Abstract
Objective
To screen for specific differentially expressed genes in small cell neuroendocrine carcinoma of the cervix (SCNEC) and to further explore their roles and mechanisms in tumor progression.
Methods
Differentially expressed genes in SCNEC compared with squamous cell carcinoma (SCC) and adenocarcinoma (AC) were screened by microarray and immunohistochemical analyses. The biological functions of the identified genes were examined in a SCNEC cell line using RNA interference and over-expression plasmid-transfection technologies. Co-expression network analysis and immunoprecipitation technology were used to explore the potential mechanisms.
Results
Compared with SCC and AC, UCHL1 (encoding ubiquitin C-terminal hydrolase L1) was identified as a specific differentially expressed gene in SCNEC, which was positively related to lymph node metastasis (LNM). Migration and invasion of SCNEC tumor cells were induced by UCHL1 over-expression and suppressed by UCHL1 down-regulation, as shown by scratch and transwell invasion assays. Co-expression network analysis suggested that Prospero homeobox protein 1 (PROX1) might interact with UCHL1, and in vivo immunoprecipitation and western blots verified that levels of ubiquitinated PROX1 were significantly decreased following UCHL1 overexpression.
Conclusion
UCHL1 is a potential biomarker of LNM in SCNEC. UCHL1 might promote SCNEC cell migration and invasion by reducing PROX1 ubiquitination.
Keywords
Introduction
Cervical cancer is a common gynecological malignancy and the fourth most common cancer in women, with a relatively high incidence and mortality in developing countries. 1 Approximately 70% of all cervical cancers are squamous cell carcinoma (SCC), 25% are adenocarcinoma (AC), and the remaining 5% are rare pathological types, 2 including small cell neuroendocrine carcinoma of the cervix (SCNEC), which accounts for only 0.31% to 2.00% of all malignant cervical tumors, with an incidence of approximately 0.06/100,000. 3 One or two positive neuroendocrine markers are needed to help establish and confirm the diagnosis of SCNEC. Synaptophysin (Syn), neural cell adhesion molecule (CD56), neuron-specific enolase (NSE), and chromogranin A (CgA) are currently considered as immunohistochemical (IHC) staining markers for SCNEC.4,5
Compared with more common pathological types of cervical cancer, SCNEC has a higher rate of lymph node metastasis (LNM) and a poorer prognosis. Approximately 40% of SCNEC patients are lymph node-positive, compared with is only 10% to 15% of patients with SCC or AC.6–8 Burzawa et al. found 5-year survival rates of patients with non-SCNEC with local occurrence, regional metastasis, and distant metastasis of 91%, 57%, and 16%, respectively, compared with only 32% to 63%, 0% to 18%, and 0%, respectively, for SCNEC.9,10
To the best of our knowledge, there have been no mechanistic studies of SCNEC to date, possibly because of the lack of any commercially available cell line for SCNEC. China has the highest number of new cancer diagnoses and cancer-related deaths worldwide, 1 which is unfortunate, but also provides opportunities to investigate rare diseases such as SCNEC. We therefore carried out gene expression microarray analysis and validated the results in clinical SCNEC specimens, primary SCNEC cell cultures, and cell biology experiments, to provide a more in-depth understanding of the occurrence and development of this disease.
Materials and methods
Patients and cervical cancer tissue specimens
We analyzed cancer tissues from patients with cervical cancer who underwent radical surgery at our hospital between January 2013 and December 2016. All specimens were confirmed by post-surgical pathological examination. Paraffin-embedded specimens were used for IHC, and fresh tissues were used for gene expression profile detection and quantitative reverse transcription polymerase chain reaction (RT-qPCR) (Figure 1). All tissues used in this study were obtained with informed consent from the patient, and the study was approved by the Ethics Committee of The Obstetrics and Gynecology Hospital of Fudan University (419 Fang-xie Road, Shanghai, P.R. China; No. 2016-40; Sep-06-2016). We also investigated the prognostic risk factors in additional SCNEC patients who underwent radical surgery at our hospital between January 2014 and December 2017. These patients were also recruited after providing informed consent. All clinicopathological information was collected for statistical analysis. The end point of the follow-up was March 2019.

Differential gene screening and validation flow chart.
Gene expression microarray
SCNEC has been reported to have mixed SCC or AC components. 11 We therefore analyzed differentially expressed genes in three SCNEC tumors compared with adjacent non-cancerous tissues, and also selected three SCC and AC tissues as control groups to specifically screen out differentially expressed genes in SCNEC. Samples were subjected to GeneChip® Human Transcriptome Array 2.0 analysis by Shanghai Biotechnology Corporation (Shanghai, China). Briefly, the samples underwent: 1) RNA extraction, purification, and quality control; 2) RNA amplification and labeling; 3) array hybridization; and 4) data acquisition and analysis.
RT-qPCR, IHC, and western blotting
RT-qPCR, IHC, and western blotting were performed as described previously.
12
For qRT-PCR, RNA was extracted from cervical cancer tissues or SCNEC cells using TRIzol Reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer's instructions. RT was performed using an RT kit (Promega, Madison, WI, USA) and RT-qPCR was performed using a SYBR Green qPCR Master Mix kit (TaKaRa, Shiga, Japan). The primers for UCHL1 were: 5′-
Primary SCNEC cell culture and validation
Tumor cells from three SCNEC patients were used for primary cell culture, but cultures were only established successfully for one patient (a 64-year-old female patient diagnosed with SCNEC via biopsy before radical surgery). A fresh tumor fragment was collected and subjected to enzymatic digestion followed by centrifugation at 225 × g. Cells were cultured at 37.8°C with 5% CO2 in Dulbecco’s Modified Eagle Medium containing basic fibroblast growth factor and epidermal growth factor (Gibco, Carlsbad, CA, USA). SCNEC cells were verified by immunofluorescence staining for the SCNEC immune markers Syn, CD56, and CgA, as described previously. 12 Anti-Syn, anti-CD56, and anti-CgA antibodies were provided by the Pathology Department, The Obstetrics and Gynecology Hospital of Fudan University, Shanghai, China.
Cytological experiments
Each biological experiment was repeated three times. An UCHL1-OE/pSuper vector was constructed by SynBio-Tech (Suzhou, China). UCHL1-specific small interfering RNA (siRNA) oligonucleotides with the following sequences were purchased from GenePharma (Suzhou, China): GGGAAUUCCUGUGGCACAATT (sense) and UUGUGCCACAGGAAUUCCCTT (antisense). Transfections were performed using Lipofectamine 2000 (Thermo Fisher, Waltham, MA, USA) following the manufacturer’s protocol. For scratch assays, transfected cells were inoculated into a 6-well plate at 1 × 106 cells/well, incubated overnight, and then scratched vertically the following day using a 100-µL micropipette tip. The cells were washed twice with phosphate-buffered saline and placed in serum-free culture medium. After 24 and 48 hours, cells in five random fields were counted under an inverted phase-contrast microscope.
Cell invasion was measured in Boyden chambers using Transwell filters (Corning Inc., Corning, NY, USA) with Matrigel (Corning). Cells (1 × 105 in 0.2 mL of serum-free medium) were placed in the upper chamber and the lower chamber was loaded with 0.6 mL of medium containing 10% fetal bovine serum. Cells that migrated to the lower surface of the filters were stained with Crystal violet solution. Five fields in each well were counted after incubation for 24 and 48 hours at 37.8°C with 5% CO2.
Ubiquitinated protein levels were detected. One day after transfection of UCHL1-OE or UCHL1-RNAi into SCNEC cells, an HA-Ubi plasmid (Tonker Biotechnology Co., Shanghai, China) was transfected into the same cells. After 2 hours, the cell lysate was immunoprecipitated with PROX1 antibody and immunoblotting was then performed with ubiquitin (ab7780; Abcam, Cambridge, MA, USA) and PROX1 antibodies. Input samples were tested for protein expression using UCHL1 and GAPDH (KC-5G4; Kangchen, Shanghai, China) antibodies.
Statistical analysis
Molecules that may interact with UCHL1 were analyzed using Cytoscape (https://cytoscape.org/) and possible transcription factors for UCHL1 were predicted using the TRANSFACT database (http://gene-regulation.com/pub/databases.html#transfac). The test level was set at α = 0.05, test efficiency 1−β = 0.80, and kappa = 1:2. With a ratio of two groups of samples (n1: n2), according to the formula in the database (http://powerandsamplesize.com/Calculators/Compare-2-Proportions/2-Sample-Equality), 13 cases were needed in the experimental group and 25 cases in the control group. Under these conditions, A positive rate of candidate genes >85% was needed to reach statistical significance. At the beginning of this study, there were 16 available SCNEC specimens in our specimen bank, and we therefore used these 16 samples in the experimental group and included 30 control samples.
Data are presented as mean ± standard error. Relationships between UCHL1/PROX1 expression and clinicopathological features were analyzed by Pearson χ2 test. Quantitative data were compared using two-tailed Student’s t-tests or ANOVA. Univariate analysis of survival was carried out using the Kaplan–Meier method and multivariate analysis was carried out using the Cox regression method. All statistical analyses were performed using SPSS 22.0 software (IBM Corp., Armonk, NY, USA). P < 0.05 was considered statistically significant.
Results
Patients
The Specimen Bank of our hospital included samples from 85 patients with cervical cancer (76 formalin-fixed paraffin-embedded tissues, including 16 SCNEC, 30 SCC, and 30 AC; and 12 fresh tissues, including 3 SCNEC, 3 paired SCNEC paracancerous tissues, 3 SCC, and 3 AC) who underwent radical surgery at our hospital between January 2013 and December 2016. The mean age of the patients was 49.83 years (range, 25–78 years). The paraffin-embedded specimens were used for IHC and the fresh tissues were used for gene expression profile detection and qRT-PCR. An additional 53 SCNEC patients (mean age 44.45 years; range, 24–75 years) who underwent radical surgery at our hospital between January 2014 and December 2017 were also recruited, to investigate the prognostic risk factors
Preliminary screening of four differentially expressed candidate genes (UCHL1, PROX1, CRMP5, and TM4SF1) from gene expression chip data
There were 5348 differential gene probes between SCNEC tumors and adjacent tissues, 3362 between SCNEC and AC, and 3244 between SCNEC and SCC, which involved many Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes pathways (Supplementary Fig. 1). It was difficult to select specific differentially expressed genes for SCNEC from these data. It has been reported that 11% to 64% of SCNECs are mixed with an AC or SCC component, 11 and it is therefore possible that the pathological components of the three SCNEC tumors were not pure. Indeed, postoperative pathology suggested that two of the three SCNECs were mixed with AC. We therefore examined the intersections of the three differentially expressed gene groups (SCNEC vs adjacent tissues, SCNEC vs AC, and SCNEC vs SCC) to narrow the screening scope. We screened out relatively specific differentially expressed genes in SCNEC as genes that were expressed differentially in SCNEC tissues compared with nonmalignant adjacent tissues, SCC, and AC. We examined the top 200 differentially expressed genes in the three groups (Figure 2a). There were 11 probes with high specific expression and one probe with low specific expression in SCNEC, most of which were related to tumorigenesis and development, especially PROX1, UCHL1, and CRMP5 (Supplementary Table 1). Finally, four genes, UCHL1, PROX1, CRMP5, and TM4SF1, were identified as preliminary candidates based on literature references and the number of participants in GO (Figure 2b, c).
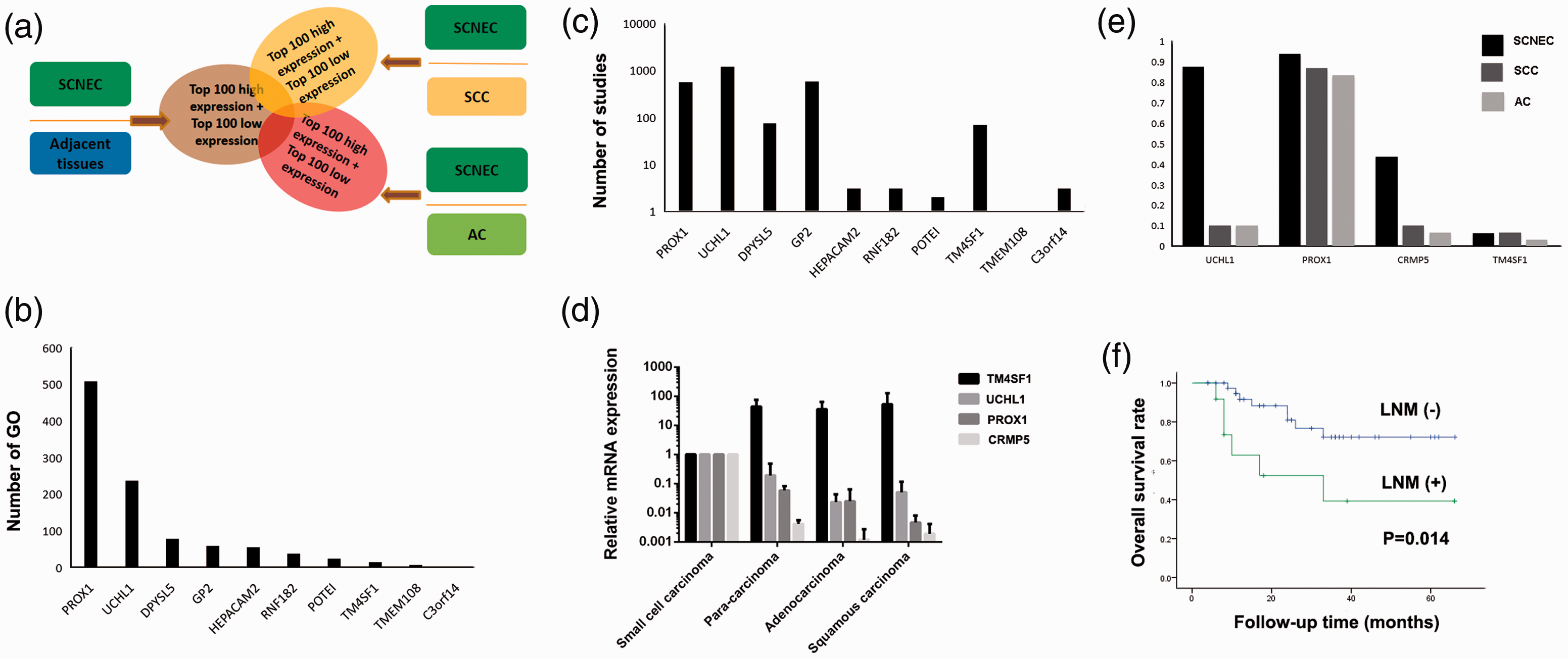
Screening of specific genes for small cell neuroendocrine carcinomas of the cervix (SCNEC) and analysis their clinical significance. (a) SCNEC-specific differentially expressed gene screening map. The overlapping parts of the three ellipses are specific genes for SCNEC. (b, c) Statistical analysis of number of specific differentially expressed genes participating in Gene Ontology pathways and number of literature reports. LOC101926908 and PTPRG-AS1 are non-coding RNAs and were not included in the analysis. (d) Expression of UCHL1, PROX1, CRMP5, and TM4SF1 in three types of cervical cancer. Histograms show ratios of different specimens to SCNEC. (e) Positivity rates for the above proteins in the three types of cervical cancer. (f) Survival analysis of 53 SCNEC patients with or without lymph node metastasis.
Further validation of UCHL1 among the four candidate genes
The gene expression results were validated by qRT-PCR and IHC. The qRT-PCR results were consistent with the microarray analysis: UCHL1, PROX1, and CRMP5 expression were higher in SCNEC than in adjacent tissue, SCC, and AC, while TM4SF1 expression levels were relatively reduced in SCNEC (Figure 2d). IHC showed that the rate of PROX1-positivity was high in all three tumors, but there was no significant difference among them (93.8% vs 86.7% vs 83.3%). A similar situation was found for reduced TM4SF1 expression (6.3% vs 6.7% vs 3.3%). UCHL1 (P < 0.001) and CRMP5 (P = 0.002) expression differed significantly among the three malignant groups. The UCHL1-positivity rates in SCNEC, SCC, and AC were 87.5%, 10.0%, and 10.0%, respectively, while those of CRMP5 were 43.8%, 10.0%, and 6.7%, respectively (Figure 2e). According to the requirement for positive expression (>85%), UCHL1 was determined to be a specific candidate for further research. The IHC-positivity rates of the other potential SCNEC markers (CgA, Syn, NSE) were lower (range 60.8%–82.5%). 11 These results suggested that UCHL1 could be a potential new diagnostic marker for SCNEC.
UCHL1 expression was positively correlated with LNM in SCNEC patients
We next divided patients into high expression (HG) and low expression groups (LG) according to their UCHL1 IHC scores. Statistical analysis showed that UCHL1 expression was positively correlated with LNM in SCNEC patients (Table 1; P = 0.003). However, there were no significant correlations with other factors, including tumor stage, tumor size, parametrial infiltration, lymphatic vessel infiltration, muscle infiltration depth, age, and histological homology. There were no correlations between UCHL1 protein expression and clinicopathological parameters in patients with SCC or AC (Supplementary Tables 2 and 3). These results further suggested that UCHL1 might be a specific marker for SCNEC from a clinical perspective.
UCHL1 expression level and clinicopathological parameters in 16 patients with small cell neuroendocrine carcinoma of the cervix.

LG, low-expression group; HG, high-expression group.
In 53 SCNEC patients, univariate analysis showed that LNM, parametrial infiltration, and FIGO stage were prognostic risk factors for survival (Table 2). Multivariate analysis confirmed the LNM (P = 0.021, odds ratio [OR]=3.49, 95% confidence interval [CI] 1.20–10.09) and FIGO stage (P = 0.012, OR=3.49, 95%CI 1.06–1.60) were independent risk factors. The 5-year survival rate was 72.2% in SCNEC patients without LNM, and this decreased to 39.3% in patients with LNM (Figure 2f). These results indicated that UCHL1 expression might be negatively related to patient prognosis.
Univariate analysis of prognostic factors in patients with small cell neuroendocrine carcinoma of the cervix.

OS, overall survival; HPV, human papillomavirus; LVSI, lymphovascular space invasion; LNM, lymph node metastasis.
Altering UCHL1 expression influenced the migration and invasion of primary SCNEC cells
Given the potential clinical relevance of UCHL1 in SCNEC, we carried out cytological experiments to clarify its underlying molecular mechanism. Three fresh SCNEC tumor tissues were collected and primary tumor cells (PTCs) were cultured for nearly 2 years, resulting in one successful culture. SCNEC cell morphology in primary culture differed from that in tumor tissues: cultured cells appeared irregular, partly spindle-shaped, partly polygonal, with large nuclei. SCNEC is generally diagnosed when two of the three specific markers (CgA, Syn, and CD56) are positive. We therefore carried out immunofluorescence analysis to validate the PTC, and showed that CgA and Syn were positive while CD56 was negative (Figure 3a), which met the diagnostic criteria. However, postoperative pathology of the patient's tissue specimens suggested that all three markers were positive (Figure 3b). This discrepancy may have been related to the heterogeneity of the tumor cells. This PTC cell line was used for subsequent experiments.

Effect of UCHL1 expression on motility of primary tumor cells (PTCs). (a) Identification of PTCs by immunofluorescence with small cell neuroendocrine carcinoma of the cervix markers (SNEC). (b) Immunohistochemistry of SCNEC markers in patient-derived tumor tissues. (c) Up-/down-regulation of UCHL1 at mRNA and protein levels. (d) Upregulation of UCHL1 enhanced (d) migration and (e) invasion of PTCs. (d and e are averages of three repeated experiments). *P < 0.05; **P < 0.01; ***P < 0.001.
UCHL1-overexpression plasmid and siRNAs were used to up- and down-regulate UCHL1 expression in SCNEC cells, respectively (Figure 3c). The migration and invasion abilities of SCNEC cells were significantly increased following UCHL1 overexpression and significantly decreased after silencing UCHL1, as shown by scratch and transwell experiments (Figure 3d, e). These cell behaviors may underlie the clinical phenotype of lymphatic metastasis.
UCHL1 may promote lymphatic metastasis of SCNEC by altering PROX1 ubiquitination
We further investigated the molecular mechanism of UCHL1 in SCNEC by constructing a co-expression network of differential probes (Figure 4a). The interaction probes included 12 up- and 15 down-regulated mRNA coding genes, and 12 up- and four down-regulated long non-coding RNAs. Interestingly, this network contained PROX1. PROX1 is a transcription factor that is critical for lymphatic duct formation. 13 We therefore hypothesized that there might be a mechanistic link between UCHL1 and PROX1, leading to lymphatic metastasis of SCNEC.

UCHL1 promoted lymph node metastasis of small cell neuroendocrine carcinoma of the cervix (SCNEC) via PROX1. (a) Molecular probes that may interact with UCHL1. Genes expressed as dots; long non-coding RNAs expressed as dots with bands. Red, high expression; blue, low expression. (b) Proportion of high PROX1 expression in three kinds of cervical tumors. (c) Changes in PROX1 ubiquitination levels after up-/down-regulation of UCHL1. (d) Correlation between UCHL1 and PROX1 in tumor tissue samples from SCNEC patients; R = 0.766. (e, f) Immunohistochemical staining of UCHL1 and PROX1.
We hypothesized that PROX1 acted as a transcription factor upstream of UCHL1, and that its high expression promoted UCHL1 transcription and translation. To test this hypothesis, we predicted the transcription factors for UCHL1; however, the results did not include PROX1, thus negating this hypothesis (Supplementary Table 4). We then tested if UCHL1 acted on PROX1. Proteins that need to be degraded are first ubiquitinated and then degraded by proteasomes. UCHL1 is a member of the deubiquitinase family, which plays important roles in protein degradation and other processes.14,15 We therefore hypothesized that PROX1 may be abnormally deubiquitinated by UCHL1 in SCNEC, resulting in lower ubiquitinated PROX1 levels and enhanced PROX1 activity. Detection of levels of ubiquitinated PROX1 showed that UCHL1 overexpression reduced PROX1 ubiquitination levels, while silencing UCHL1 increased these levels (Figure 4b).
According to this hypothesis, SCC and AC should also be prone to LNM if PROX1 expression is high. We therefore quantitatively analyzed PROX1 expression in SCC and AC and divided patients into high and low expression groups (high expression: immune score ≥4). PROX1 expression was significantly higher in the SCNEC group compared with the SCC and AC groups (68.8% vs 20.0% vs 23.3%, P < 0.001) (Figure 4c). IHC further indicated that high UCHL1 expression was correlated with PROX1 expression in SCNEC (P = 0.005), but not in SCC or AC (Table 3). Correlation analysis between UCHL1 and PROX1 in SCNEC showed that UCHL1 was positively correlated with PROX1 (R = 0.766, P < 0.001) (Figure 4d). UCHL1 and PROX1 expression were both positive in the cytoplasm (Figure 4e, f). These results further confirmed that UCHL1 might reduce PROX1 degradation through its deubiquitination activity, thus promoting lymphatic metastasis of SCNEC.
Expression of UCHL1 and PROX1 in three types of cervical cancers.

SCNEC, small cell neuroendocrine carcinoma of the cervix; SCC, squamous cell carcinoma; AC, adenocarcinoma; LG, low-expression group; HG, high-expression group.
Discussion
Similar to other endocrine tumors, SCNEC is mainly treated by surgery combined with radio-chemotherapy; however, the high risk of surgery and serious decline in quality of life do not improve the prognosis. The main reason for the poor prognosis is the low incidence of SCNEC, which makes it difficult to investigate its pathogenesis, develop effective drugs, and optimize surgical techniques. For example, researchers from MD Anderson Cancer Center (Houston, TX, USA) initiated a Phase II clinical trial of weekly paclitaxel and bevacizumab for SCNEC, but only three patients were recruited over a 19-month period from 2008 to 2010 and the study was therefore terminated. 9 Basic research into this disease has also been sparse and superficial. Most previous studies used IHC of paraffin sections to detect the expression of proteins associated with SCNEC, such as cyclin D1, E-cadherin, P-cadherin, N-cadherin, p53, BCL2, BRG-1, P40, SST2, SST5, and CD44.16,17 Notably however, the current study used gene chip and IHC analyses, combined with clinical data, and identified UCHL1 as a specific and highly expressed gene in SCNEC tumor tissues compared with SCC and AC.
Because of its low incidence, the chemotherapy regimen for SCNEC mainly reflects that for small cell neuroendocrine lung cancer. Interestingly, the specific differential expression of UCHL1 in SCNEC compared with other common pathological types has thus been similarly reported in lung cancer. Shimada et al. found that UHCL1 was highly expressed in small cell neuroendocrine lung cancer and associated with its prognosis, while another study showed that UCHL1 was down-regulated in non-small cell lung carcinoma but was not associated with patient survival.18,19
Our biological experiments suggested that UCHL1 and PROX1 might play important roles in SCNEC progression. UCHL1 is a member of the ubiquitin carboxyl terminal hydrolase family, which primarily hydrolyzes molecules linked to the ubiquitin carboxyl terminus. Current studies suggest that UCHL1 plays a role similar to proto-oncogenes in some tumors. 20 For example, UCHL1 is highly expressed in gastric cancer and is positively correlated with tumor size and TNM stage, and negatively correlated with cumulative survival rate. 21 PROX1 is a transcription factor and a key regulator of lymphatic development. It is also associated with a variety of tumors, such as papillary thyroid carcinoma, in which PROX1 was shown to be highly expressed and to promote the metastasis of cancer cells to lymph nodes. 21 These studies provide theoretical support for our experimental results.
The mechanism underlying our clinical findings may involve deubiquitination of PROX1 by UCHL1, as indicated in other studies. For example, Hussain et al. found that UCHL1 overexpression promoted lymphoma formation and was closely related to its deubiquitination activity. 21 Other downstream UCHL1 pathways, including Noxa, hypoxia-inducible factor 1α, and β-catenin, have also been reported. 22 UCHL1 may thus play a switching role and its downstream substrates may be diverse and complex, suggesting its potential as a wide-spectrum target for cancer therapy. However, UCHL1 is also widely expressed in neurons and gonadal cells in vivo, 23 suggesting that the development of drugs targeting UCHL1 should pay attention to potential systemic side effects.
Our study also had some limitations. First, our in vitro experiments were only based on one successfully cultured cell line, which may not fully represent the characteristics of all SCNEC tumors. Second, the sample size was still small. Further studies are needed to expand the sample size for this rare tumor through multi-center cooperation to verify our clinical conclusions, especially the relationship between UCHL1 expression and the prognosis of SCNEC patients. Future clinical trials may be able to estimate the prognosis of patients or develop drugs based on UCHL1.
In conclusion, our data showed that UCHL1 was highly expressed in SCNEC and positively correlated with LNM. We suggest that the underlying molecular mechanism for this effect involves reduced ubiquitination of PROX1 through the deubiquitination activity of UCHL1. UCHL1 could represent a new diagnostic and/or therapeutic target for SCNEC.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221087620 - Supplemental material for Ubiquitin C-terminal hydrolase L1 promotes lymph node metastasis in small cell neuroendocrine carcinomas of the cervix
Supplemental material, sj-pdf-1-imr-10.1177_03000605221087620 for Ubiquitin C-terminal hydrolase L1 promotes lymph node metastasis in small cell neuroendocrine carcinomas of the cervix by Yunqiang Zhang, Jingxin Ding, Xuyin Zhang and Keqin Hua in Journal of International Medical Research
Footnotes
Availability of data and materials
The datasets supporting the conclusions of this article are included and indicated within the article.
Declaration of conflicting interest
The authors declare that they have no conflict of interests.
Funding
This work was supported by Shanghai Hospital Development Center [grant/award number SHDC12015117].
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
