Abstract
Sulphur-containing compounds have been linked to colorectal cancer by factors such as the presence of methyl mercaptan in intestinal gas and long-term dietary intake associated with sulphur-metabolizing microbiota. Therefore, this current case report hypothesized that active sulphur metabolism in colorectal cancer results in the formation of sulphur compounds in the intestine and, thus, examined sulphur metabolites possibly associated with sulphur respiration in colon cancer tissues. The patient was a 73-year-old female that underwent laparoscopic right hemicolectomy for ascending colon cancer. During the surgery, colon cancer tissues and normal intestinal mucosa samples were collected. After optimizing the sample concentrations for homogenization (pre-treatment), the samples were stabilized using a hydroxyphenyl-containing derivative and the relevant metabolites were quantified using liquid chromatography with tandem mass spectrometry. The results showed that cysteine persulfide and cysteine trisulfide levels were higher in colon cancer tissues than in normal mucosal tissues. Thus, sulphur metabolism, possibly sulphur respiration, is enhanced in colon cancer tissues.
Introduction
Actively proliferating cancer cells require large amounts of energy. 1 In general, eukaryotic cells gain energy through oxidative phosphorylation using oxygen as an external electron acceptor. However, because cancer cells generally exist in hypoxic environments,2–5 they rely on less efficient substrate-level phosphorylation. This process, likely occurring through glycolysis, occurs in cancer cells even under aerobic conditions and is known as the Warburg effect. 6 As such, the detailed mechanisms underlying energy production in cancer cells remain largely unclear.
In addition to substrate-level phosphorylation, cysteine persulfide, present in mammals,7–9 has been shown to function as an external electron acceptor in sulphur respiration.10,11 There are reports of energy production by sulphur respiration in archaea and bacteria, with sulphur, thiosulfate or dimethyl sulfoxide electron receptors.12–16 However, this current study focuses on sulphur respiration involving cysteine persulfide in mammals. In this type of respiration, the final electron acceptor in the electron transfer system is cysteine persulfide, which is broken down into hydrogen sulphide and cysteine. 10 The enzyme sulfide quinone oxidoreductase (SQR), which transfers electrons from hydrogen sulfide to the mitochondrial ubiquinone cycle, was reported to play an important role in this sulphur respiration system. 10 This anoxic respiration system may be involved in cancer cell growth in hypoxic environments as an alternative oxidative phosphorylation system. The large intestine lumen, which is highly anaerobic,17,18 is a location where this system might be observable.
The association between sulphur-containing compounds and colorectal cancer has been reported from several perspectives. For example, sulphur-containing compounds, such as methyl mercaptan, have been reported present at significantly higher levels in flatulence gas from colorectal cancer patients than from healthy individuals. 19 Methyl mercaptan, produced from L-methionine by an enzyme called methionine ɤ-lyase (MGL), is also known as a gas that causes halitosis. 20 Some intestinal microorganisms that have been linked to colorectal cancer, such as Porphyromonas gingivalis, Prevotella intermedia and Fusobacterium nucleatum, have MGL. 21 Therefore, methyl mercaptan in a colorectal cancer patient may be produced by intestinal bacteria. Recently, long-term dietary patterns associated with sulphur-metabolizing microorganisms, such as Bilophila wadsworthia, have been reported to increase the risk of distal colorectal cancer. 22 Therefore, this current study hypothesized that active sulphur metabolism in colorectal cancer results in the formation of sulphur compounds in the intestine.
This current study investigated the presence of sulphur respiration in colon cancer by measuring the level of metabolites potentially relevant to sulphur respiration in cancerous and normal mucosal tissues from a 73-year-old female patient with colon cancer.
Case report
In August 2020, a 73-year-old female patient was admitted to the Yokohama City University Hospital, Yokohama, Kanagawa, Japan for treatment of ascending colon cancer. When she underwent a medical check-up, the faecal occult blood test was positive. She had diabetes mellitus and hypertension. Physical examination revealed no significant findings. Laboratory test results revealed a positive faecal occult blood test, 1.4 ng/ml serum carcinoembryonic antigen (normal range: <5.00 ng/ml) and 1.4 U/ml cancer antigen 19-9 (normal range: <39.0 U/ml). Colonoscopy revealed elevated lesions located at the ascending colon. Biopsy revealed moderately differentiated adenocarcinoma. Computed tomography showed that the patient had no lymph node metastasis or distant metastasis. The final diagnosis was ascending colon cancer. Consequently, the patient underwent laparoscopic right hemicolectomy in September 2020. During the surgery, cancerous and normal mucosal tissues were sampled (10 cm away from the anal side of the tumour) using a sterile pair of scissors (Figure 1). Tissue from cancerous and non-cancerous areas were collected from two locations in close proximity to each other (Tumour 1 and Tumour 2; Normal 1 and Normal 2). Each sample had an area of 3 mm2 (depth: cancerous tissue, 3 mm; normal mucosa, 2 mm; wet weight: cancerous tissue, 31 and 33 mg; normal mucosa, 18 and 23 mg). To remove adhered stool from the tissue samples, each sample was placed in a bottle containing 50 ml of Otsuka Normal Saline (Otsuka Pharmaceutical Factory Inc., Tokushima, Japan) using a sterile pair of forceps and stirred for approximately 5 s. The samples were then placed in cryotubes, frozen with liquid nitrogen and stored at −80°C until analysis. The time from arterial ligation to freezing in liquid nitrogen was noted (max: 20 min; min: 15 min).

Sample collection points of the specimens taken from a 73-year-old female patient diagnosed with ascending colon cancer. The colour version of this figure is available at: http://imr.sagepub.com.
The study protocol was approved by the Ethics Committee of Yokohama City University (no. B190600051) and was registered with UMIN (no. UMIN000038703). This study was conducted in accordance with the Declaration of Helsinki. Informed written consent was obtained from the patient for the institutional use of her data and results in addition to the publication of this report and any accompanying images. The reporting of this study conforms to CARE guidelines. 23
As a suitable method for analysing the metabolome of colon cancer for sulphur respiration has not yet been reported, tissue sample homogenization was undertaken as a pre-treatment step for metabolomic evaluation. The tissues were homogenized in 10% methanol (w/v) using a ShakeMaster Auto (BMS-A20TP; Bio Medical Science, Tokyo, Japan) at 1100 rpm for 2 min. Next, 50 µl of the homogenates of each sample were pooled to create a 10% quality control (QC) homogenate. The 10% QC homogenate was further diluted to yield 0.1%, 0.2%, 0.5%, 1%, 2% and 5% QC homogenates. These homogenates were treated with 2 mmol/l β-(4-hydroxyphenyl)ethyl iodoacetamide (HPE-IAM) and then centrifuged at 21 500
Turbo spray ionization parameters.

For all the conditions tested, it was confirmed that HPE-IAM remained in the mixtures, indicating that the tested conditions had sufficiently excess levels of the derivative to react with the relevant compounds. Increasing the amount of the tissue specimen in homogenized samples resulted in an increase in the observed levels of cysteine and cysteine persulfide derivatives, with the measurement signals saturating at tissue concentrations ≥2% (Figure 2). As signal saturation represents the upper limit of quantification for the MS used in this experiment, a tissue concentration of 1% was used for all the subsequent measurements.

The homogenate concentrations of β-(4-hydroxyphenyl)ethyl iodoacetamide (HPE-AM) showing that increasing the amount of the tissue specimen in homogenized samples resulted in an increase in the observed levels of cysteine and cysteine persulfide derivatives.
Subsequently, all samples were pre-treated based on the homogenate concentration. For the QC sample, 700 µl of 1% QC sample was mixed with 70 µl of internal standard (IS) ([13C3, 15N1] L-cysteine; CNLM-3871-H-0.25; Cambridge Isotope Laboratories, Tewksbury, MA, USA). Next, 70 μl of 20 mM HPE-IAM methanol solution (N-iodoacetyltyramine; I685880; Toronto Research Chemicals, Toronto, ON, Canada) was added to this solution, and the samples were incubated for 1 h at 37°C using either a heating block or constant-temperature bath and then vortexed. The samples were centrifuged for 5 min at 21 500
For tissue samples, 150 µl of 1% homogenate and 15 µl of IS were mixed, and then 15 μl of 20 mM HPE-IAM methanol solution was added. The samples were incubated for 1 h at 37°C using a constant-temperature bath and then vortexed. After vortexing, samples were centrifuged for 5 min at 21 500
Each sample was analysed in triplicate using LC/MS/MS. The presented results are the mean of three measurements. The levels of IS, which was added exogenously in equal amounts, were consistent across each sample (mean range: 591 412–6 292 013 count per seconds [cps]), indicating reliable measurements. Differences in the levels of endogenous cysteine and its persulfide and trisulfide compounds between the samples were observed, with the mean levels being considerably higher in the cancer tissues than in the normal mucosal tissues (tumour versus normal mucosa: cysteine: 17 640 838 versus 9 719 277.5 cps; cysteine persulfide: 282 652 versus 188 443.5 cps; cysteine trisulfide: 202 599 versus 52 168.5 cps) (Figure 3).
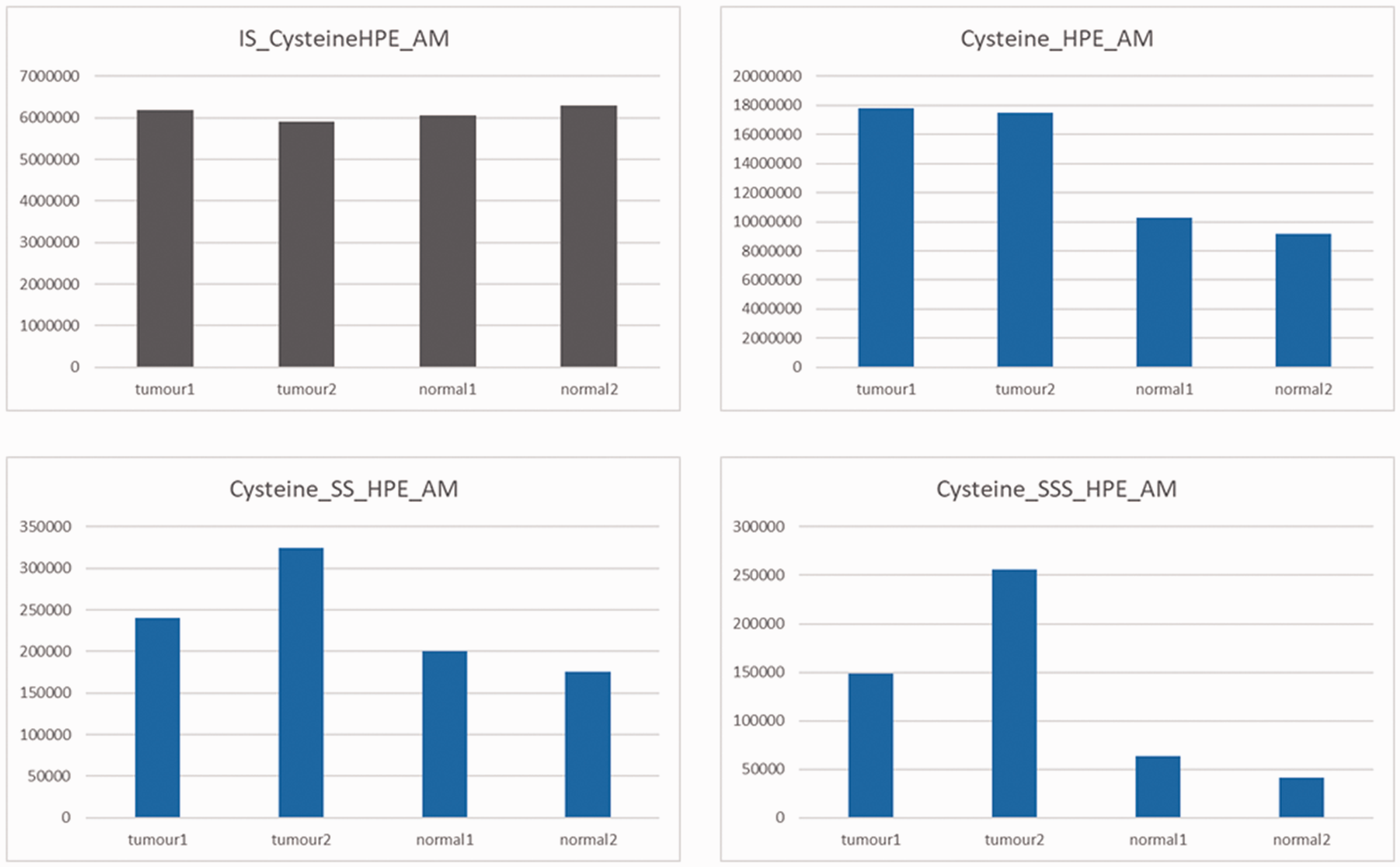
The mean levels of internal standard (IS), cysteine, and cysteine persulfide and trisulfide compounds in tumour and normal tissue specimens taken from a 73-year-old female patient diagnosed with ascending colon cancer. HPE-AM, β-(4-hydroxyphenyl)ethyl iodoacetamide.
Discussion
This current case report and pilot investigation demonstrated that compounds associated with ‘sulphur respiration’ are formed in greater amounts around colon cancer cells than around non-cancer cells. This suggests active sulphur metabolism in cancer cells, implying that sulphur respiration may serve as an energy-generating process under hypoxic conditions.
A comparison of metabolites between colorectal cancer and non-cancerous tissues in colorectal cancer patients has shown that sulphur-related substances, including methionine and cysteine, are increased in colorectal cancer tissues. 24 Although cysteine polysulfide was not measured in this report, methionine is a source of methyl mercaptan in flatus gas,20,21 and cysteine is also a source of cysteine persulfide,7,9–11 which may support the results of this study.
Recent studies concerning chronic obstructive pulmonary disease, diabetic retinopathy and multiple sclerosis have reported cysteine metabolism in human samples.25–27 However, the results of these studies were controversial and the reported levels of cysteine metabolites were inconsistent.25–27 To verify the current preliminary findings, the levels of enzymes associated with sulphur respiration, such as SQR, mitochondrial cysteinyl-tRNA synthetase, cystathionine gamma-lyase, cystathionine beta-synthetase, and 3-mercaptopyruvate sulphurtransferase,7,10,28–30 need to be investigated in tumours. If sulphur respiration is shown to be important for energy production in cancer cells, the pathways involved in this process can be potential targets for anti-cancer drugs.
This current study had several limitations that warrant consideration when interpreting the results. First, to confirm the pathogenic mechanism of a colon cancer disease entity hypothesized here, further experimentation, including additional tissue samples, is required. Secondly, duplicate samples were taken from each of the cancerous and non-cancerous areas. Triplicate samples would have enhanced the robustness of the current findings.
In conclusion, this study shows that metabolites potentially relevant to sulphur respiration are present in increased concentrations in colon cancer tissue. The levels of cysteine and its persulfide and trisulfide compounds were higher in colon cancer tissues than in normal mucosal tissues. To better understand the mechanism underlying sulphur metabolism in cancer tissues, further patients should be studied.
Footnotes
Author contributions
All authors were involved in the conception and design, acquisition of data, analysis and interpretation of data, drafting the article and all provided final approval of the version to be published.
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: This study was supported by a scholarship donation from Chugai Pharmaceutical Co., Ltd. (no. AC-1-20200415123903-602601).
