Abstract
Ivabradine is an effective treatment for focal atrial tachycardia. However, it may also be effective for re-entrant atrial arrhythmia. An 85-year-old woman with a history of underlying ischaemic cardiomyopathy complained of worsening symptoms of heart failure because of rapid atrial tachycardia that was resistant to several rate-controlling drugs, but responded well to ivabradine. An electrophysiology study demonstrated a roof-dependent macro-re-entrant tachycardia of the left atrium. Linear ablation of the left atrial roof resulted in termination of the tachycardia. Thus, ivabradine can be an effective treatment for re-entrant atrial tachycardia.
Keywords
Introduction
Atrial tachycardia is common in patients with heart failure with reduced ejection fraction and is often difficult to treat. Cardioversion therapy is the preferred treatment, but thrombosis in the left atrial appendage commonly prevents this. Therefore, the administration of rate-controlling medication is a reasonable choice in patients with severe symptoms and a rapid ventricular rate. However, because of the potential for hypotension and other adverse effects, there are few pharmaceutical options 2 . We hypothesised that ivabradine might represent a good option for ventricular rate control.
Case presentation
The reporting of this case conforms to the CARE guidelines. 1 An 85-year-old woman with ischemic cardiomyopathy, hypertension, and diabetes mellitus was referred to our hospital because of recurrent heart failure and rapid atrial arrhythmia. Pulmonary vein isolation with radiofrequency ablation was performed successfully in February 2019, and the patient was prescribed metoprolol succinate 23.75 mg once daily. She was not administered any other anti-arrhythmic medication because a resting heart rate of ≤60 bpm was achieved, and she remained asymptomatic and in sinus rhythm for a year. Echocardiography revealed that her left atrial diameter was 44 mm, her left ventricular end-diastolic diameter was 54 mm, and her left ventricular ejection fraction (LVEF) was 40% during regular follow-up examinations.
During April 2020 the patient was admitted several times because of her heart failure, and each time she had rapid atrial arrhythmia. Electrocardiography (ECG) showed atrial tachycardia (AT), with a 250-bpm atrial rate and 2:1 atrioventricular (AV) conduction (Figure 1). Her left ventricle had enlarged to 63 mm and her LVEF had decreased to 22%. Because her atrial tachycardia was closely related to the deterioration of her heart failure, electrical cardioversion was attempted. However, trans-oesophageal echocardiography (TEE) showed left atrial appendage thrombosis. Metoprolol succinate 23.75 mg was administered once daily, diltiazem 15 mg was administered three times daily, digoxin 0.125 mg was administered every other day, and amiodarone was administered intravenously to control her ventricular rate. However, her mean heart rate remained at 120 bpm and she became hypotensive. Therefore, the amiodarone administration was discontinued, because of a lack of efficacy and the presence of hepatic injury, indicated by three-fold increases in the circulating alanine aminotransferase and aspartate aminotransferase activities.

Electrocardiographic trace obtained on admission, showing atrial tachycardia (atrial rate 250 bpm), a 2:1 atrioventricular conduction ratio, and complete left bundle branch block.
Although we had followed the existing recommendations regarding the use of anti-arrhythmic drugs 2 , the results were unsatisfactory. Therefore, we attempted off-label treatment with ivabradine 5 mg twice daily, to which the patient consented. Four days after beginning this treatment, her ventricular rate had decreased and her other symptoms had abated. Her AV conduction ratio had decreased to 4:1 and her atrial tachycardia had decreased to 230 bpm (Figure 2). A continuous ECG recording showed that she had a mean heart rate of 62 bpm throughout the day. Thus, her heart failure had been relieved and she was discharged.
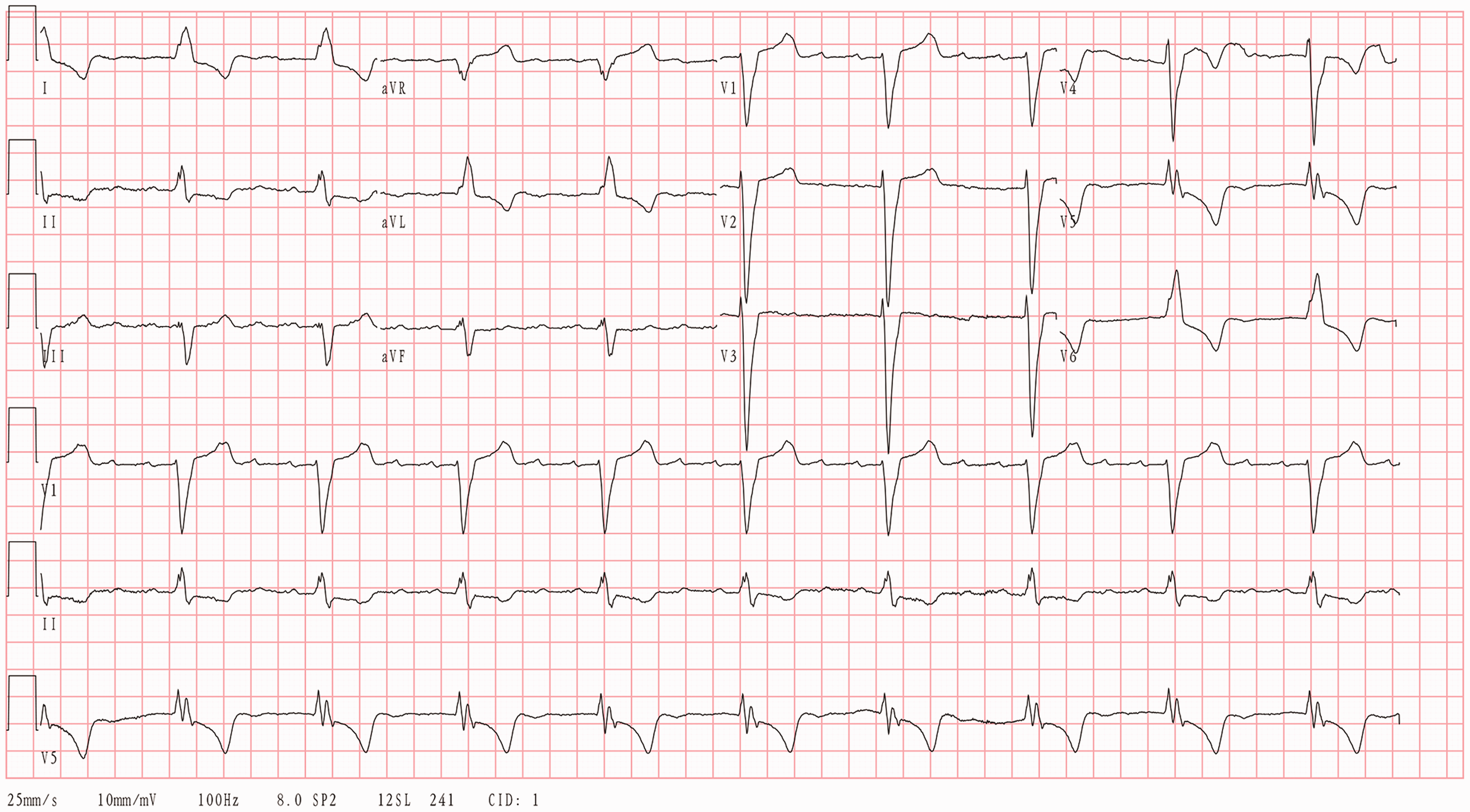
Electrocardiographic trace obtained 4 days after starting treatment with ivabradine 5 mg twice daily, showing atrial tachycardia (atrial rate 230 bpm), a 4:1 atrioventricular conduction ratio, and complete bundle branch block.
One month later, in May 2020, an electrophysiology study (EPS) was performed after the resolution of the atrial appendage thrombus had been confirmed using TEE. Ensite Navx 3D (Abbott Laboratories, Chicago, IL, USA) mapping revealed a re-entry isthmus on the left atrial roof, with a re-entry circumference of 260 ms. The patient’s tachycardia ceased after the ablation of the left atrial roof (Figure 3) and her ivabradine was discontinued after the procedure. In January 2021, 8 months post-ablation, the patient continued to be symptom free.

Electrophysiology study of the patient. The colours depict the progression of atrial depolarisation: red, then orange, yellow, green, blue and purple, in sequence. The presence of adjacent purple and red areas, connected head to tail, is characteristic of re-entry. The isthmus of the re-entrant circuits, where the conduction is slowest, is located on the left side of the roof.
Discussion
For the patient described herein, cardioversion was unquestionably the best therapeutic option, but the presence of thrombus in her left atrial appendage made this impossible. Therefore, we followed the international guidelines for the treatment of supraventricular tachycardia 2 by administering anti-arrhythmic drugs (digoxin, a beta-blocker, and a calcium channel blocker), but this did not stop or significantly ameliorate the arrhythmia. However, the administration of ivabradine slowed the excessive ventricular rate owing to re-entrant atrial tachycardia.
The effects of ivabradine to reduce spontaneous atrial activity and that from pulmonary veins, also referred to as “focal atrial tachycardia”, have been reported previously.3,4 The latest guidelines recommend considering the use of ivabradine if other measures fail in the treatment of focal atrial tachycardia. 2 However, whether this recommendation applies in cases of atrial tachycardia with a re-entry mechanism is unclear. Ivabradine is hypothesised to affect the I(f) current, which is carried by hyperpolarisation-activated, cyclic nucleotide-gated (HCN) channels. HCN4 is the predominant subtype present in the sinoatrial and AV nodes,5,6 and ivabradine has been shown to ameliorate tachycardia by acting on HCN4 channels in the AV nodes of pigs. 7 Therefore, it may be that the same mechanism permits the control of ventricular rate in arrhythmias generated by a re-entry mechanism.
In the present case, the ventricular rate had decreased by day four of ivabradine treatment. P-wave morphology similar to that during tachycardia on ECG suggested that AV conduction had been reduced by the ivabradine, and an EPS confirmed left atrial re-entry tachycardia. The circumference of the re-entrant circuits was minimally affected, implying that the drug has its effects on the AV node rather, than in the atrium.
Ivabradine is a selective I(f) inhibitor that is currently used in the management of patients with stable angina pectoris 8 or heart failure. 9 It has a dose-dependent effect to reduce the ventricular rate, but has no effect on myocardial contractility, which is important for the treatment of heart failure, especially in patients with low LVEF.
In our experience, atrial macro-re-entry-related tachycardias are common in older patients and in those with structural heart disease. Ivabradine is a candidate treatment for atrial re-entry tachycardia when it cannot be treated using established drugs and cardioversion is not possible.
Footnotes
Author contributions
Lanting Zhao and Rong He contributed equally to this work. All the authors read and approved the final version of the manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
This study was approved by the ethics committee of Beijing Tsinghua Changgung Hospital (January 2021). Written informed consent was obtained from the patient for the publication of this case report and the accompanying images.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: This study was funded by the Beijing Municipal Administration of Hospitals Incubation Program (grant number PX2019036), the Beijing Municipal Administration of Hospitals Clinical Medicine Development Program (grant number ZYLX201831), and the Beijing Hospitals Ascent Plan (grant number DFL20190902).
