Abstract
Objective
To investigate the effect of atorvastatin on serum periostin level and blood eosinophil count in patients with asthma.
Methods
Patients diagnosed with asthma were enrolled and randomised into an intervention or placebo group, to receive 40 mg atorvastatin or similar placebo, daily, for 8 weeks. Spirometry was performed at baseline, and at the end of weeks 4 and 8; patients also provided blood samples and completed an asthma control test (ACT) at baseline and at the end of week 8. Primary study outcomes were blood eosinophil count and serum periostin levels.
Results
Eighty patients completed the study (40 per group). Mean ACT scores were similar between the intervention and placebo groups at baseline (17.95 ± 3.75 versus 17.98 ± 3.77, respectively), and improved in the intervention group (19.88 ± 3.28), but remained unchanged in the placebo group (18.6 ± 3.26) during the treatment period. No statistically significant differences in spirometric changes, blood eosinophil count or serum periostin levels were observed between the groups during the treatment period.
Conclusion
Spirometric parameters and inflammatory markers did not change significantly in response to atorvastatin treatment, and did not differ between the placebo and intervention groups.
Introduction
Asthma is a common chronic disease affecting both children and adults, with a worldwide prevalence of more than 300 million people that is estimated to rise each year. 1 The social impact of asthma is significant, it is associated with many school and work days lost to absenteeism, and it represents a great economic burden. A 2017 study estimated overall annual costs per patient with asthma to be approximately 3100 USD in the USA and 1900 USD in Europe, with over half of asthma-related costs caused by a small percentage of patients (less than 10%) with severe asthma. 2 Several guidelines differ in their diagnostic criteria for asthma, but most state that asthma should be suspected in patients with signs and symptoms that include wheezing, cough, dyspnoea, and chest tightness, and should be confirmed by diagnostic tests, such as spirometry. 3
The main asthma symptoms result from bronchoconstriction, mucus hypersecretion, airway inflammation, and hyper-responsiveness. 4 Several inflammatory cells, such as mast cells and lymphocytes that release specific types of cytokines (e.g. interleukins [ILs]), are increased in the airways of patients with asthma, and chemical mediators, such as macrophage-derived chemokines, are expressed in airway epithelial cells, helping to attract inflammatory cells to the patient's airways. 5 Eosinophils are differentiated forms of granulocytes that release toxic granule proteins and reactive oxygen species in defence against pathogens. They also release chemokines, including chemokine (C-C motif) ligand 5 (CCL5, also known as RANTES), chemokine (C-C motif) ligand 11 (CCL11, also known as eotaxin), and C-C motif chemokine ligand 3 (CCL3); cytokines, such as IL-4, IL-5, IL-13, and IL-25; lipid mediators; and other growth factors. In addition, increased eosinophils in blood – eosinophilia – may cause airway damage and remodelling, and is associated with severe asthma and an increase in exacerbations.6,7 Eosinophilia is observed in approximately half of patients with asthma, but cannot be used as an asthma diagnostic marker, due to its presence in several inflammatory conditions. Thus, research is ongoing to find more specific markers for asthma diagnosis.6,8
Periostin, an extracellular matrix protein of the fasciclin family, has been identified as a downstream molecule of IL-4 and IL-13, and is associated with subepithelial fibrosis in bronchial asthma. 9 Periostin is suggested to be a systemic marker of eosinophilic airway inflammation and is overexpressed in the lungs of patients with respiratory disorders, specifically asthma; however, it has not been significantly correlated with forced expiratory volume in the first second (FEV1).10,11 Sputum and serum periostin levels have been associated with the proportion of eosinophils in sputum, but may not have sufficient ability in predicting eosinophilic asthma. 12 Research into the role of periostin in asthma progression, and into the use of serum periostin levels as a marker of asthma severity or treatment efficacy, is ongoing. 13
The complex aetiology of asthma and associated treatment difficulties have resulted in a continuing search for more appropriate treatment, targeting inflammation.
14
Atorvastatin has shown beneficiary effects in reduction of inflammatory cell infiltration and airway hyperresponsiveness, by reducing serum levels of inflammatory markers, such as transforming growth factor (TGF)–
Patients and methods
Study population
The present triple-blind, placebo-controlled, randomized clinical trial (RCT) was conducted in Shiraz, Iran, between January and July 2018. The study protocol was approved by the Ethics Committee of Shiraz University of Medical Sciences (Ethics code: IR.SUMS.MED.REC.1396.111) and was recorded in the Iranian Registry of Clinical Trials (IRCT at https://www.irct.ir/user/trial/15332/view; code: IRCT2017102216367N2). The reporting of this study adheres to CONSORT guidelines. The required study sample size was calculated to be 34 per group, allowing for a standardized effect size of 0.65, using previously published results. 17 Considering a 95% confidence interval (CI), study power of 80%, and the chance of lost to follow-up, 40 patients were planned for each group.
Patients aged 18–60 years with a history of asthma for >1 year, who were referred during the study period to the Lung Clinic of Shahid Faghihi Hospital and Motahari Clinic, both in Shiraz, Iran, were considered for study inclusion. Additional inclusion criteria comprised: no history of smoking during the previous 3 months or <10 pack/year, no history of lower respiratory tract infection during the previous 4 weeks, and no oral steroid or statin use during the previous 3 months. Exclusion criteria were: use of drugs that interact with statins, such as antifungals, macrolide antibiotics, cyclosporine, gemfibrozil, verapamil, and amiodarone; diagnosis of hepatitis or active hepatic disease, myopathy, or myositis; and lactating or pregnant women. Asthma was diagnosed by a pulmonology specialist (SM) according to history taking, physical examination, pulmonary tests, and imaging. The study design and objectives were explained to all prospective participants, and written informed consent was obtained from all patients included in the study, with clarification that they were free to leave the study at any time.
Study design
A simple randomization method was used. A statistician, who was not involved in other steps of the study, assigned patients to two groups A and B, using random allocation software. The researcher collecting the data, the statistician who analysed the data, and the nurse who administered the drugs to patients were blinded to the group allocations, and included patients were enrolled into the intervention or placebo groups based on the randomly assigned codes. The intervention group received 40 mg atorvastatin (Sobhan Pharmaceutical Co., Tehran, Iran), orally, once daily for 8 weeks, and the placebo group received a placebo following the same schedule. The placebo was prepared by the School of Pharmacy of Shiraz University of Medical Sciences, and was identical in shape and colour to atorvastatin, but only contained neutral ingredients without pharmacological effect. Following study commencement, patients who did not adhere to the treatment schedule, did not attend any of the study-period examinations, developed any complications related to the administered drug, or who refused to continue the study, were excluded from the study and the final analysis.
Study parameters
All patients underwent spirometric examination at baseline, and at the end of weeks 4 and 8: FEV1, forced vital capacity (FVC), FEV1/FVC, total lung capacity (TLC), residual volume (RV), RV/TLC, total airway resistance (Raw), and specific airway resistance (sRaw) were recorded for each patient. Patients also completed the asthma control test (ACT), 21 before and after the intervention (at baseline and at the end of week 8). The ACT is a simple five-item method for assessing the adequacy of asthma control by providing a numerical score for asthma control during the previous 4 weeks. The validity of the Persian version of this test, used in the present study, has been confirmed previously. 22
Furthermore, a venous blood sample (7 ml) was obtained from each patient, following an overnight fast, at baseline and the end of week 8, to evaluate white blood cell (WBC) count, blood eosinophil percentage, total cholesterol, triglyceride, low-density lipoprotein (LDL), high-density lipoprotein (HDL), alanine transaminase (ALT), and aspartate transaminase (AST). Collected blood was immediately separated into two samples per patient: 2 ml into an anticoagulation tube containing ethylenediamine tetra-acetic acid that was sent to the immunology and haematology lab at Faghihi Hospital for immediate processing; and 5 ml that was allowed to clot for 30 min at 4°C then centrifuged at 3000
The primary study outcomes were blood eosinophil count and serum periostin levels. Secondary study outcomes were spirometric changes and ACT scores. Patient demographics, including age, sex, weight, height, and smoking status were extracted from the medical records. Participants were categorized by age, either <50 years or ≥50 years, and smoking status was recorded as positive or negative (former and current smoking were considered as positive). Body mass index (BMI) was calculated based on the recorded weight and height.
Statistical analyses
Continuous variables are presented as mean ± SD and categorical variables are presented as
Results
A total of 80 patients were included in the final analysis: 40 patients in each group (Figure 1). There were no statistically significant differences between the intervention and placebo groups in terms of demographic or clinical characteristics, including age, sex, BMI, or smoking status (all
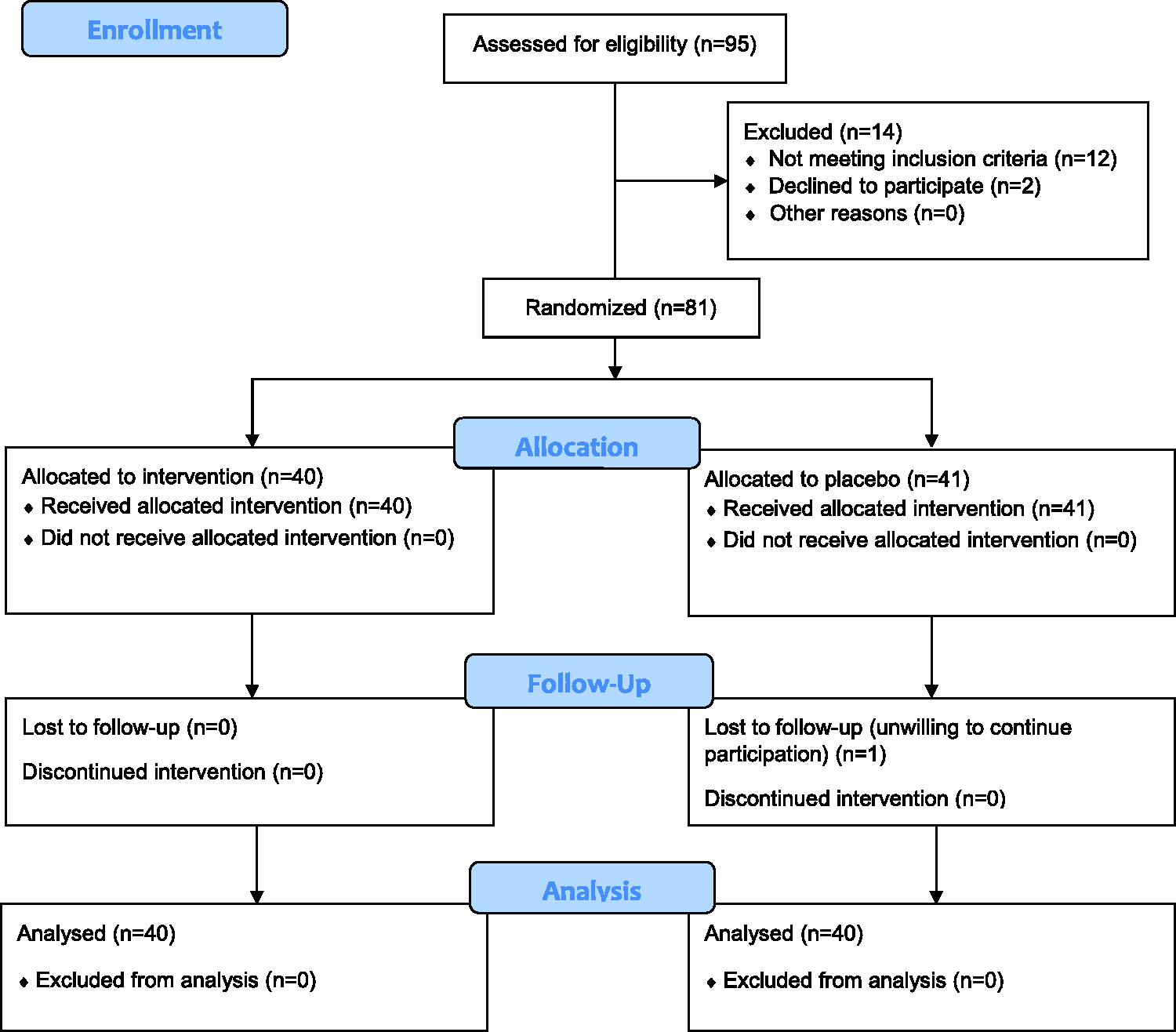
Flow diagram for study enrolment.
Distribution of demographic and clinical variables in patients with asthma allocated to receive 40 mg atorvastatin (intervention group) or placebo (control group) for 8 weeks.

Data presented as mean ± SD or
BMI, body mass index (underweight, <18.5; normal, 18.5–24.9; overweight, 25–29.9; obese, ≥30); GINA, Global Strategy for Asthma Management; LABA, long-acting beta-agonist.
There were no statistically significant between-group differences (all
There was no statistically significant difference in baseline ACT score between the intervention and placebo groups (17.95 ± 3.75 versus 17.98 ± 3.77, respectively;

Comparison of asthma control test (ACT) scores in patients with asthma who received 40 mg atorvastatin, daily (intervention group) or placebo (control group) at baseline and at the end of treatment (week 8). Data presented as mean ± SD;
Analysis of spirometric parameters (Table 2) showed that TLC and RV were statistically significantly reduced in the intervention group at the end of week 8 versus baseline (both
Spirometric parameters at baseline, end of week 4 and end of week 8 in patients with asthma who received 40 mg atorvastatin, daily (intervention group) or placebo (control group).

Data presented as mean ± SD.
FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; TLC, total lung capacity; RV, residual volume; Raw, total airway resistance; sRaw, specific airway resistance.
*Between-group difference at each time-point (independent samples
NS, no statistically significant difference (
Mean WBC count, total cholesterol, HDL, LDL, and triglyceride showed statistically significant changes at the end of the treatment period compared with baseline values in the intervention group (all
Blood parameters at baseline and at the end of treatment (week 8) in patients with asthma who received 40 mg atorvastatin, daily (intervention group) or placebo (control group).
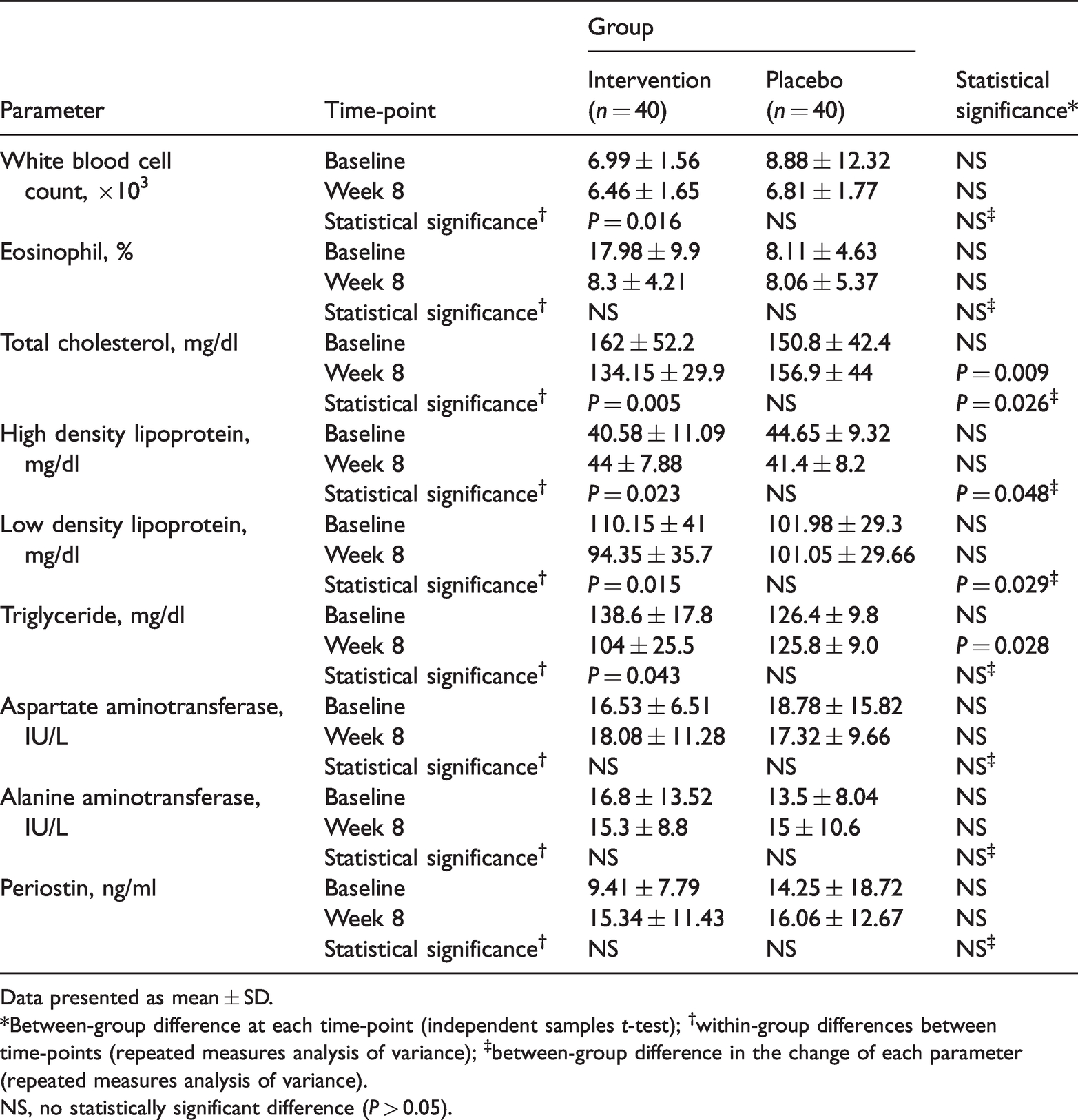
Data presented as mean ± SD.
*Between-group difference at each time-point (independent samples
NS, no statistically significant difference (
Discussion
In the present RCT, the effects of 8-week treatment with 40 mg/day atorvastatin on asthma control and spirometric and serum parameters were compared against placebo in patients with asthma. The ACT scores were shown to be significantly improved in the intervention group but remained unchanged in the placebo group. Furthermore, spirometric parameters, serum periostin level and blood eosinophil counts were not different between the groups. Although FEV1 was found to differ between the groups at the end of week 8, this was likely due to the nonsignificant inverse changes in FEV1 in the intervention and placebo groups. Each patient’s serum lipid profile was evaluated before and at the end of intervention to determine adherence to treatment, which was confirmed by the significant reduction in serum levels of total cholesterol, LDL, and triglyceride in the intervention group.
Atorvastatin is a cholesterol-lowering drug with anti-inflammatory effects, and animal models have shown its efficacy in reducing inflammatory cell infiltration and airway hyper-responsiveness.15,16 However, variable results have been obtained with human studies regarding its efficacy in treating asthma. Some have reported better asthma control in patients with severe asthma when using statins, while others, like the present study, have reported no significant difference.23–27 In the study by Fahimi et al.,
25
17 adult patients with moderate to severe asthma received 10 mg/day atorvastatin or placebo for 4 weeks in a cross-over design study. Similar to the present study, they reported no difference in spirometric parameters between the intervention and placebo group; however, the studies differed regarding improvement of spirometric parameters over time in the intervention group, as Fahimi et al. reported a significant increase in FVC (
In the present study, ACT scores demonstrated significant improvement in asthma control in the intervention group (from 17.95 ± 3.75 to 19.88 ± 3.28;
Atorvastatin has shown beneficial effects on reducing inflammatory markers, such as tumour necrosis factor-α, C-reactive protein, and ILs in other diseases, such as ischaemic stroke, rheumatoid arthritis, and diabetes.28–30 Accordingly, the present study examined the efficacy of atorvastatin on eosinophil count and serum periostin levels, as these are considered important inflammatory markers in asthma.
12
Nevertheless, the present results showed no significant changes in eosinophil count or serum periostin in either group, and no between-group differences were shown: blood eosinophil count was 8.11 ± 4.63 at baseline and 8.06 ± 5.37 after 8 weeks the placebo group, and 17.98 ± 9.9 at baseline and 8.3 ± 4.21 after treatment in the intervention group, with no statistically significant change in either group or significant between-group difference at either time-point. In a previous study, eosinophil count significantly reduced in the placebo group (from 221.31 ± 134.01 to 204.62 ± 134.64) and intervention group (from 244.35 ± 154.4 to 179.14 ± 117.43; both
As described above, results vary between the few studies addressing the efficacy of atorvastatin on asthma control and inflammatory markers in patients with asthma. Similar discrepancies have been reported regarding the effect of other statins, such as simvastatin, on asthma control and inflammation reduction in patients with asthma.31,32 In addition, wide variations are observed among studies, in terms of disease severity, parameters, and main treatments to control asthma, suggesting that further studies are required to determine the exact efficacy of statins on asthma control. Reviews have indicated that atorvastatin reduces airway inflammation in patients with asthma, but does not improve lung function; however, the investigated inflammatory markers vary among the studies. 20 Investigations into general inflammatory markers, such as eosinophil count and estimated sedimentation rate, have reported the necessity to use more specific markers.26,27 In the present study, the change in serum periostin levels in patients with asthma receiving atorvastatin was investigated as a novel potential biomarker in such patients; 33 but the results showed no significant difference in serum periostin levels between the groups.
The present results may be limited by several factors. Changes in airway tissue, endothelium, or cellular levels were not examined for a more accurate assessment of any change in patients' airway hyper-responsiveness and inflammation. Only clinical and serological parameters were investigated, while histopathological assessment of the patients may have produced more revealing results. Furthermore, the time-course of the present study was short and the sample size was limited, although they were comparable to similar studies. As the intervention included both sexes, and young and middle-aged patients with varying body weights and severity of asthma, the results of the present study may be attributed to a wide variation between patients with asthma. Finally, further research involving a population of patients with elevated levels of blood eosinophils and/or serum periostin, together with uncontrolled severe asthma, may help elucidate the potentially beneficial effects of atorvastatin.
In conclusion, the results of the present RCT showed no significant difference in spirometric parameters between patients with asthma receiving 40 mg/day atorvastatin or placebo for 8 weeks. There was also no significant change in eosinophil count or serum periostin levels, nor any between-group difference in these inflammatory biomarkers. The only significant difference between the groups in this study was an improvement in the asthma control scores in the intervention group. As this clinical finding was not supported by lung function test or inflammatory markers, we cannot conclude significant efficacy of atorvastatin on asthma control, and the clinical effect may have been due to other factors, such as increased patient awareness of controlling asthma. Accordingly, further studies with a larger sample size and longer follow-up periods are required to determine the exact effect of statins, such as atorvastatin, on asthma control. We suggest the measurement of more accurate parameters, such histopathological changes, in future studies.
Footnotes
Acknowledgments
The present article was extracted from the thesis written by Jalal Torkan.
Author contributions
SM designed the study. SM, JT, and MH were responsible for data collection. SM and JT wrote the initial manuscript draft. SM and JT conducted the analyses. SM, JT and MH contributed to writing the manuscript. All authors contributed to the draft and approved the final version of the manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: The present study was financially supported by the Vice-chancellor for research of Shiraz University of Medical Sciences (grant No. 15216).
