Abstract
Pulmonary diseases often cause significant health issues and nutritional disorders. Weight loss and malnutrition are related to the severity of obstructive disorders. Therefore, patients with such conditions often experience low nutritional energy. Acetonemic vomiting is caused by acetonemic syndrome. Previously, it was believe that acetonemic vomiting was observed only in childhood. However, it was recently suggested that acetonemic vomiting can also occur in adults. It is also considered that acetonemic vomiting can occur in subjects with low body weight because stored carbohydrate levels are reduced and fats are mainly used for energy. Consequently, large amounts of acetone are produced, ultimately resulting in nausea and vomiting. In this study, we report a case of adult acetonemic vomiting complicated by low body weight in a subject with Mycobacterium avium complex pulmonary disease.
Keywords
Introduction
Pulmonary diseases often cause significant health issues and nutritional disorders. In addition, nutritional disorders are commonly complicated by various pulmonary diseases, such as chronic obstructive pulmonary disease, 1 pulmonary tuberculosis, 2 and Mycobacterium avium complex (MAC) pulmonary disease. 3 It has been noted that the complications of nutritional disorders have negative effects on the condition and prognosis of chronic respiratory failure. Weight loss is related to the severity of obstructive disorders, and subjects with a body mass index (BMI) of less than 20 kg/m2 comprise approximately 30% of patients with obstructive disorders.
Acetonemic vomiting is caused by acetonemic syndrome, which is also called acetonemic vomiting syndrome and cyclic vomiting syndrome. Acetonemic vomiting is typically observed in childhood, and the median age of onset of symptoms ranges from 3.5 to 7 years.4–6 However, it was recently indicated that acetonemic vomiting can occur at any age from infancy to adulthood. 7 In adults, acetonemic vomiting can develop when the purine or protein balance is disturbed and ketone body levels are increased.
In this study, we report a case of adult acetonemic vomiting complicated by low body weight in a subject with MAC pulmonary disease. This case report clearly indicates that we should be aware of the possibility of acetonemic vomiting in both children and adults, especially in underweight subjects with repeated nausea and/or vomiting.
Case presentation
A 79-year-old Japanese woman was brought to the emergency room of Kawasaki Medical School General Medical Center (Okayama, Japan) with symptoms of nausea and vomiting. She was diagnosed with MAC pulmonary disease at 52 years old, and she was positive for MAC on polymerase chain reaction at 69 and DNA–DNA hybridization at 71. She was diagnosed with rheumatic polymyalgia at 73 and rheumatoid arthritis at 75, and she was taking 100 mg/day pregabalin and 2.5 to 7.5 mg/day prednisolone. Before admission, she was taking 150 mg/day rifampicin, 250 mg/day ethambutol, and 400 mg/day clarithromycin to treat MAC pulmonary disease. Chest X-ray (Figure 1a) revealed multiple small nodular shadows, and chest computed tomography (Figure 1b) disclosed consolidation including bronchiectasis. Her height, body weight, and BMI were 155.4 cm, 33.5 kg, and 13.9 kg/m2, respectively. Her vital signs were as follows: temperature, 36.8°C; blood pressure, 118/62 mmHg; heart rate, 84 beats/minute; and oxygen saturation, 98% (room air). Table 1 presents the patient’s laboratory data in the emergency room. Her infection markers were markedly elevated. In addition, her nutrition-associated data revealed malnutrition and mild hypoglycemia. Moreover, her urinary test disclosed elevated urinary and serum ketone body concentrations.
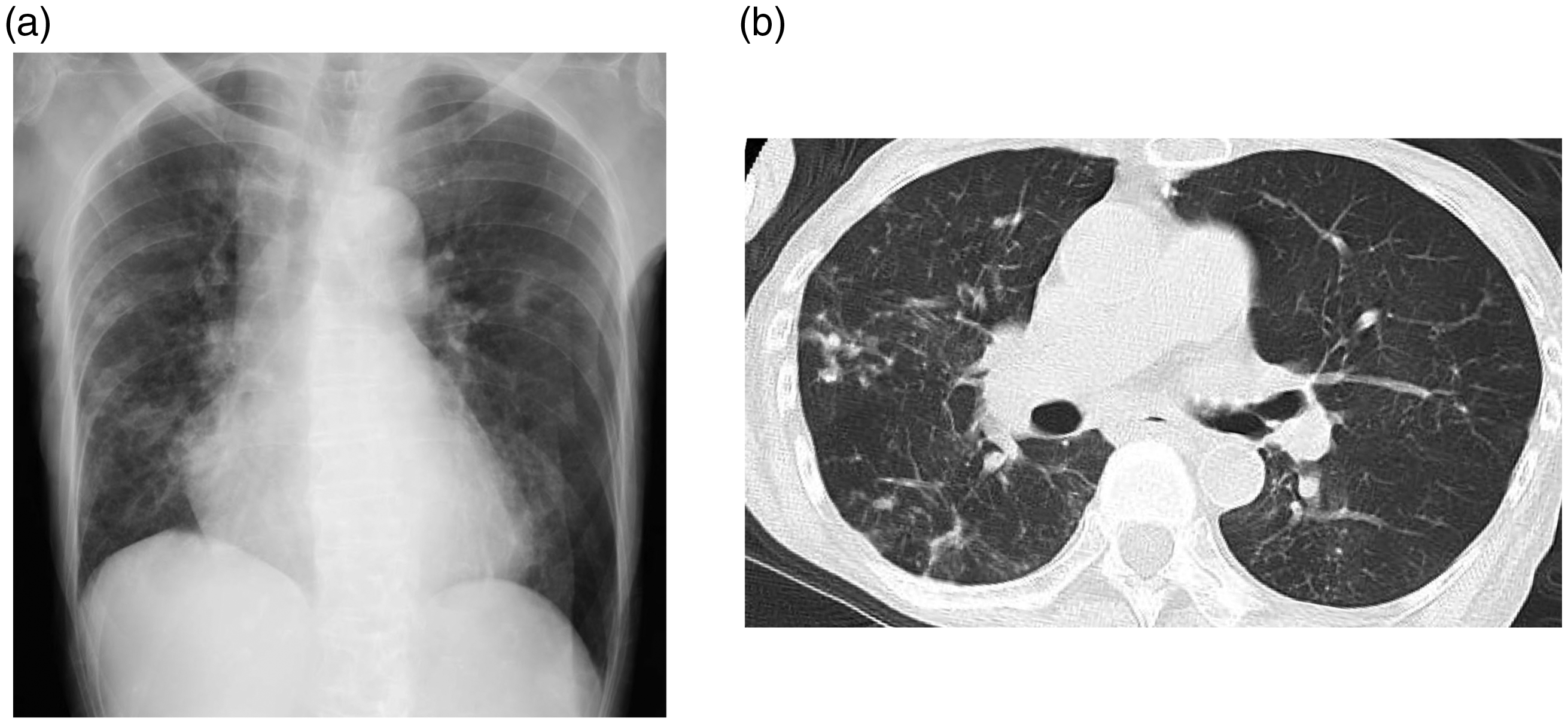
Imaging findings. (a) Chest X-ray revealed multiple small nodular shadows and (b) Chest computed tomography revealed consolidation including bronchiectasis.
Laboratory data in an emergency room in this subject.

AST, aspartate aminotransferase; ALT, alanine aminotransferase; LDH, lactate dehydrogenase; ALP, alkaline phosphatase; γ-GTP, γ-glutamyl transpeptidase; BUN, blood urea nitrogen; CRP, C-reactive protein.
After admission, we started antibiotic therapy (9.0 g/day tazobactam/piperacillin for 3 days) even though pathogenic bacteria were not detected in sputum and blood. Her fever was improved the next day (peak temperature, 37.5°C), and her infection markers were decreased (white blood cell [WBC] count, 5260/μL; C-reactive protein [CRP], 5.42 mg/dL). Surprisingly, she had been hospitalized four times within 1 year. Therefore, we investigated the details of her laboratory data during hospitalization. Her body weight decreased to less than 35.0 kg approximately 1 year ago. Her main complaints during each hospitalization were nausea and vomiting, and her condition was complicated by MAC pulmonary disease. Moreover, during each admission, she exhibited mild hypoglycemia and elevated urinary ketone bodies (Figure 2). Although we examined the possible reasons of repeated nausea and vomiting with gastroscopy after the third admission, we failed to detect any abnormality in the upper gastrointestinal tract. There were no findings of malignancy in the imaging examination and tumor marker assessment. Based on such findings, we finally diagnosed her with acetonemic vomiting complicated by low body weight. In addition, her low body weight was caused by MAC pulmonary disease. We started education and diet therapy for the patient (total, 1500 kcal/day [approximately 30 kcal per ideal body weight] without skipping meals including carbohydrates). After then, her symptoms of nausea and vomiting disappeared without anti-nausea drug therapy after admission, and she was not hospitalized for 1 year.

Time course of clinical parameters in this subject. This patient was hospitalized five times including this admission for only 1 year. She was malnourished (low albumin and cholinesterase levels). In addition, during each hospitalization, she exhibited nausea and vomiting together with hypoglycemia and ketosis.
Discussion
Pulmonary diseases including MAC pulmonary disease often cause low body weight and malnutrition.1–3 Therefore, patients with these diseases often experience low nutritional energy. Conversely, acetonemic vomiting is likely to occur in subjects with low body weight because stored carbohydrate levels are reduced and fats are mainly used for energy. Consequently, large amounts of acetone are produced, leading to nausea and vomiting.
Acetonemic vomiting is induced in subjects with acetonemic syndrome, especially during childhood.4–6 Being related to protein catabolism, carbohydrates and fats are associated with the tricarboxylic acid cycle under normal physiological conditions. Ketosis is mainly induced by fasting or the excessive consumption of protein and fatty foods. In addition, under relative or absolute carbohydrate deficiency, lipolysis is activated to provide supplemental energy. Consequently, ketone bodies are synthesized from acetyl coenzyme A in the body with increased lipolysis. However, it was recently demonstrated that acetonemic vomiting can arise even in adults when the purine or protein balance is disturbed, and ketone body levels are increased. 7 Our patient experienced MAC pulmonary disease for a long period, and this condition could easily lead to energy deficiency with low body weight. Therefore, we believe that she developed relapsing acetonemic vomiting despite being an adult. The current case report revealed that when subjects with pulmonary disease and low body weight display nausea and vomiting, we should consider the possibility of acetonemic vomiting even in adulthood.
This case report had several strengths. First, this report clearly demonstrated that when subjects with pulmonary disease and low body weight exhibit nausea and vomiting, we should consider the possibility of acetonemic vomiting, even in adults. Second, this report clearly revealed that diet therapy without skipping meals including carbohydrates is extremely important for mitigating the symptoms of nausea and vomiting in adults with acetonemic vomiting induced by low body weight and pulmonary diseases. Conversely, several limitations should be considered. She was taking 5 to 10 mg/day prednisolone for the treatment of MAC pulmonary disease for at least for 5 years. Therefore, it is possible that she had relative adrenal insufficiency. In general, many cases of relative adrenal insufficiency are complicated by fatigue, loss of appetite, weight loss, hypotension and hyponatremia, hypoglycemia, and/or relative hypereosinophilia. Our patient displayed nausea, vomiting, loss of appetite, and weight loss during prednisolone therapy. In addition, she had mild hypoglycemia while receiving a dose of 40 to 70 mg/dL. However, she did not experience hypotension and hyponatremia. Therefore, we did not consider the possibility that she had relative adrenal insufficiency, and we did not measure adrenocorticotropic hormone and cortisol levels. Based on these findings, we considered that her hypoglycemia was mainly caused by low body weight. Moreover, she was treated with antibiotics repeatedly because her WBC count and CRP levels were markedly elevated. However, her fever was immediately improved and her infection markers were decreased within a few days after each admission. Therefore, we believe her elevated WBC count and CRP levels might have been, at least in part, associated with elevated ketone body levels because WBC counts and CRP levels are increased in subjects with diabetic ketoacidosis.
As described in the Case presentation section, the patient was diagnosed with MAC pulmonary disease at 52, and she experienced a long disease duration, although her MAC pulmonary disease radiologic findings had not been severe over the last 10 years (Figure 3). Therefore, we believe that weight loss in this subject might have been, at least in part, associated with MAC pulmonary disease. However, this patient developed polymyalgia rheumatic and rheumatoid arthritis at 73 and 75, respectively, and subsequently, her body weight further decreased (Figure 3). Therefore, we cannot deny the possibility that polymyalgia rheumatic and rheumatoid arthritis were also involved in body weight loss in this subject. We assume that MAC pulmonary disease and subsequent collagen diseases were both associated with weight loss in this subject.

Time course of MAC pulmonary disease radiologic findings in this subject over the last 10 years. Positivity for MAC was confirmed by antibody testing and polymerase chain reaction, and the patient exhibited severe weight loss. This patient experienced polymyalgia rheumatic and rheumatoid arthritis, but she had already displayed severe weight loss before the onset of these conditions.
Conclusions
Taken together, we should remember that pulmonary diseases including MAC pulmonary disease are often complicated by low body weight, and therefore, patients could have acetonemic vomiting with mild hypoglycemia and ketosis. In addition, we should consider the possibility of acetonemic vomiting in both children and adults, especially in underweight subjects with repeated nausea and/or vomiting. Moreover, it is important for such patients to receive appropriate education and diet therapy to mitigate their symptoms.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221118506 - Supplemental material for Adult acetonemic vomiting complicated with low body weight in a subject with Mycobacterium avium complex pulmonary disease: a case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605221118506 for Adult acetonemic vomiting complicated with low body weight in a subject with Mycobacterium avium complex pulmonary disease: a case report by Hitomi Tanaka, Takatoshi Anno, Haruka Takenouchi, Katsumasa Koyama, Fumiko Kawasaki, Katsumi Kurokawa, Niro Okimoto, Koichi Tomoda and Hideaki Kaneto in Journal of International Medical Research
Footnotes
Ethics statement
Written informed consent was provided by the patient. All details have been de-identified such that the identity of the patient may not be ascertained in any manner. The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Research Ethics Committee of Kawasaki Medical School and Hospital (protocol code: 5420-00; date of approval: 27 September 2021). The reporting of this study conforms to CARE guidelines. 8
Data availability statement
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author contributions
Hi.T. and T.A. analyzed data and wrote the manuscript. Ha.T., Katsuma K., F.K., and Katsumi K. analyzed data and contributed to the discussion. N.O., K.T., and H.K. reviewed the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
