Abstract
Erythema nodosum migrans (ENM) is usually considered as a rare clinical variant of erythema nodosum and is characterized by unilateral, migratory, relatively painless, nodular lesions. ENM cases are rarely reported and most cases are idiopathic. Therefore, the appropriate treatment modality of ENM is unknown. We report a 72-year-old woman with highly suspected valsartan-induced ENM. She experienced painful, infiltrated, centrifugally spreading, slightly morpheaform, erythematous plaques on the flexor side of her left leg. Her symptoms were relieved after discontinuation of valsartan and temporary administration of oral prednisone once daily (20 mg for the first 7 days and 10 mg for the next 7 days).
Introduction
Erythema nodosum migrans (ENM) is usually considered as a rare clinical variant of erythema nodosum, 1 which is also thought to be a separate clinical entity. 2 ENM typically presents as unilateral, migratory, nodular lesions, which are relatively painless, and plaques that are usually located on the lower extremities.3–4 The overwhelming majority of patients reported with ENM were women aged 30 to 60 years, 5 although patients younger than 20 years have also been reported. 6 ENM subcutaneous nodules persist for a longer period than EN nodules. Because of the paucity of ENM cases, there is little consensus on its appropriate therapy. We report an unusual case of ENM that appeared to be induced by valsartan in a female patient. The reporting of this study conforms to the CARE guidelines. 7
Case presentation
A 72-year-old female patient presented to our clinic because of aggravated symptoms of unilateral plaques on the flexor side of the left leg for 1 month (Figure 1a). She complained of recurring edematous purple-red plaques on her left calf in the past 1 year, with a hard texture and mild pressure pain. The plaques had a tendency to exhibit central clearing resulting in a peripheral ring, and these plaques resolved without treatment. She also had a history of high blood pressure and concurrently took oral nifedipine once daily (30 mg), but had no other complications. One year previously, valsartan capsules (80 mg, once daily) were added to her drug regimen because of a poor antihypertensive effect, and then edematous purple-red plaques appeared after 15 days.
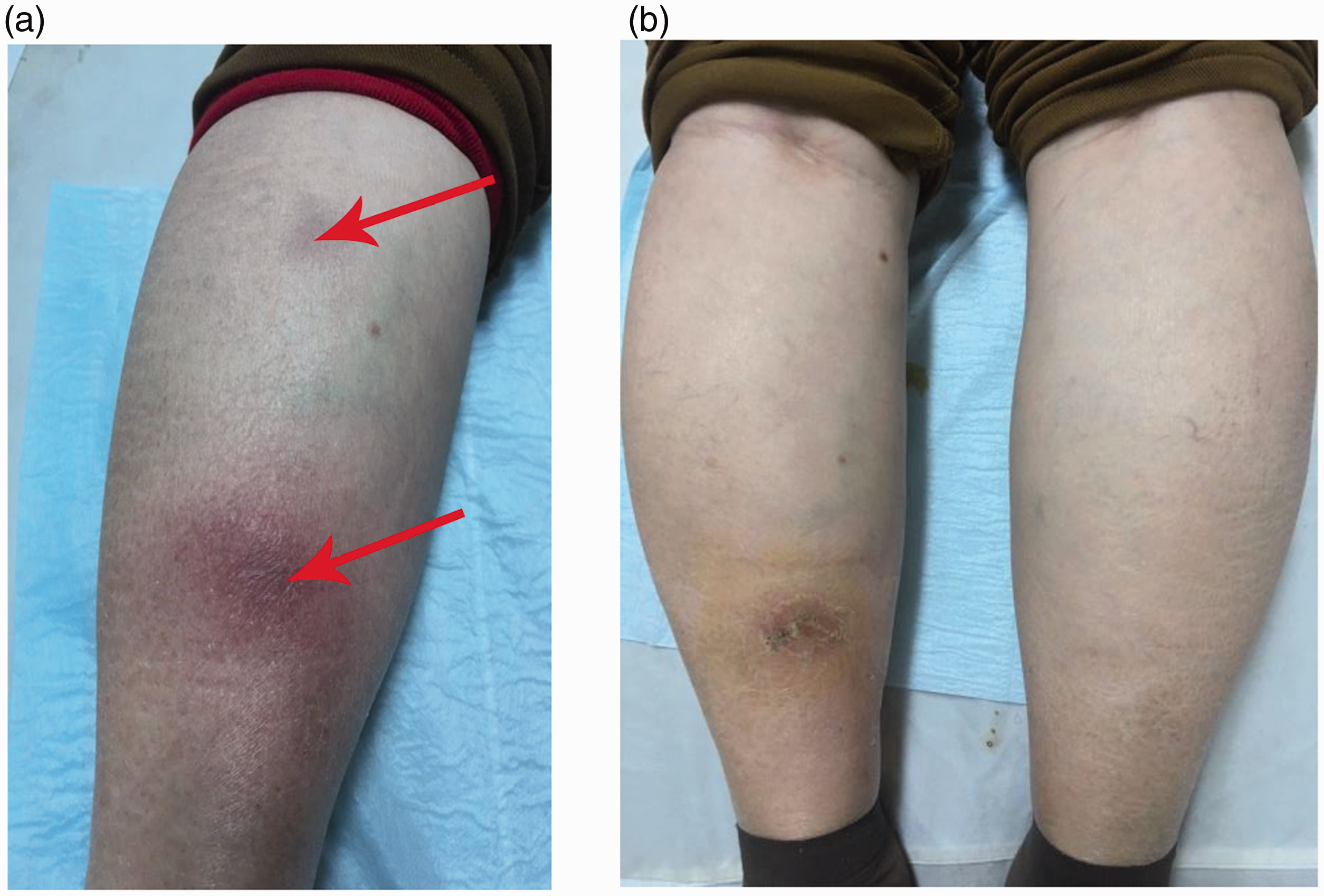
Photographs of erythema nodosum migrans (ENM). (a) A unilateral plaque on the flexor side of the left leg and visible hyperpigmentation can be seen (red arrows). (b) The lesion is completely resolved on the left calf at 14 days after treatment started (the dark spot is due to remaining pigmentation), and there are no skin lesions on the right calf.
At admission, a physical examination showed no other abnormal symptoms in the patient’s respiratory system, circulatory system, or digestive system, except for hypertension. Except for an increased white blood cell count (14.18 × 109/L), neutrophil absolute value (8.01 × 109/L), lymphocyte absolute value (5.43 × 109/L), hemoglobin concentration (155 g/L), and platelet count (356 × 109/L), a laboratory examination showed no other abnormalities. The C-reactive protein concentration, erythrocyte sedimentation rate, fasting blood glucose concentration, liver and kidney function, hepatitis B surface antigen concentration, anti-nuclear antibody concentration, gamma-interferon release test, tuberculin test, chest X-ray, antistreptolysin O titer, and thyroid tests were within normal limits. A histopathological examination showed massive infiltration of lymphocytes and a small amount of neutrophil and histiocyte infiltration. Septal panniculitis, non-specific lymphocytic vasculitis with cuff-like lymphocyte distribution, and histiocyte infiltration around blood vessels of the dermis were observed (Figure 2). With regard to medication, the patient self-reported that she had not taken any other drugs, except for antihypertensive drugs. On the basis of the clinical symptoms, physical examination, laboratory examination, histopathological examination, and medication required, valsartan-induced ENM was highly suspected.

Histopathological examination shows massive infiltration of lymphocytes and a small amount of neutrophil and histiocyte infiltration. (a) Septal panniculitis can be seen (×40, red arrow). (b) Septal fibrosis and a large number of inflammatory cells can be seen (×100, red arrow). (c) Non-specific lymphocytic vasculitis in the superficial dermis can be seen (×200, red arrow). (d) Para-septal extension of inflammation into the lobules can be seen (×200, red arrow). (e) Multinucleated giant cells are clearly present (×400, red arrow). (f) Lymphocytes and a small number of neutrophil and histiocyte infiltration can be seen (×400, red arrows).
Valsartan was discontinued because of its possible adverse reaction. An increased dosage of nifedipine once daily (60 mg) and oral prednisone once daily (20 mg in the first 7 days and 10 mg in the next 7 days) were temporarily administered for 14 days. The patient's symptoms were relieved after 14 days of treatment (Figure 1b). Since then, only nifedipine once daily (60 mg) was maintained, and there was no recurrence during 8 months of follow-up.
Discussion
EN is a type of septal panniculitis, which can be associated with a number of systemic diseases. 8 Predisposing factors of EN may be infections (e.g., Streptococcus, Yersinia, Mycobacterium tuberculosis, Chlamydia, and systemic fungi), drugs (e.g., estrogen/oral contraceptives, sulfa drugs, penicillin, verofenil, and infliximab), pregnancy, and systemic diseases (e.g., sarcoidosis, inflammatory bowel disease, autoimmune diseases, and malignant tumors). Approximately 50% of EN cases are thought to be idiopathic with no cause found.9–10 ENM is a rare variant and special type of EN, and was first described by Bafverstedt in 1954. 11 There have been few studies on ENM, and no case induced by valsartan has been reported.
ENM is more common in women than in men, and its diagnosis is made clinically. ENM is characterized by the presence of unilateral, painless, erythematous to brownish nodules that gradually migrate or expands outward in a centrifugal pattern. These nodules form a plaque with central clearing resulting in a peripheral ring, which may assume a yellowish or morpheiform appearance, and persist for several months to several years, mainly on the lower legs.4–5 Systemic symptoms of ENM are mild or absent, and the pathological manifestations are interlobular panniculitis and lymphocytic vasculitis. In our patient, lesions were typically unilateral on the flexor side of the left leg. The pathophysiology of ENM showed massive infiltration of lymphocytes, and a small amount of neutrophil and histiocyte infiltration, which is in contrast to a previous study. 12 In the late stages of ENM, chronic inflammation predominates. ENM has less systemic symptoms and lesions, and tends to last longer, but shows similar histopathological findings compared with those in classic EN. The diagnosis of ENM was highly suspected in our patient on the basis of her clinical symptoms, and physical, laboratory, and histopathological examinations.
Most cases of ENM are idiopathic in origin. Although an underlying etiology is not usually identified in ENM, an infectious etiology, autoimmune disease, thyroid disease, α1-antitrypsin deficiency, medication adverse effects, and cancer may result in ENM.1,6,13,14 In our case, a physical examination showed no other systemic illness, except for hypertension. With regard to medication, the patient self-reported that she had not taken other drugs, except for antihypertensive drugs. The appearance of lesions was highly synchronized with the use of valsartan. Valsartan works as a specific angiotensin (AT) II receptor antagonist, which selectively acts on the AT1 receptor subtype. This may trigger an immune mechanism. 15 thus leading to the occurrence of ENM. The AT1 receptor is associated with vasoconstriction, water and sodium retention, sympathetic nerve stimulation, aldosterone release, and cell growth and proliferation. AT1 receptors are mainly distributed in the human kidney, heart, vascular smooth muscle cells, adrenal cortex, brain, platelets, and placenta. In the skin, including basal epithelial cells, and eccrine and apocrine glands, the ATII receptor of the AT1 subtype has also been found. 16 Therefore, no other possible predisposing factors could have been associated with ENM in our patient, except for the presence of the antihypertensive drug valsartan. Consequently, valsartan-induced ENM in this case was highly suspected. Valsartan is widely used as an antihypertensive drug in clinical practice, 17 and few adverse skin reactions are reported. One patient developed psoriasis within 2 months after being treated with valsartan. 18 There have been other reports of one case of valsartan-induced photosensitive rash and angioedema, 19 and three cases of valsartan-induced melanoma.20–22 In clinical practice, temporary symptomatic treatment with steroids (oral prednisone) does not improve the recurrence rate of ENM. 23 In our study, the patient's symptoms were relieved after 2 weeks of discontinuing valsartan and temporary anti-inflammatory treatment with prednisone. There was no recurrence of ENM during 8 months of follow-up, and the patient only received once daily nifedipine (60 mg) treatment for hypertension.
To the best of our knowledge, this is the first case of highly suspected valsartan-induced ENM. However, an association of ENM with our patient’s hypertension cannot be excluded, and clinicians need to be aware of this possibility. Further studies are required to establish a clear cause and effect relationship, especially the specific agent involved in the pathophysiology of valsartan-induced ENM. Predisposing factors for ENM need to be identified through a detailed medical history and various examinations. The findings in this case suggest that the correct diagnosis is essential for appropriate management and care of these patients.
Footnotes
Ethics statement
The study was conducted in accordance with the principles of the Declaration of Helsinki and Good Clinical Practice guidelines. The study was approved by the Ethics Committee of the Third Central Hospital of Tianjin (approval number SZX2021-036). The patient provided verbal informed consent for publication.
Author contributions
Yong Liu contributed to the concept and design of the study. Yong Liu, Hui Liu, and Queqiao Bian collected and assembled the data. Yong Liu and Yanmin Guan wrote the manuscript. Yong Liu and Yanmin Guan critically revised the manuscript. All authors have read and approved the final version at the time of submission.
Availability of data and materials
All data generated or analyzed during this study are included in the article. The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
