Abstract
Objective
The long-term outcomes of primary carotid artery closure after carotid endarterectomy (CEA) have not been sufficiently studied. This prospective study was performed to analyze the 5-year outcomes of the non-shunting and primary arterial repair technique for CEA.
Methods
This study involved 150 patients who underwent CEA with the primary arterial closure technique without arterial shunting and completed 5 years of follow-up.
Results
The patients comprised 107 men and 43 women. The 30-day postoperative course was uneventful in 147 (98.0%) patients; however, cerebrovascular accidents occurred in 3 (2.0%) patients. With respect to the long-term results, most cases of restenosis at 5 years were <50%. Two patients developed asymptomatic total internal carotid artery occlusion. Eleven deaths occurred (mortality rate of 7.3%); one death (0.7%) occurred in the first 30 days.
Conclusion
Primary arteriotomy closure provides very good long-term patency. Routine use of patch closure is unnecessary.
Keywords
Introduction
Ischemic stroke is the primary cause of incapacitation and mortality among the adult population worldwide. Extracranial atherosclerotic stenosis of the internal carotid artery (ICA) is the cause of nearly 18% to 25% of all ischemic strokes. The management of this type of stroke varies from optimal medical therapy to carotid revascularization, including carotid endarterectomy (CEA) and carotid artery stenting.1–3 Many randomized controlled trials have proven that CEA significantly decreases the incidence of stroke in most patients with ICA stenosis.4,5 Despite widespread agreement regarding the basic goals of CEA, the optimal CEA procedure continues to be debated. The two main controversies are focused on shunting (whether to use it in all cases, selected cases, or no cases) and the use of patching for arteriotomy closure and its effect on long-term postoperative stenosis. The groups who support the use of routine shunting believe that it lowers the risk of cerebral hypoperfusion, whereas surgeons who are against it or rather more selective in using it argue that shunting by itself might lead to distal embolization and stroke. 6
The main cause of restenosis within 2 months postoperatively is considered to be technical errors; restenosis that occurs 2 months to 2 years postoperatively is mainly due to fibrosis, and restenosis that occurs beyond 2 years postoperatively is believed to be caused by atherosclerosis plaque formation.1,3–5 According to the current literature, most cases of >50% restenosis of the ICA detected during long-term follow-up occur during the first 2 years postoperatively.3–5 The latter issue regarding patching has necessitated researchers to study the fine balance of whether to patch the arteriotomy site. Patch angioplasty can be beneficial in maintaining the lumen diameter in patients with a kinked or long carotid artery; it can also maintain physiological flow in the transitional zone from the CEA area to the distal ICA artery. However, using a patch is not without risks because of the longer clamp time. In addition, complications resulting from the use of two suture lines instead of one and from the patch material itself include pseudoaneurysm formation, infection, arterial rupture, and occlusion. 5
This prospective study was performed to analyze the 5-year outcomes of non-shunting and primary repair for CEA. The patients’ clinical characteristics, lesion categories, and diagnostic and therapeutic approaches were also evaluated. Finally, we compared our results for treating extracranial ICA stenosis with those in the international literature to obtain reproducible conclusions.
Patients and methods
From January 2010, 150 patients who underwent CEA for extracranial ICA stenosis and completed 5 years of follow-up were included in this prospective study. All patients with unilateral or bilateral ICA stenosis amenable to CEA (≥50% stenosis in symptomatic patients and ≥70% stenosis in asymptomatic patients) were consecutively included. All patients were under the care of a single consultant vascular surgeon who performed all surgeries at the same vascular center. The indications for CEA remained the same during the entire duration of the study.
All patients were evaluated by two consultant neurologists and an ophthalmologist as required. The patients’ information was prospectively collected in a dedicated database. Patients with asymptomatic ICA stenosis were treated with best medical therapy.
The diagnostic workup included a detailed history, physical examination, carotid duplex ultrasound, and computed tomography angiography. The morphological plaque types were based on a previously established international classification of plaque morphology 7 and were recorded as follows. In type 1 plaques, the appearance was uniformly and completely (≥90%) sonolucent. Type 2 plaques were >50% sonolucent but contained an echogenic area, and the surface was either smooth or irregular. Type 3 plaques had <50% sonolucency; in other words, they were primarily echogenic. Type 4 plaques were uniformly and completely echogenic. Type 5 plaques contained heavy calcifications that obscured proper assessment.
All data were collected prospectively. The patients were assessed for neurologic deficits immediately postoperatively, during the hospital stay, and at regular clinic follow-ups. The patients were followed up at 3, 6, 12, 18, and 24 months and 3, 4, and 5 years thereafter. All patients who completed 5 years of follow-up were included in this study. ICA restenosis was assessed during each visit using a duplex ultrasound scan. Clopidogrel was stopped 5 days before surgery, restarted in the first week postoperatively, and continued for 1 year. Aspirin was continued until the day of surgery, resumed when the patient started oral intake, and continued for the entire duration of follow-up.
All CEA procedures were performed under general anesthesia using the standard longitudinal neck incision over the anterior border of the sternocleidomastoid muscle. All patients received 5000 IU of unfractionated heparin and 100 mg of aspirin intravenously 10 minutes before clamping of the ICA, common carotid artery, and external carotid artery.
Because our institution has a no-shunt policy, no patients in this study underwent shunting. Arterial backflow and distal stump pressure were used to verify good cerebral perfusion. Direct primary arteriotomy closure (no patch) was used for all CEAs. No methods of monitoring intraoperative intracerebral blood flow were used.
The statistical software IBM SPSS Statistics for Windows, Version 23.0 (IBM Corp., Armonk, NY, USA) and R ver. 3.2.2 (R Foundation for Statistical Computing, Vienna, Austria; https://www.R-project.org/) were used for the data analysis, and Microsoft Word and Excel were used to generate graphs and tables.
Results
A total of 150 patients underwent CEA by the conventional longitudinal technique. The patients comprised 107 men and 43 women with a mean age of 64.71 ± 5.89 years. The median follow-up was 36 months (range, 12–50 months).
The patients’ clinical presentations and signs are listed in Tables 1 and 2, respectively. Only 26 (17.3%) patients had carotid bruit. The mean time interval from presentation to surgery was 6.9 ± 4.60 weeks (range, 2–32 weeks).
Distribution of patients’ presentations.

TIA, transient ischemic attack.
Distributions of patients’ signs.

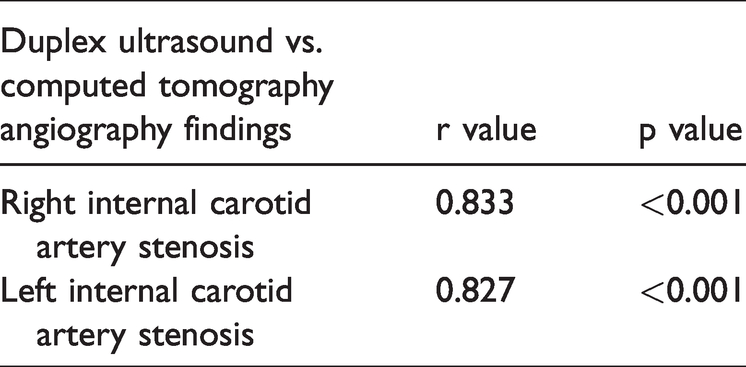
The plaques were mainly type 4 (38.7%); type 1 was the least common (5.3%) (Table 3). Comparison of the duplex ultrasonography and computed angiography findings produced a p-value of <0.001 with a positive r value, indicating equivalent competencies of duplex ultrasonography and computed tomography (Table 4). Type 2 diabetes mellitus and hypertension were the main comorbidities (86.7% and 70.0%, respectively); the patients’ other comorbidities are shown in Table 5. The mean duration of ICA clamping was 17 minutes, and the mean estimated intraoperative blood loss was 162.50 ± 100 mL (range, 20–1200 mL). Undesired intraoperative events occurred in nine patients: tachycardia in six (4.0%), fluctuating blood pressure in one (0.7%), fluctuating ST-segment elevation in lead II in one (0.7%), and frequent ectopic beats in one (0.7%). The postoperative course was uneventful in 132 (88.0%) patients; a cerebrovascular accident occurred in only 3 (2.0%) patients, and the other complications are shown in Table 6. We followed the patients closely for 5 years, particularly for postoperative restenosis. Most cases of restenosis were <50%, based only on the sonographic findings. Two patients developed asymptomatic ICA occlusion, one in the 18th month and the other in the 4th year of follow-up. The latter occurred after the patient chose to discontinue taking aspirin without consulting us.
Plaque morphology.

*Type 1: uniformly and completely (≥90%) sonolucent; Type 2: >50% sonolucent with an echogenic area and either a smooth or irregular surface; Type 3: <50% sonolucent (primarily echogenic); Type 4: uniformly and completely echogenic; Type 5: presence of heavy calcifications obscuring proper assessment.
Pearson correlation between duplex ultrasound and computed tomography angiography findings.
Comorbidities.

*Defined as a glomerular filtration rate of <60 mL/minute/1.73 m2.
Operative details.

Eleven deaths occurred (mortality rate of 7.3%); 1 (0.7%) was an early death that occurred in the first week postoperatively because of anterior myocardial infarction, and the remaining 10 (6.7%) were late deaths mostly with unrelated causes. One patient died of myocardial infarction at 1 year, and two died of myocardial infarction at 2 years. Three patients died of myocardial infarction, lung cancer, and ovarian cancer, respectively, at 3 years. Another three patients died of myocardial infarction, an ischemic cerebrovascular accident, and a hemorrhagic cerebrovascular accident, respectively, in the fourth year. One patient died of breast cancer at 5 years.
Discussion
This study, which was a continuation of our first study published in 2018, was performed to evaluate our results of CEA using the conventional longitudinal approach with no shunt, no patch, and no use of intraoperative cerebral monitoring maneuvers.
Halm et al. 8 performed a large study in which the characteristics of 1972 patients were studied; the combined rate of death or non-fatal stroke within 30 days of surgery was 2.95%. Attigah et al. 9 studied 1290 patients and found that a postoperative neurological deficit was present in 3.7%. In our last study, the 30-day mortality rate was 0.7%, and 3.2% of patients developed perioperative stroke. 6 In the present study, three (2.0%) patients developed cerebrovascular accidents.
Whether to perform routine shunting for all patients, perform selective shunting, or perform no shunting during CEA is a matter of ongoing debate. Groups who advocate the use of shunting support this practice by citing multiple studies in which carotid artery shunting eliminated cerebral ischemia caused by ICA clamping with no increase in thromboembolic complications or arterial dissections.8,9
Excellent results were documented in several large clinical series evaluated in a meta-analysis by Aburahma et al. 10 The studies involved 12,032 patients who underwent shunting, and the perioperative stroke rate ranged from 0.7% to 3.6%.
In contrast, surgeons who do not recommend shunting refer to several reports from the literature showing that shunting might be inessential in around 85% of patients and that possible complications include arterial dissection, air emboli, and acute occlusion. 11
Goodney et al. 12 focused on complete contralateral ICA in patients requiring CEA procedures and found a higher risk-adjusted chance of death or stroke when shunts were used by surgeons who did not use them routinely. Aburahma et al. 10 reviewed several studies and concluded that routine ICA shunting during CEA and selective shunting had similar results in terms of perioperative stroke and 30-day mortality.
In our center, we use a non-shunting technique for all patients, check the backflow frequently, and aim to maintain the mean systolic blood pressure at >90 mmHg. Our results regarding perioperative cerebral events have been comparable to the published literature.
Another issue of debate is the method of arteriotomy closure, which stems from the complexity of the carotid artery anatomy. This anatomical intricacy affects the blood flow dynamics, being laminar (straight) in the common carotid artery and non-laminar (helical or swirling) in the bifurcation. Because of the nature of flow at the branching points, the distribution of wall shear stress (WSS) varies. WSS is defined as a frictional force that is exerted parallel to the vessel wall by the blood viscosity. Up to the point of division, laminar flow is highest; opposite the division point, the flow pattern slows down and becomes complicated by zones of vortices and recirculation. These areas of low WSS are prone to atherosclerosis and intimal thickening, which explains why atherosclerosis develops mainly at major branches and bends of the arterial network (which differs the systemic nature of cardiovascular risk factors).4,13,14
The guidelines of the European Society for Vascular Surgery and the Dutch Society for Vascular Surgery recommend use of the patch angioplasty closure technique. Multiple meta-analyses focusing on patch versus direct closure concluded that patch closure might decrease the risk of ipsilateral stroke, arterial occlusion, and restenosis. 15
Harrison et al. 4 studied the flow patterns throughout the cardiac cycle within the arteries post-CEA. They used a well-described method to reconstruct three-dimensional models using a series of transverse ultrasound images. Their study showed normal flow patterns in some of the primary closure arteries. They also found that patching might result in lower WSS because of an increase in the cross-sectional area in addition to the creation of recirculation and flow separation. Moreover, they studied the difference between using an 8-mm and 5-mm patch and concluded that if a clinical benefit of patching is anticipated, use of a 5-mm patch is favorable because wider patches were associated with extensive regions of low WSS. 4
Bond et al. 5 studied the outcomes of seven randomized controlled trials comparing patch to primary closure. The authors found that patching was associated with a reduced 30-day risk of stroke or death, reduced rates of return to surgery, and reduced risks of arterial occlusion compared with primary closure. They also found a higher rate of anastomotic complications and no superiority of the long-term patency rate compared with the primary closure technique. 16
Currently, the decision to proceed with CEA mainly depends on the degree of stenosis. However, this approach is far from being perfect or sustainable. The degree of stenosis is usually assessed by a noninvasive imaging modality; among such modalities, duplex ultrasonography has been widely used. It not only facilitates assessment of the degree of stenosis but also allows evaluation of the atherosclerotic plaque morphology and provides images of the arterial wall.
Studying plaque characteristics and differentiating vulnerable plaques from stable ones is gaining more popularity in guiding decisions regarding whether to perform an intervention.17–19
Several classification systems are available to describe plaque morphologies. We used an international system that describes plaques according to their echolucency. Topakian et al. 19 found that plaque echolucency was associated with an increased risk of ipsilateral stroke and was an independent predictor of ipsilateral stroke even after controlling for other cardiovascular risk factors. Several classification systems are available to describe plaque morphologies. We used an international system that describes plaques according to their echolucency.20,21 Gijsen et al. 17 studied two approaches to target plaque vulnerability: histopathological classification and biomechanical stress analysis of the fibrous cap. However, they concluded that reliable identification of stable plaques might be a more fruitful approach to reduce the performance of carotid surgical interventions because of the limitation of the spatial resolution of current clinical magnetic resonance imaging techniques. 17
In our group of patients, 45 (30.0%) patients had type 3 plaques, whereas 58 (38.7%) patients had type 4.
We used the direct arteriotomy closure technique for all patients in the present study. The incidence of postoperative cerebrovascular accident was 2.0%. The incidence of restenosis was not remarkable throughout the 5-year follow-up, and all cases were <50% of the carotid artery diameter. Only two patients developed total occlusion, one of whom withheld aspirin therapy without seeking medical advice.
Based on the results of this study, which is an extension of our last series, we still believe that CEA can be performed safely with no shunt and with direct arteriotomy closure with no effect on postoperative stroke or long-term symptomatic restenosis. The use of shunt and patch closure can lead to unnecessary prolongation of the operative and clamp time and unforeseen complications.
In our longitudinal cohort, the 5-year follow-up showed very minimal symptomatic restenosis of the ICA, and such a long duration of follow-up has rarely been investigated in patients who underwent CEA using the primary closure technique. However, our sample was too small to formulate solid recommendations. Larger-sample studies and systematic reviews are required to obtain a higher level of evidence regarding this controversial subject.
Conclusion
General anesthesia is safe for CEA, and a 5-year follow-up is essential for detecting long-term complications of CEA. Routine use of carotid shunting may be unnecessary. Primary arteriotomy closure provides very good long-term patency. Routine use of patch closure is unnecessary.
Footnotes
Acknowledgements
We would like to acknowledge all personnel who assisted in serving our patients. We would also like to thank Niharika S, Ashika R, and Swathi Gupta for assisting with creation of the tables, data lists, and graphs and performance of the statistical analysis. Finally, we thank Dr. K.P. Suresh, PhD (Biostatistics) for reviewing the methodology and results.
Ethics approval
This study was approved by our institutional ethics committee (approval number 142 on 13 July 2021).
Consent to participate
Both verbal and written consent to participate was obtained from all patients.
Consent for publication
Verbal and written consent for publication was obtained from all patients.
Availability of data and material
All raw data in this study are available upon request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
