Abstract
Objective
We investigated the effect of dexmedetomidine anesthesia on postoperative cognitive function after pulmonary surgery.
Methods
A blinded, prospective, randomized, placebo-controlled study was performed on 60 patients (age range 65–74 years) undergoing lobectomy by video-assisted thoracoscopic surgery (29 in the dexmedetomidine group; 31 in the placebo group). Dexmedetomidine group patients received dexmedetomidine, and placebo group patients received an equal volume of physiological saline 20 minutes before anesthesia induction. Cognitive function was evaluated using the Montreal Cognitive Assessment 1 day before surgery and on postoperative day (POD)1, POD3, and POD7. The regional cerebral oxygen saturation (rSO2) was monitored continuously by near-infrared spectroscopy before anesthesia.
Results
The Montreal Cognitive Assessment score was significantly different between the two groups on POD1 (dexmedetomidine 26.4 ± 0.73 vs. placebo 25.5 ± 0.96) and POD3 (dexmedetomidine 27.1 ± 0.79 vs. placebo 26.6 ± 0.80). Specifically, attention and orientation scores were increased in the dexmedetomidine group on POD1 and POD3. The rSO2 was not significantly different between the dexmedetomidine and placebo groups.
Conclusion
Dexmedetomidine given before induction of anesthesia could reduce the risk of postoperative cognitive dysfunction and might not decrease rSO2. Hence, dexmedetomidine could be employed in pulmonary surgical procedures, especially for older patients with a high risk of cognitive dysfunction.
Keywords
Introduction
Postoperative cognitive dysfunction (POCD) is a condition in which consciousness, cognitive function, memory, and orientation decline after surgery. Although the pathogenesis of POCD is not completely clear, the release of a variety of cytokines that trigger the systemic inflammatory response caused by surgery, anesthesia, brain oxygen reduction, and other related factors might be a possible explanation.1,2 POCD is closely related to the trauma experienced from a surgical procedure and can impair the prognosis and quality of life of patients. The POCD prevalence ranges from 16% to 62% in patients undergoing hip fracture repair.3,4 In particular, older patients are vulnerable to memory disturbances and other types of cognitive impairment after surgical procedures. 5
Dexmedetomidine is an α2-adrenoreceptor agonist. It is a sedative and analgesic agent that has been licensed recently in the USA as intensive care unit (ICU) sedation ≤24 hours after surgery. 6 Its use has been linked to reductions in postoperative delirium, stress, and inflammatory responses, resulting in improved protection of the nervous system.7–9 Several studies on cognitive function in volunteers or patients who received dexmedetomidine have been published. They showed that preoperative administration of dexmedetomidine could effectively protect cerebral blood vessels, improve cognitive dysfunction and damage to attentional network function, and increase postoperative quality of life. 10 The effects of dexmedetomidine on cognitive function after various surgeries were reported.11–13 Recent studies have shown that addition of dexmedetomidine to postoperative intravenous analgesia pumps did not improve the patients’ postoperative cognitive function. 14 Shi et al 15 found that male patients undergoing lobectomy could benefit from continuous dexmedetomidine (0.5 µg/kg/hour) administration in terms of postoperative delirium and postoperative cognitive impairment. However, there are no reports of dexmedetomidine administration before anesthesia in pneumonectomy.
Studies have indicated that dexmedetomidine causes a reduction of cerebral blood flow (CBF) in humans.11,16 Drummond and colleagues showed that dexmedetomidine caused a dose-related reduction in CBF and the cerebral metabolic rate (CMR) in healthy individuals. 17 Furthermore, a study carried out in anesthetized dogs indicated that the reduction in CBF was not accompanied by a simultaneous reduction of CMR. 18 Regional hemoglobin oxygen saturation (rSO2) and CBF velocity were reported to be comparably preserved during sedation using dexmedetomidine or propofol. However, the effect of dexmedetomidine on rSO2, which is an indication of the actual regional cerebral oxygen supply, was not indicated, which raised concern regarding whether postoperative cognitive function would be affected. Dexmedetomidine might play a dual role during the perioperative period in patients who undergo pulmonary surgical procedures.
In this study, we investigated the effect of dexmedetomidine on rSO2 to determine whether the cerebral blood supply was affected, and we investigated the effect of dexmedetomidine anesthesia on POCD after pulmonary surgery.
Methods
Study protocol
We carried out this prospective, randomized, placebo-controlled trial in Beijing Tuberculosis and Thoracic Tumor Research Institute (Beijing, China). Ethical approval for this study was obtained from the Beijing Chest Hospital Ethics Committee on 5 April 2017 (BJXK-2017[10]). Patients who underwent an elective pulmonary surgical procedure from May 2018 to August 2021 were included. Written informed consent was obtained from all individuals participating in the trial. Our study adheres to the CONSORT guidelines and the CONSORT checklist, which was submitted as a supplementary file. 19 The original trial was registered at ClinicalTrials.gov (number: ChiCTR2000038878; principal investigators: Fei Wang and Tao Liu; date of registration: 8 October 2020).
Patients were excluded if they had cerebrovascular disease, arrhythmia, sinus bradycardia, ischemic heart disease, a history of obstructive sleep apnea, or a known allergy to dexmedetomidine; had an education level below junior high school; were taking medication for mental disorders; had previous neurologic disease (e.g., stroke, seizures, dementia); or had memory impairment.
Pulmonary resections, including wedge resection, lobectomy, bilobectomy, sleeve lobectomy, and pneumonectomy, were selected on the basis of tumor location. Video-assisted thoracoscopic surgery was the first choice, and thoracotomy was performed when necessary. Unilateral pulmonary collapse was routinely performed in pulmonary surgeries to improve the surgical view and surgical procedure.
Grouping and drug administration
During the study period, patients were randomly divided into a placebo group and a dexmedetomidine group using a computer-generated random number table. The included patients were randomly assigned to receive dexmedetomidine or physiological (0.9%) saline. In an emergency (e.g., unexpected, rapid deterioration in clinical status), an anesthesiologist could adjust or interrupt infusion of the study drug if indicated. Both the enrolled patients and the physicians in charge of POCD evaluation were blinded to the group allocation. Postoperative analgesia: all patients were given sufentanil (100 µg) plus flurbiprofen axetil (200 mg) diluted in 100 mL of physiological saline via patient-controlled intravenous analgesia after surgery.
Study drugs (dexmedetomidine hydrochloride (200 μg/2 mL) and physiological saline (2 mL)) were provided as clear aqueous solutions in identical 3-mL bottles (Jiangsu Heng Rui Medicine, Jiangsu, China). Dexmedetomidine hydrochloride was diluted in 50 mL of physiological saline (i.e., final concentration = 4 μg/mL) before administration. Premedication or sedation was not given to any participant. Patients breathed room air during the entire procedure.
Peripheral intravenous access was secured. Perioperative monitoring comprised electrocardiography, pulse oximetry, and continuous monitoring of arterial blood pressure (via a 20-G catheter inserted into the radial artery). The primary outcome was postoperative cognitive function. The Bispectral Index (BIS), peripheral oxygen saturation (SpO2), rSO2, mean arterial blood pressure (MAP), and heart rate (HR) were secondary outcomes and were acquired at baseline before patients received dexmedetomidine or physiological saline.
After the BIS, rSO2, SpO2, MAP, and HR at baseline had been recorded, patients in the dexmedetomidine group received dexmedetomidine (1 μg/kg), whereas patients in the placebo group received physiological saline (0.25 mL/kg), as recommended in other studies. 20 This dose ensured that patients in both groups received an equal dose of physiological saline regardless of randomization. Drug solutions were prepared by an anesthesiologist, and the volume was maintained at 50 mL. Dexmedetomidine or physiological saline was infused by a micro-pump for 20 minutes before anesthesia induction. Parameters were also monitored after patients regained consciousness. Demographic details (e.g., age, sex, weight, height) were also noted. Data were recorded by an investigator blinded to the study protocol, and only attending anesthetists were aware of the group allocation to guarantee quality control of the study.
Anesthesia method
Patients in both groups underwent thoracoscopic lobectomy under general anesthesia. Before anesthesia induction, patients in both groups received the blind-labeled drugs (dexmedetomidine and normal saline), and the serial number of the drug was recorded. Anesthesia induction: patients in both groups were given 1.5 mg/kg propofol, 0.05 mg/kg midazolam, 0.2 μg/kg sufentanil, and 0.6 mg/kg rocuronium. Maintenance of anesthesia: target-controlled infusion of propofol and remifentanil was performed, and 0.08 µg/kg sufentanil was given before skin suture.
Cognitive function
POCD was measured using the Montreal Cognitive Assessment (MoCA). The MoCA was developed to screen for mild or serious cognitive dysfunction. The MoCA score ranges from 0 to 30 and is divided into seven subsets: visuospatial/executive, attention, abstraction, naming, memory, language, and orientation. A MoCA score ≤25 is considered to denote cognitive impairment. Neuropsychologic evaluations were performed by a trained clinical anesthetist blinded to the study protocol. On the day before the operation, the trained anesthesiologist explained the contents of the MoCA to the patients in detail at the patient’s bedside and helped them to complete the cognitive function scale. Then, the same anesthesiologist evaluated cognitive function on the first, third, and seventh days after the operation.
rSO2 monitoring
The rSO2 level was monitored continuously by near-infrared spectroscopy (NIRS) before anesthesia (EGOS-600A; Enginmed, Beijing, China), 21 which provided continuous, non-invasive, real-time measurement of cerebral oxygenation. 22 This cerebral oximeter has two channels (right and left), which automatically register which sensor is connected, and uses sensor-dependent algorithms for rSO2 calculation. Brain oxygenation was defined as the mean oxygen saturation value of the right and left sides of the brain. The BIS is a commonly used tool to measure the sedation level. A BIS-monitoring electrode (BIS™ Quatro; Aspect Medical Systems, Norwood, MA, USA) was placed on the forehead of the patient after careful cleaning of the skin according to the manufacturer’s instructions. Then, an electrode was attached to a BIS monitor (BIS EEG Vista™; Aspect Medical Systems), which provided continuous, real-time surveillance.
Sample size
In a pilot study of the two groups, the mean MoCA score was 26 in the dexmedetomidine group and 24 in the placebo group. Hence, the sample size needed to be at least 23 for each group to achieve 90% power to detect a difference between the two groups with an alpha of 0.05 using a two-sided, two-sample t-test. Considering the prospects of inadequate cases and exclusion, we planned to enroll 30 participants for each group.
Statistical analyses
Independent Student’s t-tests were used to compare the mean values between the dexmedetomidine and placebo groups at different times. A value of p < 0.05 was considered significant. Statistical analyses were carried out using SAS v9.4 (SAS Institute, Cary, NC, USA).
Results
Sixty-one patients fulfilled the inclusion criteria. Finally, 29 patients in the dexmedetomidine group and 31 patients in the placebo group completed the study, and their data were employed for statistical analyses (Figure 1). The patient characteristics were similar in the two study groups (Table 1). Significant differences between the two groups were not observed for demographic data or the American Society of Anesthesiologists functional class. Changes in the rSO2, BIS, MAP, HR, and SpO2 during pulmonary surgical procedures in the two groups were recorded (Table 2).

CONSORT diagram showing patient allocation.
Demographic parameters of patients.

BMI: body mass index; ASA: American Society of Anesthesiologists; SD: standard deviation.
Changes in BIS, rSO2, MAP, HR, and SpO2 during pulmonary surgery in the two groups.

ap-values between the groups based on a t-test.
bp-values for the changes from baseline to consciousness recovery based on a paired t-test.
BIS: Bispectral Index; rSO2: regional hemoglobin oxygen saturation; MAP: mean arterial blood pressure; HR: heart rate; and SpO2: peripheral oxygen saturation.
The baseline rSO2 was not significantly different between the dexmedetomidine and placebo groups (dexmedetomidine 64.9 ± 2.73% vs. placebo 64.3 ± 3.29%), and no significant difference was found after the patients regained consciousness (dexmedetomidine 63.5 ± 2.52% vs. placebo 64.2 ± 3.22%). Additionally, no difference was observed between the values recorded before and after drug administration. No hypoxia was found among the patients (Table 2).
We assessed the depth of sedation using the BIS. No significant difference was found between the value at baseline and that after regaining consciousness between the two groups (97.0 ± 0.57 vs. 96.9 ± 0.40; 96.8 ± 0.60 vs. 96.9 ± 0.40).
The two groups had a similar hemodynamic profile (Table 2). The HR in the dexmedetomidine group after regaining consciousness was significantly lower than that of the placebo group (66.9 ± 13.29 vs. 76.8 ± 9.46 beats/minute, p < 0.001). The MAP, BIS, and SpO2 were not significantly different between the two groups.
The MoCA score was compared at different times between the dexmedetomidine and placebo groups. The MoCA score was significantly increased in the dexmedetomidine group on postoperative day (POD)1 (26.4 ± 0.73 vs. 25.5 ± 0.96, p < 0.001) and POD3 (27.1 ± 0.79 vs. 26.6 ± 0.80, p = 0.032). No significant difference in MoCA scores was found between the two groups on POD7. The MoCA score of the two groups reached the baseline value on POD7 (Table 3).
Change in the MoCA score, which was used to assess mild cognitive impairment in the two groups.
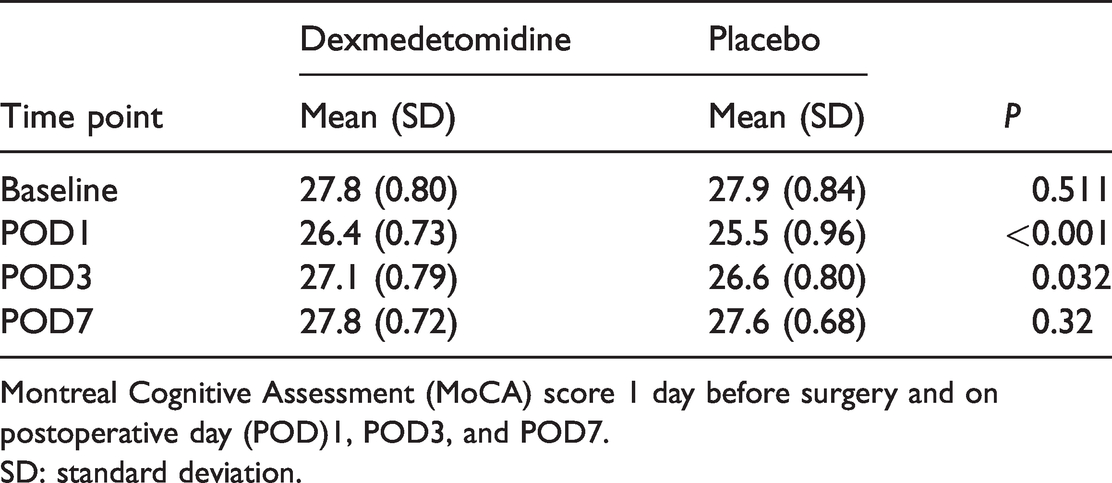
Montreal Cognitive Assessment (MoCA) score 1 day before surgery and on postoperative day (POD)1, POD3, and POD7.
SD: standard deviation.
In a comparison of the detailed MoCA score categories (Table 4), the attention score was increased in the dexmedetomidine group on POD1 (5.03 ± 0.50 vs. 4.58 ± 0.56, p < 0.05) and POD3 (5.14 ± 0.44 vs. 4.96 ± 0.48, p < 0.05) and was decreased in the placebo group on POD1 and POD3 compared with the preoperative baseline value. We found a significant decrease in the memory score in both groups without intergroup differences, and the memory score had recovered on POD7. The orientation score was also increased in the dexmedetomidine group on POD1 (5.27 ± 0.70 vs. 4.23 ± 0.72, p < 0.05) and POD3 (5.48 ± 0.57 vs. 5.06 ± 0.51, p < 0.05).
Comparison of detailed categories of the MoCA score.

D: Dexmedetomidine group; P: placebo group; MoCA: Montreal Cognitive Assessment; T0: preoperative; T1: postoperative day 1; T3: postoperative day 3; T7: postoperative day 7.
asignificant difference between the dexmedetomidine group and placebo group, p < 0.05.
bsignificant difference between the postoperative Tx and T0 (preoperative baseline value), p < 0.05.
Discussion
Our study indicated that Intravenous administration of dexmedetomidine (1 µg/kg) within 20 minutes before anesthesia can significantly improve the MoCA score and reduce the risk of POCD on the first and third days after surgery. Some studies have shown that dexmedetomidine causes a dose-related reduction in CBF and CMR, but our study found no effect of dexmedetomidine on rSO2,17,23 which suggested that concerns about a decline in rSO2 resulting in POCD could be unnecessary.
To evaluate the effect of dexmedetomidine on rSO2, it is important to obtain the precise rSO2 value. Cerebral oxygen saturation using NIRS has been widely used to monitor relative changes in the oxidative status of the brain. However, NIRS data require careful interpretation because rSO2 can be affected by age, hemoglobin concentration, bleeding, and transfusion. 20 We controlled the variables that influence rSO2, including the type of anesthetic used and oxygen inhalation, which increased the robustness of our findings.
The second key finding of the present study was that the reduction in rSO2 in the dexmedetomidine group was not significantly lower than that of the placebo group. Dexmedetomidine can act directly on selective α2-adrenoreceptors in cerebral vessels, which can cause vasoconstriction and decrease CBF. 24 Gao and colleagues found that dexmedetomidine (1 µg/kg) administration for 15 minutes before the surgical incision decreased cerebral oxygen saturation during single-lung ventilation. 25 A series of studies demonstrated that a decline in rSO2 was significantly correlated with POCD as assessed by the Mini-Mental State Examination.26,27 Casati and co workers designed a multicenter, randomized, prospective trial to estimate the efficacy of using rSO2 monitoring during abdominal surgery. 28 They found that cerebral desaturation occurred intraoperatively and was positively correlated with a decline in cognitive status; however, this was not observed during pulmonary surgery. However, we demonstrated that rSO2 does not decrease in pulmonary surgery, even after a long duration of single-lung ventilation, which suggested that dexmedetomidine does not appear to play a negative role in POCD after pulmonary surgery.
The MoCA score was calculated on POD1, POD3, and POD7. The MoCA scores in the dexmedetomidine group were significantly higher than those in the placebo group on POD1 and POD3. Additionally, the prevalence of POCD, which was measured using the MoCA, was significantly lower in the dexmedetomidine group than that in the placebo group on POD1 and POD3. Solanki and Goyal showed that dexmedetomidine can reduce the incidence of nerve injury after transient ischemia in rats. 29 However, the specific mechanisms of the organ-protective effects of dexmedetomidine are still unclear. Patients receiving dexmedetomidine were less likely to experience delirium than patients receiving midazolam, propofol, or remifentanil. It has been suggested that intraoperative dexmedetomidine treatment can significantly reduce the prevalence of delirium and POCD in both normal older patients and those with amnestic mild cognitive impairment, which suggests that it may be an effective option to prevent POCD. In our study, attention and orientation were significantly different between the two groups. This finding is in accordance with the observed reduction in cognitive dysfunction within 1 week when patients stayed in the ICU. Hence, we propose that dexmedetomidine is beneficial for patients likely to be admitted to the ICU postoperatively because of the increased incidence of perioperative cognitive dysfunction.
Dexmedetomidine is an α2-adrenoreceptor agonist that calms patients, reduces pain, and inhibits sympathetic nervous system activity but can also inhibit the inflammatory response and stress response, reduce neuronal toxicity and apoptosis, and promote brain protection through synapse formation and neurotrophic nutrition. 30 Zeng and colleagues showed that dexmedetomidine can affect proinflammatory factor expression and reduce the inflammatory response in blood vessels by activating the corresponding signaling pathways. 31 We suggest that the protective effect of dexmedetomidine on cerebral blood vessels may also have been manifested as a reduction of the level of proinflammatory factors in cerebral blood vessels, which thereby reduced the level of inflammation in the brain tissues of patients.
Our study had three main limitations. First, we did not measure cerebral autoregulation, alterations in which may have affected the brain’s response to dexmedetomidine. Second, because it would have involved collection of additional blood samples, the serum concentration of dexmedetomidine was not monitored. Third, the patients enrolled in this study had a specific cultural background and could accurately read the informed consent form, which led to the exclusion of some patients from the trial, and the lack of hierarchical analysis is also a limitation of the study. Further large-scale multicenter studies should be designed to compensate for this limitation.
Conclusion
We showed, for the first time, that a loading dose of dexmedetomidine (1.0 µg/kg) could reduce the risk of POCD and might not decrease rSO2. Hence, dexmedetomidine could be employed in pulmonary surgical procedures, especially for older patients with a high risk of cognitive dysfunction.
Footnotes
Acknowledgements
We thank Oxford Science Editing for editorial support.
Availability of data and materials
We updated the paper at research square (DOI:10.21203/rs.3.rs-152369/v1).
Author contributions
Study design: Tao Liu, Fang-Chao Liu, Yu Xia, Fei Wang
Manuscript draft: Tao Liu, Fang-Chao Liu
Data analysis: Tao Liu, Tai-Jun Luo, Bin Chen, Chun Wang, Guang-Kuo Gao
Manuscript approval: Wen-Ting Zhai, Wei Liu
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
