Abstract
A Y842D mutation within the activation loop of fms-like tyrosine kinase 3 (FLT3) has been shown to confer strong resistance to sorafenib in vitro. Whether this type of mutation exerts clinically significant effects in patients with acute myeloid leukaemia (AML) remains unclear. Here, a novel Y842D activating mutation within the kinase domain of FLT3, in a pregnant patient with de novo hyperleucocyte acute myeloid leukaemia, is described. Following induction failure with standard dose idarubicin and cytarabine (IA), the patient received re-induction combined with midostaurin, a promising agent targeting mutant-FLT3, and IA regimen. Fortunately, morphological remission was achieved. During the period of midostaurin treatment, the patient exhibited a symptom that was characteristic of differentiation syndrome, which disappeared following treatment with methylprednisolone. The present case revealed that Y842D, an uncommon activating mutation in the activation loop of FLT3, may be a midostaurin-sensitive mutation type in patients with acute myeloid leukaemia.
Keywords
Introduction
Fms-like tyrosine kinase 3 (FLT3) is a type 3 receptor tyrosine kinase. Certain alterations in this protein constitutively activate the receptor tyrosine kinase, causing uncontrolled proliferation, reduced apoptosis, and malignant transformation of myeloid cells. 1 Thus, mutant FLT3 is well known as a promising target for molecular therapy in acute myeloid leukaemia (AML).1,2 FLT3 mutations have been found to occur in 30% of patients with de novo AML, with internal tandem duplication (namely, FLT3-ITD) in the juxtamembrane domain observed in 75% of mutant patients, and point mutation within the activation loop of the tyrosine kinase domain (namely, FLT3-TKD) observed in the remaining 25%. 3 Based on previous clinical research, the adverse effect of FLT3-ITD on patients with AML cannot even be reversed by hematopoietic stem cell transplantation (HSCT). 4 Given the rare probability of FLT3-TKD mutation in patients with AML, it is reasonable that no large-sample, randomized controlled clinical trial in patients with this mutation type has been published to date. However, presently available results suggest that the negative impact of TKD mutation is comparable to that of ITD regarding duration of disease-free survival in patients with AML. 5
The D835 residue is the most common site of FLT3-TKD mutations, with other types of mutation, such as those in 836 and 840 residues, also being found in patients with AML. 6 The Y842 residue, with coding sequence located in exon 20, is key in regulating FLT3. Y842 forms a hydrogen bond with Asp811, and the latter is linked to Arg834 through an ion pair, leading to a closed (inactive) conformation of the FLT3 activation loop. 7 When the 842 tyrosine residue was substituted with cysteine (FLT3-Y842C), the FLT3 activation loop switched from the closed to open (active) conformation, leading to constitutive activation of FLT3 and signal transducer and activator of transcription 5 (STAT-5) tyrosine phosphorylation, thus contributing to malignant transformation of myeloid cells. 8 The FLT3-Y842C mutation type is potentially sensitive to sorafenib, a multitargeted kinase inhibitor that was shown to inhibit activation of FLT3 signalling and suppress proliferation of leukaemia cells in preclinical models. 8 However, replacement of the 842 tyrosine residue with aspartic acid (Y842D) exhibited different in vitro biological characteristics compared with Y842C. According to Von Bubnoff et al., 9 the Y842C mutation, but not Y842D/H/N, enabled FLT3 to form a stable binding interaction with sorafenib. Consequently, Y842D mutant leukaemia cells were much more resistant to sorafenib.
Herein, the characterization of a novel type of activating mutation (Y842D) within the kinase domain of FLT3 is described in a patient with de novo AML, and, to the best of the present authors’ knowledge, is the first such case with mutant Y842D reported to have achieved complete remission after re-induction with midostaurin plus idarubicin and cytarabine (IA) regimen. The result showed that the FLT3-ITD Y842D form of mutation may be sensitive to a chemotherapy regimen containing midostaurin.
Case report
A 29-year-old patient, gravida 1 parity 0, at 18 weeks of pregnancy, was admitted to Shenzhen University General Hospital in August 2020, for intermittent fever over the previous week. Her medical, family, and psycho-social histories were unremarkable. Physical examination revealed signs of anaemia and fever. Routine blood test showed white blood cell (WBC) count of 203.39 × 109/L (normal range, 4.0–10 × 109/L), haemoglobin (Hb) level of 78 g/L (normal range, 120–175 g/L), and platelet (PLT) count of 96 × 109/L (normal range, 100–350 × 109/L). Approximately 1 month previously, in July 2020, the patient had undergone a routine blood test during her pregnancy examination that revealed a WBC count of 7.63 × 109/L, Hb level of 121 g/L, and PLT count of 193 × 109/L. Notably, the patient's WBC count underwent a 26.66-fold increase in just 40 days. Meanwhile, a blood smear indicated 95% blast cells in peripheral blood, and a bone marrow smear showed extremely active hyperplasia, with 91% blast cells and significantly inhibited proliferation of granulocytes, macrophages, megakaryocytes and erythroblasts. Blast cells were strongly positive for esterase staining, which could be inhibited by sodium fluoride. Flow cytometry of bone marrow samples revealed that myeloid blasts exhibited monocytic differentiation tendency (CD34–, CD117–, CD33+, CD64+, CD4 dim, CD13–, and human leucocyte antigen [HLA]-DR+). Karyotype analysis of bone marrow cells revealed a translocation (t) (8;22) (p11.2; q13) mutation (Figure 1). Genetic analyses were performed using a NovaSeq™ 6000 Sequencing System that utilises real-time polymerase chain reaction (Illumina; San Diego, CA, USA), according to the manufacturer’s instructions, and fluorescence in situ hybridization (FISH). The FISH results showed that no t(15;17), t(8;21), t(16;16), or inv(16) mutations were present, or the corresponding transcript. Gene mutation analyses revealed an FLT3 point mutation (T2524G), which resulted in replacement of alanine with aspartic acid at site 842 within the receptor tyrosine kinase domain (FLT3-TKD, Y842D), with a mutant-to-wild type allelic ratio of 36.4% (0.364: 1). In addition, another mutant gene, lysine methyltransferase 2D (KMT2D), was detected with an allelic ratio of 46.6%. Comprehensively, the patient was diagnosed with AML-M5 and categorised with an unfavourable prognosis, according to the French–American–British (FAB) classification system and 2019 version of the National Comprehensive Cancer Network (NCCN) guidelines. 10
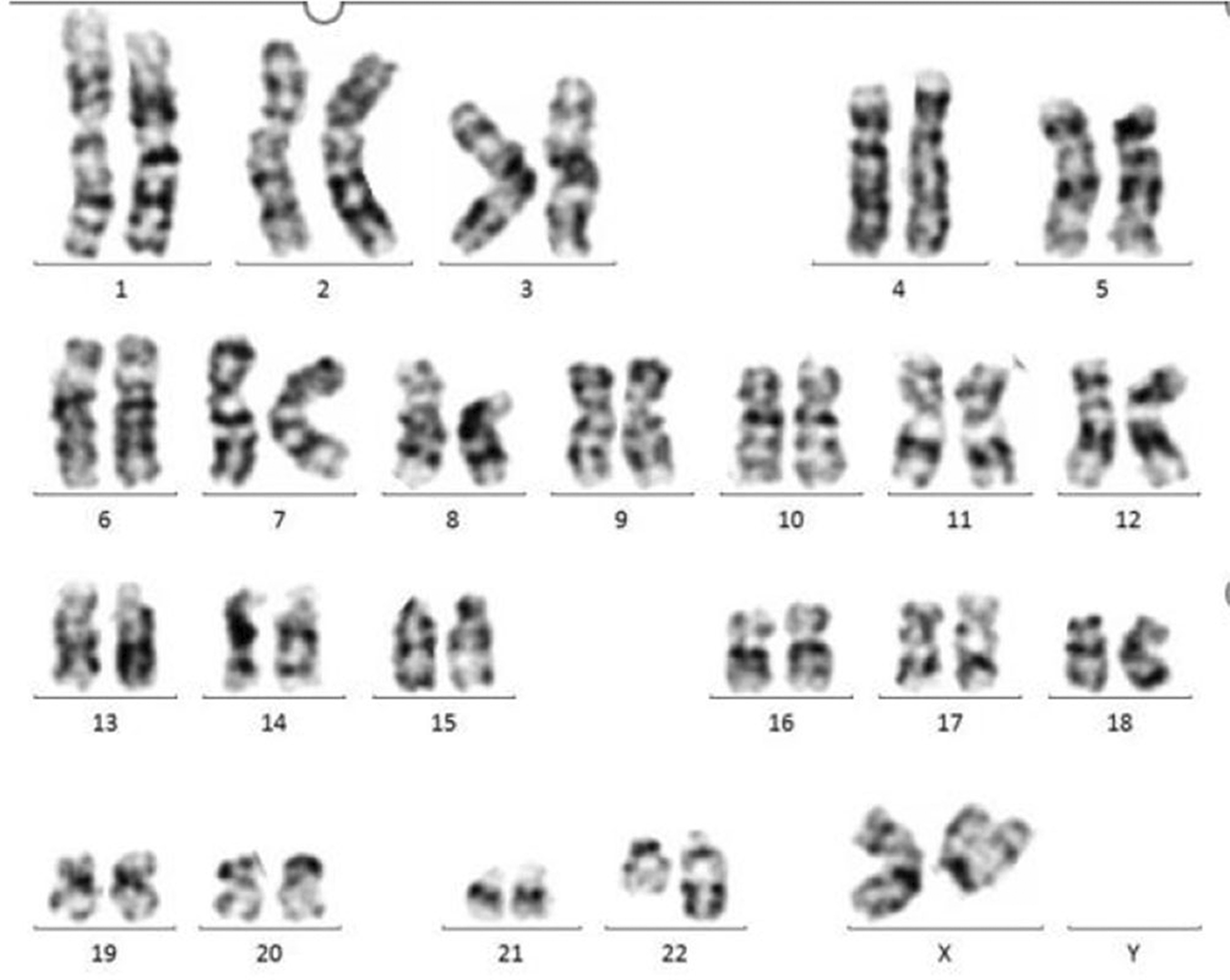
Karyotype of a 29-year-old female patient, at 18 weeks of pregnancy, at the time of diagnosis with de novo acute myeloid leukaemia.
After written informed consent for treatment was obtained from the patient, she was administered 1 g hydroxyurea, orally, 3–4 times daily for 5 days, to treat hyperleukocytosis, and artificial abortion was performed prior to induction chemotherapy. The first induction therapy was initiated on 14 September 2020, using 10 mg/m2 idarubicin, intravenous infusion, daily (days 1–3), and 100 mg/m2 cytarabine, intravenous infusion, daily (days 1–7), namely, an IA regime. Bone marrow smear analysis on 12 October 2020 revealed 8.8% primitive monocytes and 30% immature monocytes still residing in bone marrow. In addition, flow cytometry of bone marrow samples revealed 35.2% myeloid blasts showing monocytic differentiation tendency. To obtain better clinical efficacy, the FLT3-ITD inhibitor midostaurin (50 mg, orally, every 12 h, days 8–21) was combined with the IA regime in re-induction therapy initiated on 14 October 2020. On 30 October 2020 (10 days after starting midostaurin treatment), the patient developed symptoms characteristic of differentiation syndrome: a fever, with body temperature gradually rising from 38.1 to 40.2°C; and skin rash accompanied by pruritus that rapidly spread from both lower extremities to the whole body, including the face, and continued to progress over 3 to 4 days (Figure 2). Blood biochemistry analyses revealed that glutamic-pyruvic transaminase and glutamic oxaloacetic transaminase levels were 89 U/L and 160 U/L, respectively. On 6 November 2020, following treatment with 40 mg/day methylprednisolone, continuous intravenous infusion, every 12 h for 1 week, the skin rash gradually resolved. On 11 November 2020, no primitive monocytes or immature monocytes were found in the bone marrow, and the patient achieved complete remission with incomplete count recovery. Evaluation of minimal residual disease by flow cytometry revealed that myeloid blast cells accounted for <0.1% of bone-marrow non-erythroid cells, i.e., minimal residual disease was negative. Considering the adverse effect of FLT3-TKD on clinical prognosis, the patient was treated with a cycle of consolidation therapy comprising 1.5 g/m2 cytarabine, intravenous infusion, every 12 h, days 1, 3 and 5, and 50 mg midostaurin, orally, every 12 h for 14 days. On day 45 of complete remission, the patient underwent an allogeneic HSCT using a myeloablative preparative regimen of busulfan-cyclophosphamide (busulfan 0.8 mg/kg every 6 h, day –7 to day –4, and 60 mg/kg cyclophosphamide, daily, day –3 to day –2), and an HLA-matched sibling donor. In the post-transplant period, the patient developed mild chronic graft-versus host disease of the mouth and eyes, which did not require treatment. Her whole disease course and clinical outcomes are summarized in Figure 3.
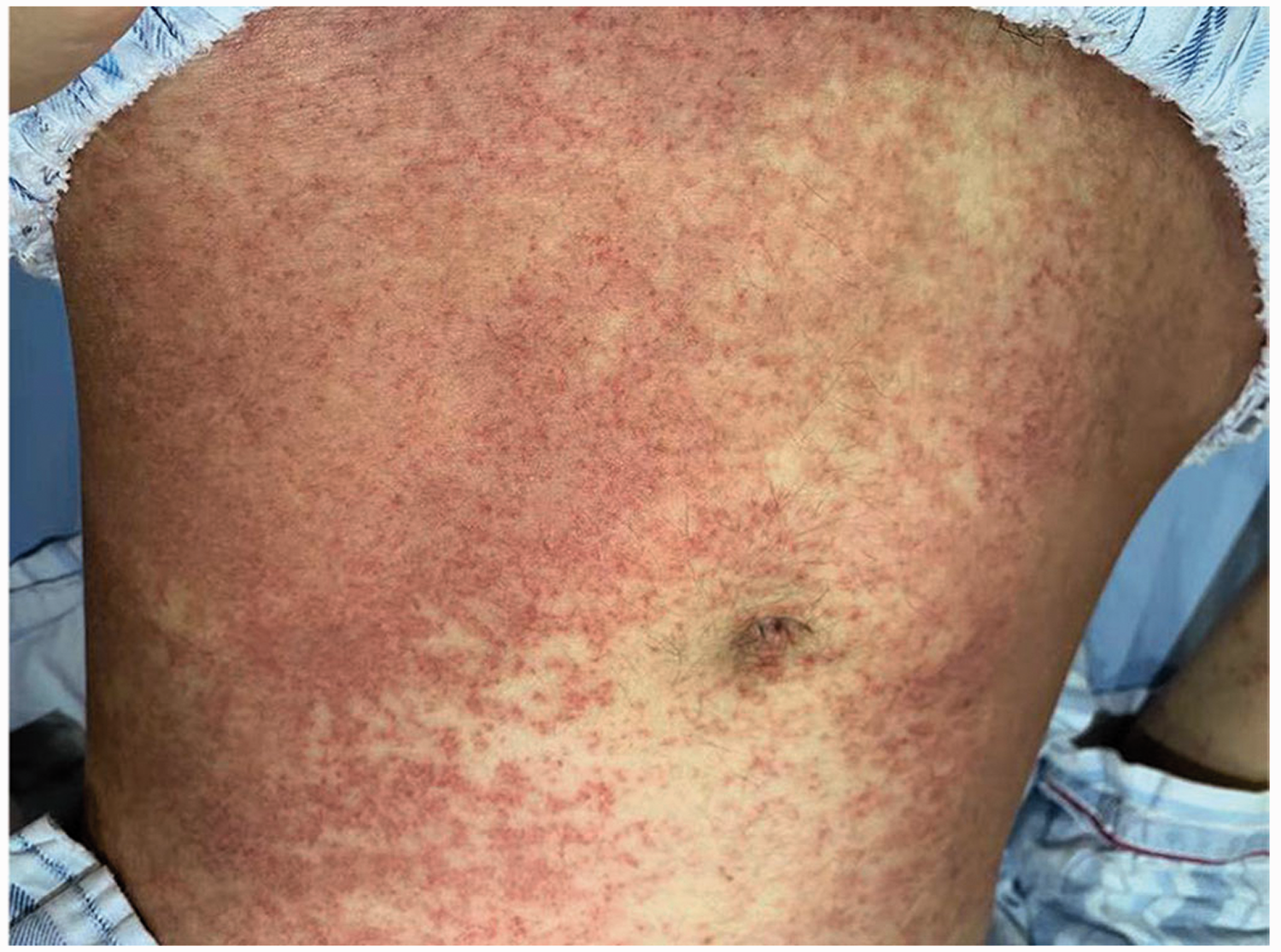
Representative image showing diffuse red subcutaneous nodules on the abdomen of a 29-year-old female patient, at 18 weeks of pregnancy, diagnosed with de novo acute myeloid leukaemia.

Timeline of clinical events for a 29-year-old female patient, at 18 weeks of pregnancy, diagnosed with de novo acute myeloid leukaemia. IA, cytarabine and idarubicin; NR, no remission; DS, differentiation syndrome; WBC, white blood cell count.
The Ethics Committee of Shenzhen University General Hospital approved the publication of this case report. Written informed consent was obtained from the patient for the publication of this article and any accompanying potentially identifiable images or data. The reporting of this study conforms to CARE guidelines. 11
Discussion
The past 30 years have witnessed the thriving development of fundamental molecular-related research on hematopoietic cells, which has greatly enriched therapeutic strategies for acute myeloid leukaemia and led the priority treatment of leukaemia into an era of molecularly-guided precision therapy.12–14 For example, the application of all-trans retinoic acid and arsenous in the treatment of acute promyelocytic leukaemia (APL) has resulted in about 90% of patients with APL being cured. 14 Therapies based on genetic and epigenetic changes have played an increasing role in the treatment of non-M3 AML. In combination with the standard 3 + 7 regimen, molecular-targeted agents have been expected to effectively improve the remission rate of induction therapy and provide more opportunities for subsequent HSCT. 15
Mutant FLT3 contains two main forms of mutation, ITD in the juxtamembrane domain and point mutation within the activation loop of the TKD. 2 FLT3 mutation occurs in 30% of patients with de novo AML and accounts for the predominant mutation type in AML. 1 Notably, FLT3-ITD is considered to be an independent unfavourable risk factor in AML, as patients with FLT3-ITD are always resistant to chemotherapy and present with a higher relapse rate and more cases of early death. Though the clinical significance of FLT3-TKD mutation in AML remains unclear due to the relatively rare incidence, meta-analysis has shown that FLT3-TKD exerts an unfavourable effect on AML similar to FLT3-ITD. 5
In patients with AML, mutant FLT3 is frequently accompanied by hyperleukocytosis. 16 Nevertheless, the particular effect of each mutation form, including Y842D, on clinical phenotype remains unclear. In the present case, the patient’s WBC count increased from 7.63 × 109/L to 203.39 ×109/L over just 40 days. The doubling time was as short as 8.45 days, which is very similar to the clinical characteristic described in patients with FLT3-ITD. Consecutive activation of FLT3 and downstream signalling pathways are generally known to result in uncontrolled proliferation of hematopoietic cells. According to the research of Kindler et al., 8 a point mutation at Y842 altered the conformation of the FLT3 receptor TKD and drove the formation of an activation loop. Mediated by a downstream signalling pathway, including signal transducer and activator of transcription 5 (STAT5), myeloid cells with Y842C acquired the ability to proliferate indefinitely. In addition, murine 32D cells transfected with human FLT3-Y842C showed activated FLT3 and STAT5 signalling, which could be inhibited by an FLT3 inhibitor (N-benzoyl staurosporine, PKC412). 9 The above results demonstrate that mutation of FLT3-Y842 plays a critical role in the development of AML. Consequently, it would be reasonable to deduce that the hyperleukocytosis observed in the present patient may have been attributed to continuous activation of FLT3 caused by the Y842D mutation.
Another important biological characteristic of FLT3-Y842D mutant AML cells is that they may be resistant to certain FLT3 inhibitors. Through culturing cells with different gradient concentrations of FLT3 inhibitors, researchers have been able to successfully screen out FLT3 inhibitor-resistant FLT3 mutant cells. Based on this screening method, Von Bubnoff et al., 9 found that, at the same concentration, myeloid cells with FLT3-Y842D were resistant to sorafenib and not midostaurin, which may be the reason why sorafenib only was used in the treatment of patients with FLT3-ITD mutant AML. 17 In a phase III, randomized controlled clinical trial, researchers compared the induction-therapy effect of daunorubicin (60 mg/m2, days 3–5) and cytarabine (20 mg/m2, days 1–7), with or without midostaurin, in 717 patients with AML and mutant FLT3, including 162 patients with FLT3-TKD. 15 Statistically, the median overall survival time in the midostaurin group (74.7 months; 95% CI 31.5, not achieved) was significantly better than 25.6 months (95% CI 18.6, 42.9) observed in the placebo group. In addition, the incidence of adverse events between the above two groups revealed no significant difference. 15 The lack of difference in adverse events is also a critical reason for the clinical application of midostaurin in patients with AML and FLT3 mutation, and why midostaurin, cytarabine, and idarubicin were selected for the re-induction regimen in the present case. At 23 days after re-induction, morphological remission was achieved, which suggests that AML associated with the FLT3-Y842D mutation in the present patient was potentially sensitive to midostaurin.
Another characteristic of the present case was that the patient presented with differentiation syndrome on day 10 of midostaurin treatment, characterised by a red nodular rash that first appeared in symmetrical parts of both lower limbs, accompanied by pruritus and fever. This type of rash is often observed in patients treated with selective FLT3 inhibitors, such as quizartinib and gilteritinib.18,19 Pathologically, FLT3 inhibitors have been shown to induce differentiation of primordial cells into neutrophils, and this rash was caused exactly by the infiltration of neutrophils in deep skin and subcutaneous tissues. 19 In patients with AML who received quizartinib monotherapy, though the proliferative degree of nucleated cells did not alter significantly 29 days after therapy, the proportions of cells with different degrees of differentiation was substantially changed. The proportion of primordial cells decreased from 77% to 6%, and the proportion of myelocyte, metamyelocyte, and neutrophils increased from 9% to 57%. As the mutation frequency of FLT3-ITD remained unchanged, it was sufficient to conclude that the mature neutrophils were derived from the differentiation of primordial cells. 18 In a study that evaluated the validity of gilteritinib in 21 patients with AML and mutated FLT3, 10 patients experienced differentiation syndrome during treatment. The overall survival time and morphological remission rate in these 10 patients was far worse than in the 11 patients who did not develop differentiation syndrome. 20 Similar to observations with the prevention of retinoic acid-induced differentiation syndrome, glucocorticoid combined with FLT3 inhibitor not only unregulated expression of the proapoptotic protein BCL2 like 11 (BCL2L11, also known as BIM), but also induced leukaemia cell apoptosis by promoting the degradation of induced myeloid leukemia cell differentiation protein Mcl-1, an antiapoptotic protein. 21 Therefore, FLT3 inhibitor combined with glucocorticoid in the prevention of FLT3 inhibitor-induced differentiation syndrome requires further study. Notably, patients with AML and mutant FLT3 combined with mutant nucleophosmin 1 (NPM1) or rearranged core binding factor (CBF) were considered to have a favourable prognosis. 22 Since the patient described herein showed only mutant FLT3 and could not be classified into a favourable prognosis subgroup, and an HLA allele-matched donor was available, it was deemed sensible to treat this patient with allogeneic HSCT as soon as possible, and to provide maintenance therapy after transplantation with midostaurin, in order to further improve the clinical outcome.
The results of the present case may be limited by the fact that sensitivity of the Y842D mutation to midostaurin could not be confirmed experimentally. Therefore, further studies are required to demonstrate the sensitivity of the Y842D mutation to midostaurin. Additionally, the mutation shown in the present patient is uncommon, whereas detection of the FLT3-Y842C mutant may be commonly performed by laboratory testing companies, thus, a suitable laboratory test was not readily available in time to monitor Y842D after midostaurin treatment and before transplantation. Flow cytometry, used to monitor the minimal residual disease, revealed that the patient was negative for minimal residual disease before transplantation.
In conclusion, the present case showed that FLT3-Y842D is a novel form of activating FLT3 mutation in AML, and patients with this mutation may be sensitive to midostaurin. In the process of midostaurin treatment, healthcare practitioners should be alert to the existence of differentiation syndrome, and once observed, glucocorticoid should be administered as soon as possible. Notably, allogeneic HSCT should be conducted to achieved complete remission as soon as possible in such patients.
Footnotes
Author contributions
MJ composed the manuscript and reviewed the literature. SJ, JY and WQ acquired and analysed the laboratory data. YL critically revised and interpreted the data. PY provided the clinical patient care. MJ and SJ wrote the manuscript. YP fully revised and improved the manuscript. All authors contributed to the article and approved the submitted version.
Data accessibility
The raw data supporting our findings can be requested from the corresponding author.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by ‘Major New Drug Development Project’ grants from the Ministry of Science and Technology of China (2019ZX09201002003), State Key Program of National Natural Science of China (82030076), National Natural Science Foundation of China (82070161, 81970151, and 81870134), Beijing Natural Science Foundation (7202186), Natural Science Foundation of Shenzhen University General Hospital (SUGH2020QD008), the project of medical science research project in 2019 in Hebei Province (grant No. 20190122) and the project of medical science research project in 2016 in Hebei Province (grant No. 20160041).
