Abstract
Objectives
To explore the impacts of 1,25-dihydroxyvitamin D3 (1,25(OH)2D3) on the proliferation, migration, and monocyte chemoattractant protein-1 (MCP-1) secretion of vascular smooth muscle cells (VSMCs) in a high glucose environment and its possible mechanism.
Methods
We extracted VSMCs from the thoracic aorta of a male Sprague–Dawley rats before culturing them in a 25-mM glucose-containing medium in the presence or absence of 1,25(OH)2D3 (10−9 –10−7 M). Cell proliferation was determined by bromodeoxyuridine incorporation assays. Subsequently, cell migratory capacity was examined by performing a transwell assay. An enzyme-linked immunosorbent assay was conducted to assess MCP-1 levels. Protein levels of matrix metalloproteinase-9 (MMP-9), mitogen-activated protein kinases (MAPKs), cyclin D1, and phosphorylated MAPKs were determined by immunoblotting.
Results
1,25(OH)2D3 significantly suppressed the proliferation, migration, and MCP-1 secretion of VSMCs mediated by high glucose in a dose-dependent manner, diminished the enhanced protein expression of MMP-9 and cyclin D1, and attenuated MAPK phosphorylation. The p38 inhibitor SB203580 and ERK1/2 inhibitor PD98059 suppressed high glucose-mediated upregulation of MMP-9 and cyclin D1 protein expression and MCP-1 secretion, respectively.
Conclusions
1,25(OH)2D3 ameliorates high glucose-mediated proliferation, migration, and MCP-1 secretion of VSMCs by inhibiting MAPK phosphorylation, implying a potential therapeutic approach using 1,25(OH)2D3 for diabetic macrovascular complications.
Keywords
Introduction
The number of people suffering from diabetes is increasing worldwide. Diabetic macrovascular complications, with atherosclerosis as a common pathological process, are the major contributors to death in patients with diabetes. 1 Vascular smooth muscle cells (VSMCs), the predominant cellular constituents of the medial layers of the arterial wall, are reportedly involved in atherosclerosis through phenotypic transformation. In response to atherogenic stimuli, VSMCs undergo transformation from the “contractile” to the “synthetic” phenotype, proliferate, migrate from the media to the intima, and generate extracellular matrix proteins, thereby contributing to intimal thickening at predisposed sites.2,3 Additionally, VSMCs with a synthetic phenotype secret some pro-inflammatory cytokines, including interleukin-6, monocyte chemoattractant protein-1 (MCP-1), and tumor necrosis factor-α, augmenting VSMC migration into the intima and monocyte-endothelial interactions and further accelerating the progression of atherosclerosis.4,5
Multiple mechanisms have been demonstrated to mediate the increased ability of VSMCs to proliferate and migrate. Among them, the activation of mitogen-activated protein kinases (MAPKs), including extracellular signal-regulated kinases (ERKs), c-Jun N-terminal kinases (JNKs), and p38 mitogen-activated protein kinases (p38 MAPKs), is considered one of the most crucial signaling pathways. In general, growth factors and mitogens activate ERKs, whereas cellular stressors and pro-inflammatory cytokines stimulate JNKs and p38 MAPKs. 6 Several studies have demonstrated that high glucose is causally related to the increased proliferation, migration, and MCP-1 secretion of VSMCs via the stimulation of MAPKs, especially p38 MAPK and ERK1/2, which play a role in the pathological process of diabetes-related vascular diseases and may be used as promising treatment targets.7–9
In recent years, considerable experimental and epidemiological studies have suggested that vitamin D deficiency is closely related to an elevated incidence and severity of cardiovascular diseases, and vitamin D supplementation prevents atherosclerosis through a variety of mechanisms.10–12 Our earlier
Therefore, the purpose of the present research was to explore the impacts of 1,25(OH)2D3 on the proliferation, migration, and MCP-1 secretion of aortic smooth muscle cells in rats under a high glucose environment and examine the role of MAPKs in this regulatory process.
Materials and methods
Cell culture
In accordance with the Institutional Guidelines and Guide for the Care and Use of Laboratory Animals (NIH Publication No.85-23, 1996 revised), Sprague–Dawley rats were housed in the specific pathogen-free grade environment of the Putian University Experiment Animal Center. Following approval from Putian University’s Animal Care and Use Committee, we sacrificed 8-week-old rats (200–250 g) for VSMC extraction. Enzymatic digestion was performed to extract VSMCs from thoracic aortas, as reported previously. 20 In addition, the experiment used cells at passages 3 to 6 only. We observed that over 98% of cells had the typical “hill and valley” structure when subjected to staining with a smooth muscle α-actin antibody (DAKO, Cambridgeshire, UK; 1:1000 dilution). Cells that reached 80% to 95% confluence were subjected to starvation for 24 hours (0.3% fetal bovine serum [FBS]). Next, all cells were classified into multiple groups, including the control (normal glucose 5.5 mM) group, high glucose (25 mM) group, and high glucose plus different doses of 1,25(OH)2D3 (10−9 M, 10−8 M, and 10−7 M; Calbiochem, San Diego, CA, USA) groups. The JNK inhibitor SP600125, ERK1/2 inhibitor PD98059, and p38 MAPK inhibitor SB203580 were purchased from Sigma, St. Louis, MO, USA. For MAPK inhibitor experiments, cells were treated with SP600125 (5 μM), PD98059 (200 μM), and SB203580 (5 μM) for 45 minutes.
Bromodeoxyuridine (BrdU) incorporation assay
The BrdU incorporation assay was performed to measure DNA synthesis in VSMCs to reflect the proliferation level in accordance with a previously discussed method.
21
Briefly, we used gelatin-coated 96-well plates covered with complete medium and plated VSMCs at a density of 2000 cells/well. To attain synchronized growth arrest, the medium was removed after 24 hours and replaced with a serum-free medium for an additional 48 hours. After 45 minutes of cell pretreatment with 1,25(OH)2D
Transwell assay
Cell migration was determined using the modified Boyden microporous membrane double groove technique. 22 We seeded VSMCs in the upper wells at a density of 1 × 105 cells and cultured them in 200 µL of serum-free Dulbecco’s Modified Eagle’s Medium (DMEM; Sigma, St. Louis, MO, USA) containing 1% FBS. In contrast, the lower wells were supplied with DMEM containing 10% FBS. In the upper wells, a high glucose-rich medium along with the experimental reagents was introduced. Then, we fixed the cell migration membrane with methanol after incubation for 24 hours at 37°C in a 5% CO2 incubator. Afterward, toluidine blue was applied to stain cells on the bottom side. The proportion of blue-stained cells in each of the five small circular sections of the migratory membrane was estimated at random (10 × objective lense, n = 6).
MCP-1 level assessment using an enzyme-linked immunosorbent assay (ELISA)
We used a commercially accessible Rat MCP-1 ELISA development Kit (PeproTech, East Windsor, NJ, USA) to assess the concentration of MCP-1 present in the cell supernatants. The assays were implemented following the instructions provided by the manufacturer. All samples were evaluated in triplicate to ensure accuracy.
Immunoblotting
After being extracted, VSMCs were lysed using a lysis buffer. The separation of total protein (50 μg in each well) was performed by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis, followed by electrophoretic transfer to nitrocellulose membranes. Membranes were blocked using 5% dried milk in tris-buffered saline (TBS) (150 mM NaCl, 25 mM Tris-HCl [pH 7.5]) containing 0.05% Tween-20 (TBST) for 3 hours at ambient temperature. Then, we rinsed the membrane three times in TBST and incubated them for another 3 hours at ambient temperature with primary antibodies to cyclin D1 (mouse, monoclonal; Santa Cruz, Paso Robles, CA, USA), matrix metalloproteinase 9 (MMP-9) (rabbit, polyclonal; Servicebio, Wuhan, China), phospho-JNK MAPK (rabbit, polyclonal; Cell Signaling Technology, Danvers, MA, USA), JNK MAPK (Cell Signaling Technology), phospho-ERK1/2 MAPK (mouse, monoclonal; Cell Signaling Technology), ERK1/2 MAPK (Cell Signaling Technology), phospho-p38 MAPK (rabbit, monoclonal; Cell Signaling Technology), p38 MAPK (Cell Signaling Technology), and β-actin (mouse, monoclonal; Santa Cruz). After rinsing the membranes five times in TBST and twice in TBS, they were subjected to 1 hour of incubation at ambient temperature in anti-rabbit and anti-mouse horseradish peroxidase-conjugated secondary antibodies (Abcam, Cambridge, UK). The samples were rinsed twice in TBST, and the stained proteins were observed with a high-performance chemiluminescence film using an electrochemiluminescence substrate (Invitrogen, Carlsbad, CA, USA). Densitometry was conducted with image analysis tools (Scion Image Software; Scion Corp., Frederick, MD, USA) to determine the intensity of the bands. The protein expression of interest was standardized to the control group.
Statistical analysis
Results were presented as the mean ± standard deviation and compared by a one-way factorial ANOVA followed by a post-hoc Scheffé comparison test. A
Results
1,25(OH)2D3 suppressed high glucose-mediated increased VSMC proliferation
To evaluate how 1,25(OH)2D3 influences the capability of VSMCs to proliferate under high glucose conditions, VSMCs were pretreated with 1,25(OH)2D

1,25(OH)2D3 suppressed the enhanced capacity of VSMCs to proliferate under high glucose settings in a dose-dependent manner. After treatment with HG, mannitol, or different concentrations of 1,25(OH)2D3, VSMC proliferation was assessed by a bromodeoxyuridine assay. Results are reported as the mean ± SD, n = 8, *
1,25(OH)2D3 inhibited high glucose-mediated increased VSMC migration
A transwell assay was performed to determine the strength of VSMC migration. The findings demonstrated a remarkably enhanced migratory capacity of VSMCs exposed to high glucose compared with those subjected to normal glucose (

1,25(OH)2D3 suppressed the increased number of migrated VSMCs under HG in a dose-dependent manner. (a) Representative microscope image of migrated cells treated with the control, HG, and different concentrations of 1,25(OH)2D3 in transwell assays (100 × magnification). Cells were stained with toluidine blue. and (b) Quantification of cell migration. Results are presented as the mean ± SD, n = 5, *
1,25(OH)2D3 inhibited high glucose-mediated increased VSMC secretion of MCP-1
The expression level of MCP-1 in the supernatant of cultured VSMCs was detected by ELISA. The results showed that VSMCs exposed to high glucose produced significantly higher MCP-1 levels than those exposed to normal glucose (

1,25(OH)2D3 dose-dependently suppressed the increased MCP-1 secretion by VSMCs under HG conditions. MCP-1 levels were determined by an enzyme-linked immunosorbent assay. Results are presented as the mean ± SD, n = 4, *
1,25(OH)2D3 attenuated high glucose-mediated upregulation of cyclin D1 and MMP-9 protein expression in VSMCs
Cyclin D1 is a crucial protein in the progression of the cell cycle, and its expression is increased in response to mitogen stimulation.23,24 MMPs, specifically MMP-9, degrade the extracellular matrix and are required for VSMC migration.
25
To thoroughly assess the impacts of 1,25(OH)2D3 on the capacity of VSMCs to proliferate and migrate and its underlying mechanism in a high glucose setting, we further explored how 1,25(OH)2D3 affects the proliferation- and migration-related proteins cyclin D1 and MMP-9, respectively. The immunoblotting findings demonstrated a remarkable increase in the protein expression levels of cyclin D1 and MMP-9 in VSMCs exposed to high glucose compared with the levels in cells subjected to normal glucose (
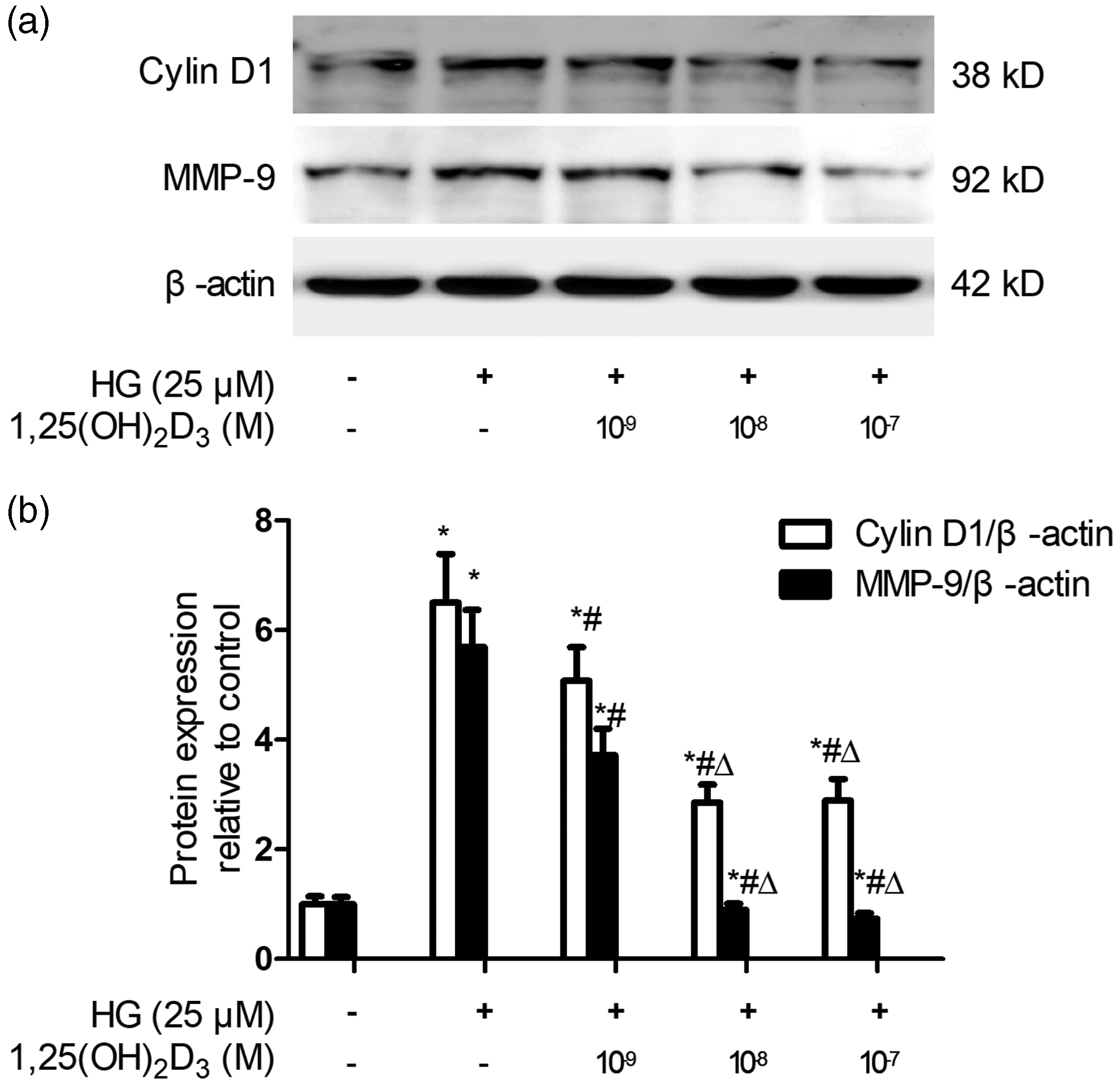
1,25(OH)2D3 suppressed the upregulated cyclin D1 and MMP-9 protein expression of VSMCs under HG conditions in a dose-dependent manner. (a) Representative immunoblot. β-actin was used as the internal control. and (b) Quantitative examination of blot band intensity. Data are presented as the mean ± SD, n = 4, *
1,25(OH)2D3 inhibited high glucose-mediated activation of MAPKs in VSMCs
Previous studies have shown that high glucose promotes VSMC proliferation and migration through the activation of MAPKs. Therefore, we further explored whether 1,25(OH)2D3 might counteract the adverse impacts of high glucose on VSMCs by inhibiting MAPK activation. The immunoblotting findings demonstrated that exposing VSMCs to high glucose resulted in enhanced JNK, ERK1/2, and p38 MAPK protein phosphorylation after 2 hours of treatment, without affecting their total levels, and 1,25(OH)2D3 (10−9–10−7 M) pretreatment for 45 minutes alleviated the increased JNK, p38 MAPK, and ERK1/2 phosphorylation mediated by high glucose in a concentration-dependent manner (

1,25(OH)2D3 suppressed the increased MAPK phosphorylation in VSMCs under HG in a dose-dependent manner. (a) Representative immunoblot. β-actin was used as the internal control. and (b) Quantitative analysis of blot band intensity. Results are presented as the mean ± SD, n = 4, *
ERK1/2 antagonist PD98059 alleviated high glucose-mediated increased proliferative and migratory capacity of VSMCs
To thoroughly clarify whether the suppressive impact of 1,25(OH)2D3 on MAPK phosphorylation mediates its protective effects against the high glucose-induced increased capacity of VSMCs to proliferate and migrate, we observed the influence of the JNK inhibitor SP600125, ERK1/2 inhibitor PD98059, and p38 MAPK inhibitor SB203580 on the proliferation and cyclin D1 and MMP-9 protein expression of VSMCs under hyperglycemic conditions. As shown in Figure 6a, PD98059 but not SP600125 and SB203580 abrogated the increased VSMC proliferation under the hyperglycemic environment (

Impacts of MAPK inhibitors on HG-mediated increased VSMC proliferation, MAPK phosphorylation, and cyclin D1 and MMP-9 protein expression. (a) VSMCs were treated with HG, mannitol, or the indicated inhibitors, and proliferation was assessed with a bromodeoxyuridine assay. The ERK1/2 inhibitor PD98059, but not the JNK inhibitor SP600125 or p38 MAPK inhibitor SB203580, attenuated HG-mediated increased VSMC proliferation. (b) The JNK inhibitor SP600125 inhibited the phosphorylation of JNK but did not affect cyclin D1 and MMP-9 protein expression. (c) The ERK1/2 inhibitor PD98059 suppressed ERK1/2 phosphorylation and cyclin D1 and MMP-9 protein expression. and (d) The p38 MAPK inhibitor SB203580 inhibited the phosphorylation of p38 MAPK but did not affect cyclin D1 and MMP-9 protein expression. The left panel depicts a representative blot, and the right panel depicts the quantitative analysis of blot band intensity. β-actin was used as the internal control. Results are represented as the mean ± SD, n = 4, *
p38 MAPK inhibitor SB203580 alleviated high glucose-mediated increased secretion of MCP-1 by VSMCs
Similarly, we observed the impacts of the ERK1/2 inhibitor PD98059, JNK inhibitor SP600125, and p38 MAPK inhibitor SB203580 on MCP-1 secretion by VSMCs under hyperglycemic conditions. The findings illustrated that the p38 MAPK inhibitor SB203580 (5 μM) significantly alleviated the elevated MCP-1 expression level in the supernatant of high glucose-cultured VSMCs (

Impact of MAPK inhibitors on HG-mediated increased MCP-1 secretion by VSMCs. VSMCs were treated with or without HG in the presence or absence of the indicated inhibitors, and the MCP-1 level in the supernatant was detected by an enzyme-linked immunosorbent assay. SP600125, PD98059, and SB203580 indicate inhibitors of JNK, ERK1/2, and p38 MAPK, respectively. Data are presented as the mean ± SD, n = 4, *
Discussion
As one of the major cellular constituents of the arterial wall, VSMCs play a fundamental role in the development of atherosclerosis through abnormal proliferation, migration, and MCP-1 secretion in response to various growth factors and stress signals. In this
MAPKs belong to a family of ubiquitous proline-directed, protein-serine/threonine kinases that play a role in mediating cellular responses to diverse stimuli, including osmotic stress, heat shock, proinflammatory cytokines, and mitogens, thereby modulating cellular activities, such as proliferation, differentiation, gene expression, apoptosis, and inflammation. 26 The most extensively investigated MAPKs are ERK1/2, p38 MAPK, and JNK. 27 Previous research reports suggested that subjecting VSMCs to high glucose markedly enhances the activation of all three members of the MAPK family compared with normal glucose, increases VSMC growth potential, and promotes MCP-1 release from VSMCs, culminating in atherosclerosis lesion development.7,9,28 Similarly, our results showed that high glucose induced p38 MAPK, JNK, and ERK1/2 phosphorylation and upregulated cyclin D1 and MMP-9 protein expression, eventually promoting the proliferation, migration, and MCP-1 secretion of VSMCs. Furthermore, using specific MAPK inhibitors, our results demonstrated that high glucose promotes VSMC proliferation, migration, and MCP-1 secretion through activating ERK1/2 and p38 MAPK phosphorylation, suggesting that high glucose not only acts as a growth factor but also as a stress signal for VSMC phenotype switching. However, He et al. illustrated that subjecting VSMCs to high glucose only results in the activation of ERK1/2 MAPK, not JNK and p38 MAPK. 29 Li et al. discovered that the stimulation of the MAPK signaling pathway in VSMCs in response to elevated glucose levels differed depending on the individual’s age. 30 Therefore, we speculate that the stimulation of MAPK signaling pathways in VSMCs exposed to high glucose levels might be modulated by age or other environmental variables.
In recent years, increasing evidence has suggested that vitamin D has an anti-atherosclerosis effect in addition to its regulatory effect on calcium homeostasis. 31 Our previous study also found that vitamin D improves diabetic-associated endothelial dysfunction and delays the progression of atherosclerosis by antagonizing high glucose-mediated oxidative stress.13–15 However, whether vitamin D may reverse the detrimental impact of high glucose on VSMCs has not been comprehensively elucidated. In the present research, we discovered that 1,25(OH)2D3, the biologically active metabolite of vitamin D, antagonized the high glucose-mediated increased proliferation, migration, and MCP-1 secretion of VSMCs isolated from the rat thoracic aorta, and this influence was associated with the suppression of ERK1/2 and p38 MAPK phosphorylation. In rat aortic smooth muscle, it has been demonstrated that high glucose activates p38 MAPK through a protein kinase C (PKC)-delta isoform-dependent pathway or a PKC-independent pathway. 32 Whether the suppressive impacts of 1,25(OH)2D3 on ERK1/2 and p38 MAPK phosphorylation under hyperglycemic conditions correlate with PKC needs to be elucidated in the future.
Several researchers have focused on the impacts of 1,25(OH)2D3 on the biological behavior of VSMCs under conditions other than high glucose, consistently supporting an anti-inflammatory potential,12,33 but the conclusions regarding the effects on the proliferation and migration of VSMCs have been inconsistent. Carthy et al. showed a dose-dependent (10−12–10−7 M) suppression of basal and epidermal growth factor-stimulated VSMC growth by 1,25(OH)2D3. 17 Chen et al. documented that 1,25(OH)2D3 (10−12–10−8 M) had no effect on the basal proliferation of VSMCs but inhibited endothelin-induced VSMC proliferation without affecting endothelin-dependent ERK activation. 18 However, Tukaj et al. illustrated that 1,25(OH)2D3 at supra-physiological doses (10−9–10−7 M) increased the proliferative and migratory activities of VSMCs while significantly attenuating the production of pro-inflammatory cytokines.19,34 Cardus et al. found that 1,25(OH)2D3 (5 × 10−9–10−7 M) dose-dependently stimulated VSMC proliferation by elevating the expression level of vascular endothelial growth factor. 16 Rebsamen et al. also demonstrated that 1,25(OH)2D3 (10−10–10−7 M) induced the migration of VSMCs via a phosphoinositide 3-kinase pathway independent of gene transcription. 35 The inconsistency in these results may be associated with the different intervention doses of 1,25(OH)2D3 and various cultural environments of VSMCs, which need to be further evaluated in future studies.
There are two limitations to our study. First, the doses of 1,25(OH)2D3 used in our experiments were rather higher than the normal serum 1,25(OH)2D3 level and too high to be realistically achieved in human serum without causing toxic effects (e.g., hypercalcemia). Therefore, a new less-calcemic vitamin D analog may be a better alternative in future research. Second, we only observed the impacts of 1,25(OH)2D3 on MAPK activation at a single time point of 2 hours, which has generally been considered a rapid nongenomic response requiring dynamic detection at multiple time points for a comprehensive assessment.
Conclusions
The present research observed that vitamin D antagonized the abnormal proliferation, migration, and secretion of MCP-1 of VSMCs induced by high glucose, and this impact was associated with the suppression of ERK1/2 and p38 MAPK phosphorylation. At the cellular level, our study further suggests the potential of using vitamin D as a therapeutic agent for future diabetic macroangiopathy.
Footnotes
Acknowledgements
The authors express their gratitude to Changsheng Xu for his technical assistance.
Declaration of conflicting interest
The authors declare that they have no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Youth Program of the National Natural Science Foundation of China (Grant no. 81800278), Young/Middle-aged Talent Cultivation Project of Fujian Provincial Health and Family Planning Commission (Grant no. 2018-ZQN-79), and Youth Project of Fujian Provincial Health and Family Planning Commission (Grant no. 2017-1-93) to LIN Liming, Natural Science Foundation of Fujian Province to XU Kaizu (Grant no. 2020J011259), and Natural Science Foundation of Fujian Province to WU Ying (Grant no. 2021J01122508).
Author contributions
The study was conceptualized and developed by Liming Lin. The tests were carried out by Meifang Wu and Ying Wu. XU Kaizu contributed to conducting the data analyses and drafting the article. The finalized manuscript was approved by all authors.
