Abstract
Objective
To compare the prognostic value of International Federation of Gynecology and Obstetrics (FIGO) 2009 and 2018 staging systems in surgical patients with small cell neuroendocrine carcinoma of the cervix (SCNEC).
Methods
We re-staged 64 surgical IB–IIA (FIGO 2009) SCNEC patients according to the FIGO 2018 system and refined stage IIIC of FIGO 2018 based on tumor local invasion. The prognostic factors were analyzed, and the advantages of FIGO 2018 were compared with 2009.
Results
The 5-year overall survival rate (OS) was 78.5% for stage I and 22.2% for stage II (FIGO 2009). In FIGO 2018, there was no difference between stage I and II, and the 5-year OS was 74.1%, 60.2%, and 0% for stage I/II, IIIC1, and IIIC2. After combining stage IIIC with the local invasion stage (T1 was limited to the cervix and vagina; T2 involved the parametrium; T3 involved the pelvic or abdominal cavity), the 5-year OS for stage IIICT1, IIICT2, and IIICT3 was 83.3%, 30.0%, and 0%, respectively.
Conclusions
For stage II SCNEC patients, FIGO 2009 underestimated the prognosis, while FIGO 2018 was more accurate. For stage IIIC, FIGO 2018 might be more individualized and accurate after combining stage IIIC with tumor local invasion.
Keywords
Introduction
Cervical cancer is a common gynecological malignancy and the fourth most common cancer in women. 1 Approximately 95% of cervical cancers are squamous cell carcinomas (SCC) and adenocarcinomas (AC), which are associated with relatively good outcomes. 2 Small cell neuroendocrine carcinoma (SCNEC) is a rare tumor with an extremely poor prognosis; compared with common cervical cancer pathological types, SCNEC has a higher rate of lymph node metastasis (LNM).3–5 While approximately 40% of SCNEC patients develop LNM, the rate is only 10% to 15% for SCC and AC patients.3–5 However, it is uncertain whether LNM affects patients’ prognosis. Most reports have shown that the prognostic factors include LNM in univariate analyses, but only staging is related to prognosis in multivariate analyses.6,7
In 2018, the International Federation of Gynecology and Obstetrics (FIGO) revised the staging system for cervical cancer.8,9 Compared with FIGO 2009, one of the main changes in FIGO 2018 is the division of stage IB from two subgroups into three subgroups (≤4 cm and >4 cm vs ≤2, 2–4, and >4 cm, respectively) according to tumor diameter to screen out patients with tumors ≤2 cm, who can be considered for fertility preservation. Another major change in FIGO 2018 is the addition of stage IIIC according to LNM to predict prognosis. However, data from SCC and AC cases show that stage IIIC is not particularly accurate in predicting prognosis and must be combined with tumor local invasion factors.10,11
The addition of stage IIIC is based on the belief that LNM is closely related to prognosis. 8 Thus, it is very important to clarify the relationship between LNM and prognosis. SCNEC, with its higher LNM rate and worse prognosis than SCC and AC, is a good candidate to validate this change. To this end, we conducted a retrospective analysis of SCNEC data in our hospital. First, we explored whether LNM was related to prognosis, then we compared the new stage with the old stage to verify the effectiveness of the new stage, and attempted to provide an optimization plan.
Methods
Patients
Data for surgically-treated patients with early-stage cervical cancer (stages IB–IIA, FIGO 2009) were reviewed after the patients provided written informed consent. The patients were treated between January 2014 and December 2018 at the Obstetrics and Gynecology Hospital of Fudan University. This study was approved by the Ethic Committee of The Obstetrics and Gynecology Hospital of Fudan University (No. 2016-40).
Among the 5544 patients, 64 patients were diagnosed with SCNEC by pathology based on morphological criteria and immunohistochemical staining for neuron-specific enolase, synaptophysin, chromogranin, and cluster of differentiation (CD)56. The morphological criteria revealed by hematoxylin–eosin staining comprised the presence of small cells with hyperchromatic nuclei and scant cytoplasm, absent or inconspicuous nucleoli, numerous mitotic figures, and extensive necrosis, and all tumors were positive for at least one neuroendocrine marker. 6 Tumors mixed with SCC or AC components were also included. All SCNEC patients underwent type III hysterectomy and pelvic lymphadenectomy, with or without para-aortic lymphadenectomy as primary treatment. All patients were advised to receive cisplatin, or carboplatin if cisplatin intolerant, + etoposide (EP) chemotherapy with or without external-beam radiation as postoperative adjuvant therapy. Follow-up ended in December 2019. The survival outcomes data for seven patients were missing. The reporting of this study conforms to the STROBE guidelines. 12
FIGO Stage
FIGO 2009 staging is based on clinical examination, while FIGO 2018 is based on pathological findings. The diagnostic tools for clinical staging comprised examination under anesthesia, cystoscopy, proctoscopy, imaging evaluation (chest X-ray, and if an abnormality was seen, then chest computed tomography (CT) without contrast was performed + abdominal CT with contrast + pelvic magnetic resonance imaging (MRI) with contrast, or positron emission tomography (PET)-CT). Once the stage has been assigned, it does not change based on intraoperative findings or progression. The details of the staging (2009 and 2018) can be found in the literature published in the official journal of FIGO.8,13 All 64 SCNEC patients were re-staged using FIGO 2018 according to the postoperative pathological report.
Patient, tumor, and treatment variables
The analyzed clinical and pathological variables were patient age, symptoms, human papillomavirus (HPV) type, pathological diagnosis (pre- and post-surgery), FIGO stage (2009/2018), tumor size, LNM, total number of LNs, depth of stromal invasion, lymph-vascular space invasion (LVSI), parametrial extension, lower segment involvement, surgical margin, vaginal involvement, and treatment modalities. The primary endpoint was any cancer-related death. All endpoints were calculated from the date of radical hysterectomy to death, or patients were censored at the last follow-up. The date of death was obtained from our hospital follow-up department.
Statistical analysis
Relationships between stage and clinicopathological features were analyzed by Pearson χ2 test. The Kaplan–Meier method was used to construct survival curves, and the log-rank test was used to examine the statistical difference between the curves. The Cox proportional hazards model was used to estimate the independent prognostic factors for overall survival (OS).14–16 All analyses were performed using SPSS 22 software (IBM Corp., Armonk, NY, USA).
Results
Compared with FIGO 2009, FIGO 2018 involves more prognostic factors
The total number of early surgically-treated cervical cancer patients was 5544, of which 64 (1.15%) had SCNEC. The mean age of the SCNEC patients was 44.48 ± 10.63 years (median, 43.50 years; range, 22–76 years). The total 5-year OS was 67.9%. The main symptom was abnormal vaginal bleeding (71.9%); other complaints comprised abnormal physical examination findings (14.1%) and abnormal vaginal discharge (10.9%). Approximately half of the patients who underwent an HPV test were HPV18-positive (56.7%); only 3.3% were HPV16-positive, and 33.3% were not classified. The remaining 6.7% patients were HPV-negative. These results indicated that more than 90% of SCNEC patients were HPV-positive, and most were HPV18-positive. However, before surgery, the rate of pathological diagnosis for SCNEC was only 48.3% because some SCNEC cases were the mixed type.
Other clinicopathological parameters are detailed in Supplementary Table 1 and were compared between the two staging systems. Because FIGO 2009 is a clinical staging system, it is not accurate in determining vaginal involvement, parametrial infiltration, and tumor size. Therefore, six stage I patients (n = 55) had pathological indications of vaginal involvement, and two stage I (n = 55) patients had parametrial infiltration, while the size of seven tumor specimens (n = 64) did not match the stage standard. However, the FIGO 2009 staging system was also related to LNM (P = 0.023), parametrial infiltration (P < 0.001), tumor size (P < 0.001), and lower uterine involvement (P = 0.014). Compared with FIGO 2009, FIGO 2018 staging, which is based on pathological staging, is more accurate regarding pathological parameters, such as vaginal involvement, parametrial involvement, tumor size, LNM, tumor invasion depth, and LVSI.
FIGO stage (2009/2018) and the degree of LNM were significantly related to prognosis
Kaplan–Meier survival analysis revealed no significant difference in 5-year OS between the different substages of stage I or II in both FIGO 2009 and FIGO 2018 (Table 1). Thus, we conducted the statistical analyses without subdividing these substages. In the univariate analysis (Table 2), LNM (P = 0.049), parametrial involvement (P < 0.001), and FIGO 2009 stage (P < 0.001) were associated with 5-year OS. Five variables with P-values of less than 0.10 in the univariate analysis (involvement of the lower uterine segment, LNM, parametrial involvement, and FIGO stage (2009 and 2018)) and another four clinically important variables (depth of tumor invasion, LVSI, tumor size, and surgical margin) were included in the multivariate Cox regression analysis. The results showed that only FIGO 2009 stage (P < 0.001) had statistical significance, and another two variables, LNM (P = 0.058) and FIGO 2018 stage (P = 0.062) appeared to be related to prognosis.
Analysis of the 5-year OS of SCNEC patients in different subgroups.
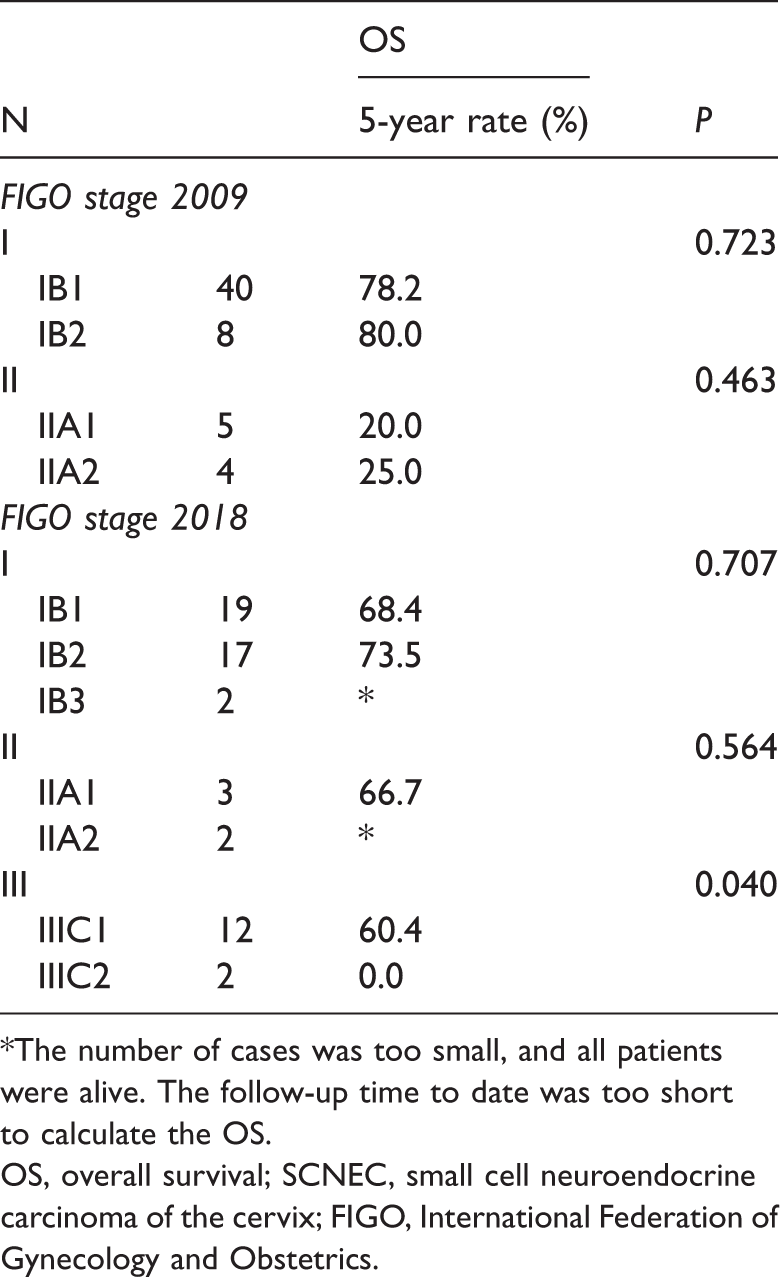
*The number of cases was too small, and all patients were alive. The follow-up time to date was too short to calculate the OS.
OS, overall survival; SCNEC, small cell neuroendocrine carcinoma of the cervix; FIGO, International Federation of Gynecology and Obstetrics.
Univariate analysis of prognostic factors in patients with SCNEC.

*The number of cases was too small, and all patients were alive. The follow-up time to date was too short to calculate the DFS or OS.
DFS, disease-free survival; OS, overall survival; HPV, human papillomavirus; LNM, lymph node metastasis; LVSI, lymph-vascular space invasion; FIGO, International Federation of Obstetrics and Gynecology.
Another interesting finding was that the LNM ratio (number of metastatic lymph nodes/total number of resected lymph nodes) was also significantly associated with prognosis. The mean number of lymph nodes removed per patient was 21.42 ± 0.939 (median, 20.00; range, 10–51). The mean LNM number in each metastatic patient was 4.94 ± 0.864 (median, 4.00; range, 1–12), and the mean LNM ratio was 0.201 ± 0.156. According to the receiver operating characteristic curve (ROC), we confirmed that the LNM ratio was more effective than the presence of LNM in predicting survival (area under the curve (AUC): 0.631 vs 0.604, respectively), and the calculated optimal threshold value was 0.20 (Figure 1a). According to this threshold value, patients were divided into different degrees of LNM: non LNM group (ratio = 0), low LNM group (ratio ≤0.20), and high LNM group (ratio >0.20). After the LNM status was replaced by the degree of LNM, multivariate Cox regression analysis showed that FIGO stage 2009 (hazard ratio (HR): 1.85, 95% confidence interval (CI) = 1.34–2.56; P < 0.001), FIGO 2018 stage (HR: 1.63, 95% CI = 0.92–2.87; P = 0.015), and the LNM ratio (HR: 2.52, 95% CI = 1.36–4.67; P = 0.003) were independent prognostic factors.

(a) Receiver operating characteristic (ROC) curve for lymph node metastasis (LNM) status, ratio, and degree. The areas under the curve (AUC) for LNM status, ratio, and degree were 0.604, 0.631, and 0.635, respectively. When the Youden index was the highest, the true positive rate (TPR) was 0.357, the false positive rate (FPR) was 0.040, and the ratio was 0.204. (b and c) Five-year overall survival (OS) of patients with small cell neuroendocrine carcinoma of the cervix (SCNEC) using the International Federation of Gynecology and Obstetrics (FIGO) stage 2009 and 2018 guidelines. d and e. FIGO 2018 stage IIIC combined with local invasion factors and LNM degree and location.
The optimized staging system (FIGO 2018) can predict prognosis more accurately
We also compared the prognosis according to the two staging systems. The 5-year OS of patients with stage I and II SCNEC (FIGO 2009) was 78.5% and 22.2%, respectively (Figure 1b). There was no difference in 5-year OS between FIGO 2018 stage I and II, and the 5-year OS for FIGO 2018 stage I/II, IIIC1, and IIIC2 was 74.1%, 60.2%, and 0%, respectively (P = 0.003; Figure 1c). We further divided stage IIIC into IIICT1, IIICT2, and IIICT3 according to pathological findings (T1 was limited to the cervix and vagina without parametrial infiltration; T2 comprised parametrial involvement; and T3 comprised pelvic or abdominal cavity involvement). The 5-year OS of stage IIICT1, IIICT2, and IIICT3 was 83.3%, 30.0%, and 0%, respectively (P = 0.010; Figure 1d). Another interesting finding was that when stage IIIC was divided into stages IIICN1a, IIICN1b, and IIICN1c according to the degree and location of LNM (N1a was limited to the pelvic cavity, with an LNM ratio ≤0.20; N1b was limited to the pelvic cavity, with an LNM ratio >0.20; and N1c involved metastasis to the para-aortic LNs), the 5-year OS was 80.0%, 26.7%, and 0%, respectively (P = 0.016; Figure 1e).
Discussion
In our study, the FIGO 2009 staging system was related to LNM, parametrial infiltration, tumor size, and lower uterine involvement, which may be related to prognosis, suggesting that the FIGO 2009 staging system has a role in predicting prognosis. In fact, before 2018, most studies reported that staging (FIGO 2009) was the consistent prognostic factor for SCNEC; other factors comprised treatment modality, LNM, tumor size, and age.6,7,17–20 For example, Wang et al. analyzed 179 patients who were initially treated with surgery and those who were treated with chemoradiotherapy. 7 Multivariate analysis showed that for the entire study population, stage (FIGO 2009) and the presence of LNM (+ vs −) were independent prognostic factors. However, in cases undergoing surgery as the primary treatment, only FIGO (2009) stage was an independent prognostic factor. 7
Many factors affect the prognosis of cervical cancer, such as parametrial involvement, tumor size, LNM, surgical margin, tumor invasion depth, differentiation grade, and LVSI. 21 The National Comprehensive Cancer Network (NCCN) guidelines classify these factors into high-risk (LNM, surgical margin, parametrial involvement) and medium-risk (LVSI, tumor invasion depth, and tumor size) groups. Compared with FIGO 2009, FIGO 2018 was associated with more prognostic factors in this study, including two medium-risk factors: tumor invasion depth and LVSI, consequently leading to a more accurate prediction of patient outcomes. Meanwhile, FIGO 2009 underestimated the prognosis of most stage II patients, which might negatively affect patients psychologically, and which is not conducive to accurate treatment.
Compared with FIGO 2009, another major change in the 2018 staging is the addition of stage IIIC, based on LNM, which is based on the theory that LNM can affect prognosis and which is supported by several studies and by our results.22,23 In one study, the 5-year OS of patients with SCC was 88% in the non-LNM group compared with 57% in the LNM group. Therefore, this change in staging is basically correct; however, some researchers suggest that optimization is required.11,24 LNM in cervical cancer is related to multiple tumor characteristics, such as tumor size, LVSI, tumor invasion depth, and parametrial involvement.22,25 LNM-positive patients are classified as stage IIIC in FIGO 2018, which be inaccurate. Thus, some scholars have combined LNM with local invasion to improve the ability of the staging to predict prognosis. One study including SCC and AC showed that survival of patients with stage IIIC1 disease differed significantly according to T stage, (5-year rates: 74.8% for T1, 58.7% for T2, and 39.3% for T3). 11 In this study, we found similar results for SCNEC, suggesting that this strategy might be applicable to all types of cervical cancer. In addition, prognosis differed in patients with different LNM locations or numbers (LNM involving the common iliac region vs lower regions, ≥3 vs 1–2, respectively).24,26 Therefore, as in our study, it is reasonable to refine stage IIIc into subgroups according to the LNM degree and location.
The novelty of our study is that SCNEC, in which LNM occurs easily, was selected to validate the effectiveness of FIGO 2018, especially for stage IIIC. However, our study had limitations. First, the number of cases was small because of the rarity of this disease. Second, our data did not include patients with advanced-stage disease, who are not suitable candidates for surgery.
Conclusion
FIGO (2009/2018) stage and LNM ratio are important prognostic factors in SCNEC. For stage II SCNEC patients, FIGO 2009 underestimated the prognosis, while FIGO 2018 assessed prognosis more accurately. For stage IIIC SCNEC, FIGO 2018 might be more individualized and accurate when combined with tumor local invasion as well as the LNM degree and location.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605211067397 - Supplemental material for Data from small cell neuroendocrine carcinoma of the cervix: FIGO 2018 staging is more accurate than FIGO 2009
Supplemental material, sj-pdf-1-imr-10.1177_03000605211067397 for Data from small cell neuroendocrine carcinoma of the cervix: FIGO 2018 staging is more accurate than FIGO 2009 by Yunqiang Zhang, Jingxin Ding and Keqin Hua in Journal of International Medical Research
Footnotes
Availability of data and materials
The dataset supporting the conclusions of this article is included within the article and its additional files.
Author contributions
KQ Hua: study conception and design. YQ Zhang: data collection and analysis, manuscript writing. JX Ding: data collection and manuscript editing. The manuscript has been read and approved by all the authors. The requirements for authorship have been met. Each author believes that the manuscript represents honest work.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
