 0.31 pg/mL. The maximum measurable dose of human IL-4 was 10.0 pg/mL. The sensitivity or the minimum detectable dose of human IL-6 was 0.81 pg/mL. The maximum measurable dose of human IL-6 was 50.0 pg/mL. The sensitivity or the minimum detectable dose of human IL-8 was 12.3 pg/mL. The sensitivity or the minimum detectable dose of human IL-10 was 0.98 pg/mL. The maximum measurable dose of human IL-10 was 50.0 pg/mL. The sensitivity or the minimum detectable dose of human IFN-γ was 0.69 pg/mL. The maximum measurable dose of human IFN-γ was 25.0 pg/mL. No cross-reactivity or interference between human IL-2, IL-4, IL-6, IL-10, or IFN-γ and analogues was observed.
The MMP-9 (DMP900, Quantikine™ ELISA, Human MMP-9 Immunoassay; R&D Systems, Inc.) assay used the quantitative sandwich enzyme immunoassay technique. The sensitivity or the minimum detectable dose of human MMP-9 was 0.156 ng/mL. The maximum measurable dose of human MMP-9 was 20.0 ng/mL. No cross-reactivity or interference between human MMP-9 and analogues was observed.
Statistical analysis
The Shapiro–Wilk test was used to determine if a variable was normally distributed in the population. The Mann–Whitney test was used to compare quantitative variables between two groups. The relationship between two quantitative variables was assessed with Spearman’s coefficient of correlation. Odds ratios (ORs), which were calculated by univariate logistic regression, were used to analyse the effect of selected variables on the dichotomous outcomes (PPG/NPG tests). The significance level for all statistical tests was set to 0.05. R 4.1.2 (https://www.R-project.org/ and MS Excel 365 (Microsoft, Redmond, WA, USA) were used for computations.
Ethics statement
The study was approved on 18 May 2021 by the Collegium Medicum in Bydgoszcz, Nicolaus Copernicus University in Torun, Bioethical Committee (classification number: KB 334/2021). All patients provided informed written consent to participate in the study.
Results
We included 40 women (age: 27–44 years, mean: 34.4 ± 4.5 years) in this study. All women underwent IVF. In 20 patients, the result of a pregnancy test was positive 10 to 15 days after transfer, and it was negative in 20 patients. The PPG and NPG groups were comparable regarding age, ethnicity, height, weight, and causes of infertility, and experienced a comparable course of ovarian stimulation and oocyte retrieval. The general characteristics of the patients are shown in Table 1. Figure 1 shows the diagnosed cause of infertility in both groups. Progesterone, oestradiol, anti-Müllerian hormone, and prolactin concentrations were not different between women who had successful IVF and those who did not become pregnant. In the PPG group, 4 (20%) women had a miscarriage and 16 (80%) were able to successfully continue the pregnancy.
General characteristics of the patients.

SD, standard deviation; BMI, body mass index; AMH, anti-Müllerian hormone.
Because of the non-normal distribution of the data, the median and range (or interquartile range) are shown, and non-parametric tests were used in the statistical analysis. The Mann–Whitney test was used to analyse quantitative variables.

Diagnosed underlying reasons for infertility.
The results of ovarian stimulation were compared between the groups, including the total number of good quality COCs retrieved, the number of oocytes in metaphase (M) II and MI, and the number of germinal vesicles. Assessment of the embryos was performed on the basis of the Gardner and Schoolcraft criteria on the third and fifth days after fertilization. 5 Assessed embryos were divided into three subcategories using the Istanbul consensus workshop on embryo assessment (2011) and standards for the assessment of oocytes and embryos – Polish Society of Reproductive Medicine and Embryology recommendations as follows: 7 top-quality embryos, non-top-quality embryos, and non-viable embryos (Table 2).
Retrieved oocytes and the number of fertilized embryos.

SD, standard deviation; COC, cumulus–oocyte complex; MII, metaphase II; MI, metaphase I; GV, germinal vesicle.
The Mann–Whitney test was used to analyse quantitative variables.
The number of MII cells had a positive effect on the chance of conceiving after ET, with an OR of 1.326 (univariate logistic regression; 95% confidence interval: 1.026–1.712; p = 0.031). Every additional MII led to a 32.6% greater chance of a PPG test in patients. The results of the univariate logistic regression were not significant for COCs; MI, top-quality and non-top-quality embryos, body mass index (BMI), and body weight.
All assessed cytokines were found in FF. The results of cytokines and MMP-9 are shown in Table 3. The effect of MMP-9 and cytokine concentrations on the results of the IVF procedure was analysed (Table 4). We found that high-sensitivity (hs) IL-4 concentrations were significantly higher in the group of women who became pregnant during the procedure than those who did not (p = 0.035).
Concentrations of cytokines and MMP-9 in follicular fluid of the patients.

SD, standard deviation; Min, minimum; Max, maximum; Q, quartile; IL, interleukin; INF, interferon; hs, high-sensitivity; MMP-9, matrix metalloproteinase 9.
Effect of MMP-9 and cytokine concentrations on results of the IVF procedure.

SD, standard deviation; IL, interleukin; INF, interferon; hs, high-sensitivity; MMP-9, matrix metalloproteinase 9.
The Mann–Whitney test was used to analyse quantitative variables.
The age of women and the diagnosed reason for infertility were not significantly correlated with the concentrations of measured immunological parameters, MMP-9, or the results of the IVF procedure. Interestingly, hsIL-6 concentrations were positively correlated with the BMI of patients (r = 0.49, p = 0.001, Figure 2).
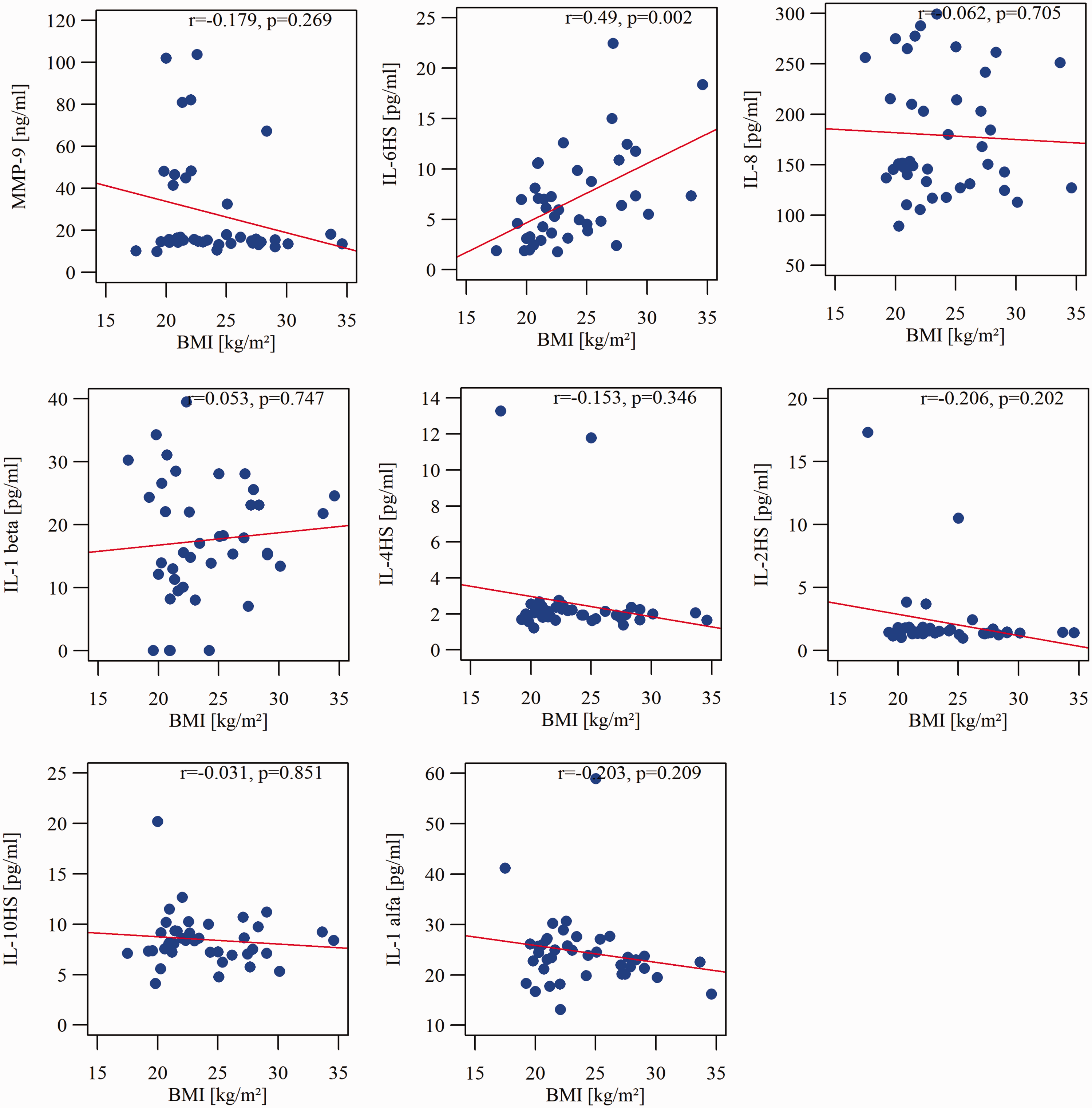
Spearman rank correlations between cytokine concentrations in follicular fluid and BMI of the patients.
There was a positive correlation between IL-2 concentrations and COC-1 (r = 0.33, p = 0.038) and the number of oocytes in MII (r = 0.332, p = 0.037). Additionally, the number of oocytes in MII was negatively correlated with IL-8 concentrations (r = −0.43, p = 0.006). The number of top-quality embryos was negatively correlated with IL-8 concentrations in FF (r = −0.43, p = 0.006). The number of non-viable embryos was not correlated with any immunological parameters assessed in this study. INF-γ was positively significantly correlated with hsIL-2 concentrations (r = 0.419, p = 0.008). hsIL-2 concentrations were positively significantly correlated with hsIL-4 concentrations (r = 0.487, p = 0.002). hsIL-6 concentrations were negatively significantly correlated with MMP-9 concentrations (r = −0.324, p = 0.042).
Discussion
FF is rich in cytokines, and a balance within the immune system is suspected to play an important role in the proper development of ovarian follicles. Furthermore, a balance between the concentrations of cytokines is crucial for the development and maturation of the follicle. An immunological balance has an effect on the natural course of ovulation and corpus luteum formation, but also on embryo implantation. The maintenance of pregnancy is also related to the immune system. Unfortunately, the knowledge of cytokines in FF is limited, not only because of the complexity of the interactions between cytokines and the general condition of the female body, but also owing to problems with obtaining FF from healthy women. The analysis of FF requires its collection only by puncturing the ovary. This procedure is usually executed in connection with various gynaecological procedures. Clinical indications for a given procedure, the underlying medical condition of the patient, and the pharmacological method of preparation need to be taken into account when interpreting such results. 1 The current study is based on FF obtained from women with different causes of infertility. We found several interesting findings.
MMP-9 in infertility
MMP-9 is one of the most widely investigated matrix metalloproteinases. MMP-9 and MMP-2 have been examined in FF in animal models. Hrabia et al. 8 found a relatively higher expression of MMP-2 and MMP-9 mRNA as follicle development progressed toward ovulation (3 vs 22 hours). Peralta et al. 9 found that MMP-9 activity was increased in follicular cysts and absent in the FF of pre-ovulatory follicles in Bovine cystic ovarian disease. Malvezzi et al. 10 investigated the differences in the activity of MMP-2 and MMP-9 and their tissue inhibitors (TIMPs) in FF of women with endometriosis undergoing IVF. They found that an increase in MMP-2 and MMP-9 activity, as well as a decrease in TIMP-1 expression, were associated with poor oocyte and embryo development. Singh et al. 11 studied 340 infertile patients and found that, in women who qualified for IVF owing to endometriosis, there was an extensive MMP-9/TIMP-1 imbalance. Poor oocyte and embryo development in these patients was associated with an increase in MMP-2 and MMP-9 and a decrease in TIMP-1 expression. Progesterone supplementation appeared to be effective in improving this imbalance.
Horka et al. 12 published an interesting study on cytokine concentrations in FF and sera of 58 female patients undergoing IVF treatment (29 with PPG tests and 29 with NPG tests). They found that women with IVF and a PPG test showed the highest MMP-9 concentrations in blood serum (833.5 [686.0–958.7] ng/mL) and FF (9.6 [6.0–17.0] ng/mL) compared with women with unsuccessful IVF. The authors concluded that MMP-9 concentrations could be a good predictor of a successful outcome of IVF. 12 Their finding is not in line with our finding that FF MMP-9 concentrations were not correlated with the success rate of IVF. The two studies are comparable to some extent, but the lack of agreement between them suggests the necessity of further research in a larger population and taking into account different factors that may contribute to successful conception. In this study, there were no significant differences between the concentrations of the analysed cytokines or MMP-9 concentrations and the underlying disease. However, the subpopulations of patients with different causes of infertility were relatively small, which could have affected our results.
T helper 2-dependent immune response and pregnancy
One of the most interesting results in this study is that there was a positive correlation between FF IL-4 concentrations and the success rate of IVF, which was defined as an elevated blood serum ß-hCG concentration between the 10th and 15th day after transfer. IL-4 and IL-13 are important cytokines in the development of allergic diseases. IL-4 and its STAT6-signalling pathway are necessary for the expression of T helper 2-dependent immune responses in many tissues, such as the skin, lungs, and gut. 13 There are several mechanisms in which allergic disorders may influence fertility, including the presence of persistent, mild inflammatory processes, semen allergy, and intolerance to medication used in infertility treatment. 14 Gade et al. 15 pointed out that bronchial asthma is a chronic disease, and its pathogenesis is associated with many factors, such as numerous mediators of inflammation. In asthmatic patients, increased activity of pro-inflammatory cytokines in peripheral blood (e.g., IL-6 and tumour necrosis factor [TNF]-α) was found. A cytokine imbalance promotes neutrophil and natural killer cell recruitment. Moreover, there are some risk factors for both infertility and asthma, such as overweight in polycystic ovary syndrome and obesity-associated bronchial asthma.
With regard to IVF, Ostanin et al. 16 found that an unsuccessful IVF cycle was associated with lower concentrations of IL-2, IL-4, IL-7, granulocytic cerebrospinal fluid, and macrophagic inflammatory protein 1b in the FF of patients. They also found that IL-8 and IL-13 concentrations were elevated. This result is in line with our finding on IL-4 concentrations, although we did not find such a correlation for IL-2 or IL-8.
Gaafar et al. 17 examined FF in 38 patients, including 18 with polycystic ovaries and 20 with male infertility/unexplained infertility. In FF, they determined the concentrations of granulocyte colony-stimulating factor, regulated upon activation of normal T cell expressed and presumably secreted (RANTES), TNF-α, IFN-γ, and IL-4 and IL-2. Granulocyte colony-stimulating factor concentrations showed a significant positive effect on the pregnancy outcome. None of the concentrations of the cytokines were associated with the quality of the embryo. Furthermore, there was no association between the concentrations of cytokines in FF and polycystic ovaries as the cause of infertility. A previous study used the magnetic multiplex immunoassay to measure the concentrations of some intrafollicular cytokines in 60 patients who underwent controlled ovarian hyperstimulation. These cytokines included granulocyte macrophage colony-stimulating factor, INF-γ, TNF-α, IL-10, CXCL8/IL-8, IL-6, IL-5, IL-4, IL-2, and IL-1β. Intrafollicular TNF-α was significantly and inversely correlated with the severity of ovarian hyperstimulation syndrome. The other nine measured cytokines showed no significant correlation with the incidence of ovarian hyperstimulation syndrome. 18
Bersinger et al. found that, in modified natural and gonadotropin-stimulated controlled IVF cycles, IL-6, IL-8, IL-10, IL-18, monocyte chemoattractant protein-1, vascular endothelial growth factor, and leukaemia inhibitory factor showed notably higher median concentrations in FF than in serum. These authors also found that IL-8 was reduced in controlled IVF cycles. 19 In our study, all analysed cytokines were present in FF, which additionally suggests that FF is immunologically diverse and active, and also suggests ovarian production. The question of why IL-4 can have a positive effect on the results of IVF is still unknown. This finding might be due to the fact that the shift in Th1/Th2 immunological balance towards Th2 is beneficial for the development of follicles. A bias from the T helper 1 cytokine profile toward the T helper 2 profile contributes to the maintenance of successful pregnancy. 20 An interesting study by Kalu et al. 21 analysed certain cytokines in the serum of women with a history of recurrent failed implantation as a result of IVF. They found that the ratios of IFN-γ:IL-4 and TNF-α:IL-4 at oocyte retrieval were higher than the pre-treatment ratios, which suggested a Th1 bias. In women with successful IVF, these ratios at oocyte retrieval were significantly lower than the pre-treatment ratios, which suggested a Th2 bias.
Effect of IL-8 on oocyte and embryo quality
In the current study, increased IL-8 concentrations in FF had a negative effect on the number of top-quality embryos achieved during IVF. Furthermore, the number of MII cells were negative correlated with IL 8 concentrations, and the number of MII cells had a positive effect on the chance of conceiving after ET. IL-8 is a neutrophil-specific factor involved in inflammatory processes and angiogenesis. 22 In a recent study by Gica et al., 23 152 patients with non-obstructive endometriosis were analysed. The women were divided into the fertile and infertile groups. The patients with infertility had significantly higher serum IL-6 serum concentrations than the fertile patients, but serum IL-8 concentrations were not significantly different between these groups. In an interesting study by Malizia et al., 24 samples of FF from 63 patients were analysed for IL-8 concentrations. They found that large follicles contained higher IL-8 concentrations than small follicles. Additionally, IL-8 concentrations in the large follicles of younger women were higher than those in older reproductive-age women. However, there was no significant correlation between FF IL-8 concentrations and the IVF outcome, which was defined as achieving pregnancy. The activity of IL-8 in FF, correlations with serum IL-8, and the effect of IL-8 on fertility require further analysis.
Study limitations, unanswered questions, and future studies
The main limitation in the interpretation of the results of this case–control study is the relatively small and diverse research population. A sample size/power analysis was not performed, and the number of patients was based on previous research and the financial support received. The cytokine profile of women in blood serum and FF needs to be analysed in a larger population of infertile women, especially in selected groups of patients suffering from polycystic ovarian syndrome or endometriosis. A multivariate analysis in a study of a larger population is also required to exclude confounding factors.
Furthermore, the interpretation of our results is difficult because the measured cytokines play important roles in many other conditions. The interactions between the current state of health of the patient and the interactions between cytokines in FF and blood serum are complex and require in-depth analyses. There are many examples of diseases with a prominent cytokine profile, such as in asthma, where IL-4 and IL-5 have crucial roles. In inflammatory bowel disease and rheumatoid arthritis, the pathogenesis is different, and TNF-α concentrations are increased as a result of chronic inflammation. Furthermore, in chronic obstructive pulmonary disease, cigarette smoke promotes IL-8 production.25,26 IL-6 and IL-8 are relevant markers of a cytokine storm in coronavirus disease 2019 (COVID-19) pneumonia. 27 That this study was performed during the COVID-19 pandemic is also a relevant factor. COVID-19 decreases male fertility. Impaired spermatogenesis via COVID-19 decreases testosterone concentrations by disturbing cytokines, such as TNF-α, IL-4, IL-6, and IL-12. Finally, this situation leads to a decreased sperm count. 28
The role of key cytokines in FF, such as their role in male infertility, requires further investigation. Saxena et al. 29 showed that seminal IL-8 concentrations were negatively correlated with semen parameters, such as sperm motility and morphology. The authors found that, in male patients with leukocytospermia, IL-8 concentrations can be five times higher than those in controls. The cytokine profile of seminal fluid of male partners of patients could provide more insight into the immunological background of success or failure of IVF. To the best of our knowledge, no study has determined the immunological profile of female and male partners and analysed it in terms of embryo development and success of IVF. This “one-sided” approach could explain the differences between results achieved by different researchers.
One of the most crucial questions to be answered is if knowledge of the immunological profile of FF affects the treatment course of infertile patients. Modifying cytokines in FF to increase the chances of conception would be useful. Identifying universal immunological markers of success or failure of IVF could potentially reduce the cost and health burden of the procedure, and provide patients with better insight into treatment options. In general, treatment of the underlying reason for disrupting the cytokine profile is beneficial and may reverse the disease’s negative effects on fertilization and pregnancy outcomes. However, whether target treatment would help biological treatment aimed at a specific immunological path is unknown There is some evidence that biological treatment used in several different conditions, such as arthritis or allergies, can be relatively safe in pregnant or breastfeeding women. Research on modifying cytological profiles in infertile women would be interesting, but difficult to achieve because of bioethical concerns.1,30,31
Another interesting aspect of the FF environment that we did not examine is the complex metabolism of phospholipids. This represents a highly controlled cellular signalling network that is essential for mounting an effective innate immune response and may also affect the cytokine composition. 32 Controlled ovarian stimulation during IVF affects the concentration of phospholipids in serum and FF. 33 Comparing these concentration with those of cytokines and enzymes from the FF that we evaluated in our study could be important. Therefore, the lack of these data in our analysis is another potential limitation.
Conclusion
This study provides some insight into the cytokine profile of FF in patients undergoing IVF. The main finding in this study is that there is a correlation between a higher concentration of IL-4 and a PPG test. Lower IL-8 concentrations are significantly correlated with the number of top-quality embryos achieved during IVF. Further research on the role of IL-4 and IL-8 in FF is required to establish the potential clinical benefit of determining their concentrations in FF of infertile women. Knowledge of the immunology of FF could lead to individualized treatment, focussed on modifying the immunological balance to increase the chances of conceiving.
Footnotes
Author contributions
RA contributed to the study design, collected research material, interviewed with the patients, obtained consent, and prepared the manuscript. NUS contributed to the study design, and helped to prepare the manuscript. KL contributed to the study design and performed immunoassays. ZB evaluated and revised the manuscript. MD evaluated and revised the manuscript. All authors gave final approval of the version to be published and agree to be accountable for all aspects of the work related to its accuracy or integrity.
Data availability statement
The dataset supporting the conclusions of this article is included within the article. The source dataset is available from authors upon request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
