Abstract
Objective
To investigate the clinical effect of extracorporeal circulation compression perfusion (ECCP) in the treatment of diabetic foot.
Methods
We retrospectively evaluated 89 patients with diabetic foot admitted from January 2017 to April 2019. The patients were grouped according to whether they received ECCP treatment; experimental group: 27 patients, controls: 62 patients. After applying the inclusion criteria and exclusion criteria, there were 21 patients in the experimental group and 21 patients in the control group. Foot microcirculation was evaluated by measuring the percutaneous oxygen partial pressure (TcPO2) and infrared thermography (IRT). Wound healing time and ulcer recurrence rate 1 year after discharge were compared between the groups.
Results
TcPO2 and IRT values in the experimental group differed significantly compared with the control group. Foot ulcer healing time in the experimental group was shorter than that in the control group (17.10 ± 3.08 days vs 25.38 ± 4.40 days, respectively), and the recurrence rate after 1 year in the experimental group was lower than that in the control group (2/21, 9.5% vs 9/21, 42.8%, respectively).
Conclusion
ECCP improved foot microcirculatory perfusion in diabetic foot treatment. ECCP has clinical practicality and may accelerate wound healing speed and reduce ulcer recurrence.
Keywords
Introduction
The number of people living with diabetes in the world is currently 366 million among those aged 20 to 70 years, and this number is expected to increase to 552 million by 2030. 1 Globally, 70% of patients with diabetes undergo amputations, with the number of patients with diabetes with lower limb amputation exceeding 1 million annually. Diabetic foot is a serious complication of diabetes. Infection and peripheral artery disease (PAD) often lead to lower limb amputation, with a patient with diabetes losing a limb every 20 s. 2 The main principle for the treatment of diabetic foot is blood glucose level control, infection control (including the use of antibiotics), 3 improved local blood circulation, and the application of various dressings. However, 84% of patients with diabetic foot still must face a choice regarding amputation. 4
Comprehensive surgical treatment of diabetic foot involves assessing chronic wounds as the starting point, including the repair of infected and non-infected wounds.5− 7 For patients with diabetic foot with ischemic lesions in the lower extremities, it is feasible to reconstruct the main blood vessels and the microcirculation in the lower limbs.4–6 Although endovascular and open surgical techniques have progressed in the treatment of severe limb ischemia (CLI), lower limb amputation remains a continuing challenge, especially for patients with diabetes. 8 Khin et al.8 reported the early results in humans of hypertensive extracorporeal limb perfusion (HELP) to prevent major limb amputation due to ischemia, and the results showed that major amputation was avoided in 8 of 20 patients (40%). Additionally, amputation was delayed in four patients by an average of 4 months during long-term treatment over 22 months (range: 12–54 months). 8
During surgical microcirculatory vascular reconstruction of the diabetic foot, we used ECCP to achieve circulatory perfusion of ischemic limbs under high perfusion pressure. ECCP and HELP are similar treatments with the same mechanism. Both methods expand the stenosis site of the diseased vessel, increase the blood supply to the tissue by increasing blood flow through the cross section of the vessel per unit time, and promote the reconstruction of the peripheral vascular network during perfusion. 8
When we reviewed our department’s case records, we found that some patients received ECCP intervention, while others received routine treatment, and the therapeutic effects differed. We performed this retrospective study to evaluate the correlation between ECCP and changes in foot microcirculatory perfusion.
Materials and methods
Patients
This study was approved by the Ethics Committee of Capital Medical University Affiliated Beijing Shijitan Hospital (approval number: sjtkyll-lx-2017(6)). The reporting of this study conforms to the STROBE guidelines. 9 Written informed consent was obtained from all participants, and their privacy has been protected. In this study, all data were collected by the Hospital Information System (HIS) of Capital Medical University Affiliated Beijing Shijitan Hospital.
This was a retrospective cross-sectional analysis of patients with diabetic foot admitted to our hospital from January 2017 to April 2019. In the retrospective medical record review, the definitions of the variables and which variable data to collect were agreed upon and recorded in detail before data collection, and the definitions and coding methods of the variables were provided to the data extractor. In this way, bias during collecting the variable data was avoided.
We estimated the sample size before beginning the study by comparing the rates of two independent samples. A previous data analysis showed that the 1-year wound recurrence rate was 45% in the control group and 9% in the experimental group. We set the power of a test to 80% and the test standard to 0.05. The calculated sample size was at least 20 cases in each group to reach the statistical standard.
Inclusion criteria: (1) diabetes mellitus; (2) lower limb ischemia (limb numbness, pain at rest, low foot skin temperature); (3) ulcers on the bottom of the foot (ulceration, infection); and (4) imaging data after admission (lower limb arterial angiography) showing a lower limb vascular stenosis rate between 50% and 75%.
Exclusion criteria: (1) poor systemic symptoms; (2) acute foot infection; (3) hypersensitivity to iodine; (4) Charcot foot ulcer (neuropathy); (5) imaging data after admission (lower limb arterial angiography) showing a lower limb vascular stenosis rate >75%.
Initially, we collected the medical records for all cases of diabetic foot after treatment, and recorded the following information: demographic data (age and sex), duration of diabetes mellitus, duration of ulcer, ulcer area, ankle–brachial index (ABI), infrared thermography (IRT) of the affected foot, percutaneous oxygen partial pressure (TcpO2) of the affected foot, and albumin and total cholesterol concentrations. The patients were divided into groups according to whether they received ECCP treatment (Figure 1). Foot microcirculation was evaluated by measuring the TcPO2 and IRT. Wound healing time and the ulcer recurrence rate 1 year after discharge were compared between the groups.

Study flow chart.
Procedures
Routine treatment
All patients underwent wound bacterial culture, and sensitive antibiotics were used according to the culture results. Drugs to improve circulation and control blood glucose, and nutritional support were also given. The wound was treated locally, including debridement and dressings.
ECCP
Four days before debridement, all patients in the experimental group underwent 12 episodes of ECCP (the cumulative treatment time was 72 hours).
The strongest pulsating site 1.5 cm above the femoral artery on the affected side was the determined puncture point, which was injected with 1% lidocaine. After achieving local anesthesia, the femoral artery was punctured anterograde on the affected side. Then, a 6-F arterial sheath was inserted, and 3000 units of heparin sodium was given for systemic heparinization. Intraoperative angiography showed the location of the stenosis(es) (superficial femoral artery, deep femoral artery, popliteal artery, anterior tibial artery, posterior tibial artery, and/or peroneal artery). A balloon catheter was then placed at the beginning of the occluded segment, and the balloon catheter was inflated to block the main blood flow to the affected limb. The balloon catheter acted as a circulatory perfusion catheter (arterial end). The ipsilateral femoral vein was placed into the balloon catheter as the blood reflux tube (venous end), and the arteriovenous line of the extracorporeal circulation perfusion system was connected to the balloon catheter retained in the femoral artery and vein. The treatment cycle was 4 days, and the perfusion tube was removed at the end of the fourth day.
ECCP was performed through the lower limb artery with a perfusion flow rate of 450 mL/min (200 mL/min within 10 minutes after the start of treatment, then adjusted to 450 mL/min for 10 minutes, and maintained at this perfusion rate until the end of treatment). During perfusion, the temperature was set to 35°C to 36°C, the oxygen volume fraction of the membrane lung device was set to 10%, the flow rate was 3 L/min, the perfusion frequency was three times per day, and the perfusion time was 6 hours per treatment. To prevent the circulating blood from the perfused limb flowing back to the human body, tourniquet pressure was applied to the proximal thigh of the diseased lower limb. At the end of each perfusion, the tourniquet was released, and the balloon of the balloon catheter was deflated to the initial state, restoring blood circulation to the patient's limb. Postoperatively, patients received anticoagulation therapy to avoid thrombosis (Figure 2, Figure 3, Figure 4).

Schematic diagram of extracorporeal circulation compression perfusion ① extracorporeal circulation pump (roller pump); ② hemofiltration device; ③ artificial heart-lung machine membrane oxygenator (infant type); ④ extracorporeal circulation blood route (arterial end, venous end).

Balloon catheterization was performed in the interventional catheterization room using extracorporeal circulation compression perfusion therapy.

Extracorporeal circulation compression perfusion in the diabetic foot intensive care unit (DFICU).
Surgical treatment
Surgical debridement was performed in both groups to thoroughly remove necrotic tissue. For patients with toe gangrene, toe amputation was performed first, followed by wound debridement. For patients without bone destruction, secondary wound skin grafting was feasible once sufficient granulation tissue developed. For patients with bone destruction, the necrotic bone tissue was removed first, and after achieving good wound granulation tissue, the wound was sutured without tension. All patients were given anti-infection treatment postoperatively, and the dressing was changed every other day until the wound healed.
Postoperative assessment indicators
The influence of this technique on foot microcirculation was evaluated by measuring TcPO2 and IRT for the patients in the experimental group on day 1, 2, 3, 4, 11, and 18 of intervention. Wound healing time and ulcer recurrence rate 1 year after discharge were compared between the experimental and control groups.
Statistical analyses
IBM SPSS Statistics for Windows, Version 24.0 (IBM Corp., Armonk, NY, USA) was used to calculate the mean standard deviation of continuous data. The independent samples t-test was used for inter-group comparisons, the paired samples t-test was used for intra-group comparisons, and the chi-square test was used for intra-group rate comparisons. Statistical significance was set at P < 0.05.
Results
General patient information
Eighty-nine patients were admitted to the Orthopedic Department, Capital Medical University Affiliated Beijing Shijitan Hospital from January 2017 to April 2019. After applying the inclusion criteria, this retrospective study involved 42 patients diagnosed with diabetic foot. The patients were divided into groups according to whether they received ECCP (n = 27, experimental group; n = 62, control group (no ECCP)). In the experimental group, six patients were excluded: four patients who did not meet the inclusion criteria and two with no follow-up data. In the control group, 41 patients were excluded: 26 who did not meet the inclusion criteria and 15 with no follow-up data. Finally, there were 21 patients in the experimental group (ECCP) and 21 patients in the control group (routine treatment). There was no significant difference in the patients’ general data between the groups (Table 1).
General patient information
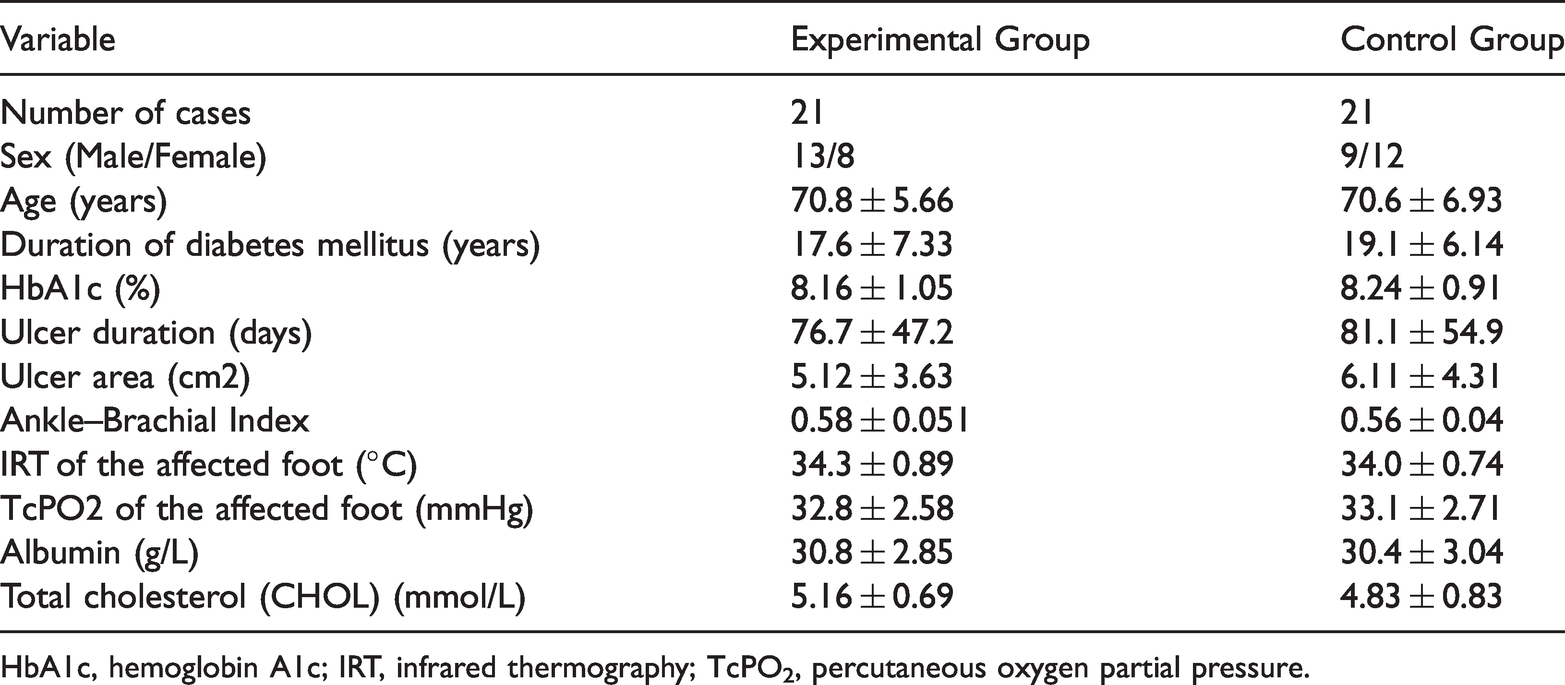
HbA1c, hemoglobin A1c; IRT, infrared thermography; TcPO2, percutaneous oxygen partial pressure.
We reviewed the collected clinical data using the Society for Vascular Surgery Wound, Ischemia, and foot Infection (WIfI) classification system for diabetic foot, which uses a combination of scores for the wound (according to the ulcer depth or extent of gangrene), ischemia (according to the ABI, toe pressure, or TcPO2), and foot infection (according to the International Working Group on Diabetic Foot/Infectious Diseases Society of America (IWGDF/IDSA) criteria) to provide a 1-year risk for amputation and 1-year benefit for revascularization. This approach has benefit over perfusion pressures alone by including associated wound and infection criteria to provide a more holistic wound overview in revascularization decision-making (Table 2).
WIfI system for diabetic foot classification.

WIfI, Society for Vascular Surgery Wound, Ischemia, and foot Infection.
Changing trends in TcPO2 and IRT in the two groups of patients
TcPO2 and IRT values in the experimental group were significantly different and showed an increasing trend compared with the control group at the same time point on day 1, 2, 3, 4, 11, and 18 of intervention (P < 0.05) (Table 3, Table 4).
Percutaneous oxygen partial pressure (TcPO2, mmHg) measured at different time points.

TcPO2, percutaneous oxygen partial pressure.
IRT of the foot measured at different time points in the two groups of patients (T, °C).

IRT, infrared thermography.
Follow-up of wound healing time and ulcer condition after discharge in the two groups
After surgical treatment and dressing changes, the wounds healed completely after a healing time in the experimental and control groups of 17.10 ± 3.08 days and 25.38 ± 4.40 days, respectively. Healing time was significantly shorter in the experimental group (t = 7.06; P < 0.05).
One year after discharge, 0, 0, and 2 cases of foot ulcer recurred during follow-up at 3 months, 6 months, and 12 months in the experimental group. In the control group, foot ulcer recurred in 1, 3, and 5 cases during the 3-, 6-, and 12-month follow-ups. Compared with the control group, the recurrence rate in the experimental group was significantly lower than that in the control group (2/21, 9.5% vs 9/21, 42.8%, respectively) (χ2 = 6.04; P < 0.05).
Typical case in the experimental group
The patient, a 66-year-old woman, was admitted to our hospital mainly because of numbness in her left lower limb for more than 6 months and gangrene of the left great toe for 1 month. Physical examination after admission revealed gangrene in the left toe, and the surrounding skin was red and swollen. There was purulent exudation at the base of the great toe, the skin temperature of the left foot was low, tenderness was obvious, pulsation in the dorsalis pedis artery and posterior tibial artery were absent, and activity in the left foot toes was slightly restricted. The activity of the left ankle was not limited. Auxiliary examination revealed the following: left foot TcPO2: 34 mmHg, IRT: 34.3°C, WIfI grade: 2,2,2, and amputation risk level: H. The patient agreed to undergo ECCP after the admission examination, and 12 episodes of ECCP were performed 4 days before debridement. After ECCP, left lower limb angiography showed that abundant collateral circulation had formed in the P3 segment of the popliteal artery and around the posterior tibial artery and anterior tibial artery, and that the blood supply to the left lower limb had improved significantly. The TcPO2 value after treatment was 36 mmHg, and the IRT was 36.4°C. Amputation of the left great toe was then performed. Intraoperatively, the blood supply to the wound was abundant after amputation, and the wound was sutured in one stage. The wound healed after 2 weeks. The wound treatment cycle for this patient was 2 weeks (Figure 5, Figure 6, Figure 7, Figure 8).

The condition of the wound on the left foot when the patient was admitted to the hospital.
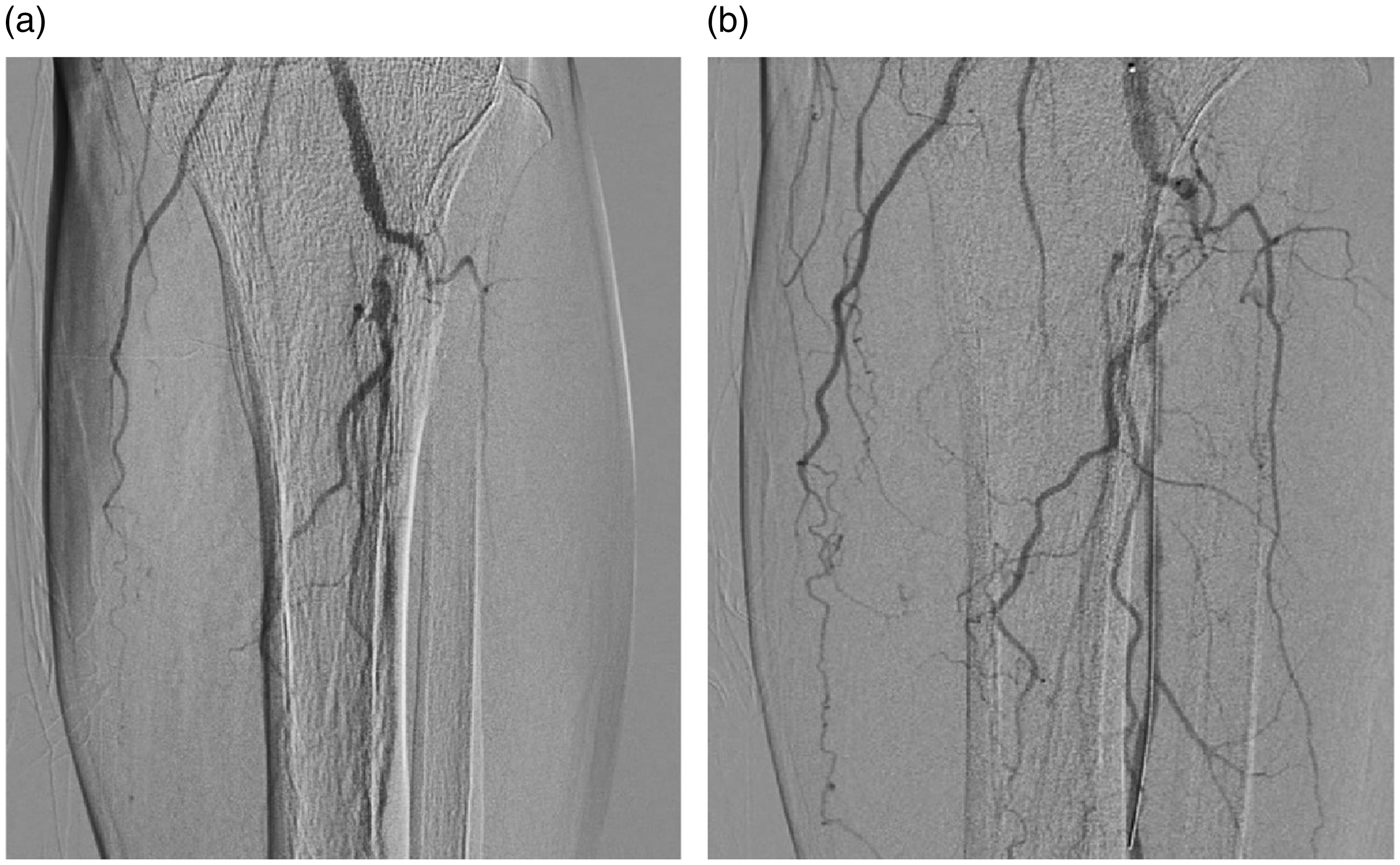
Lower limb angiography a: Below-knee angiography of the left lower limb before extracorporeal circulation compression perfusion showing that the anterior tibial artery was occluded at its origin, the initial segment of the tibiofibular trunk was severely stenosed, the middle and distal segments of the peroneal artery were not visible, and the initial segment of the posterior tibial artery had begun to occlude. b: Three days after extracorporeal circulation compression perfusion, below-knee angiography of the left lower limb showed that abundant collateral circulation had formed in the P3 segment of the popliteal artery and around the posterior tibial artery and anterior tibial artery, and that the blood supply to the left lower limb had improved significantly.
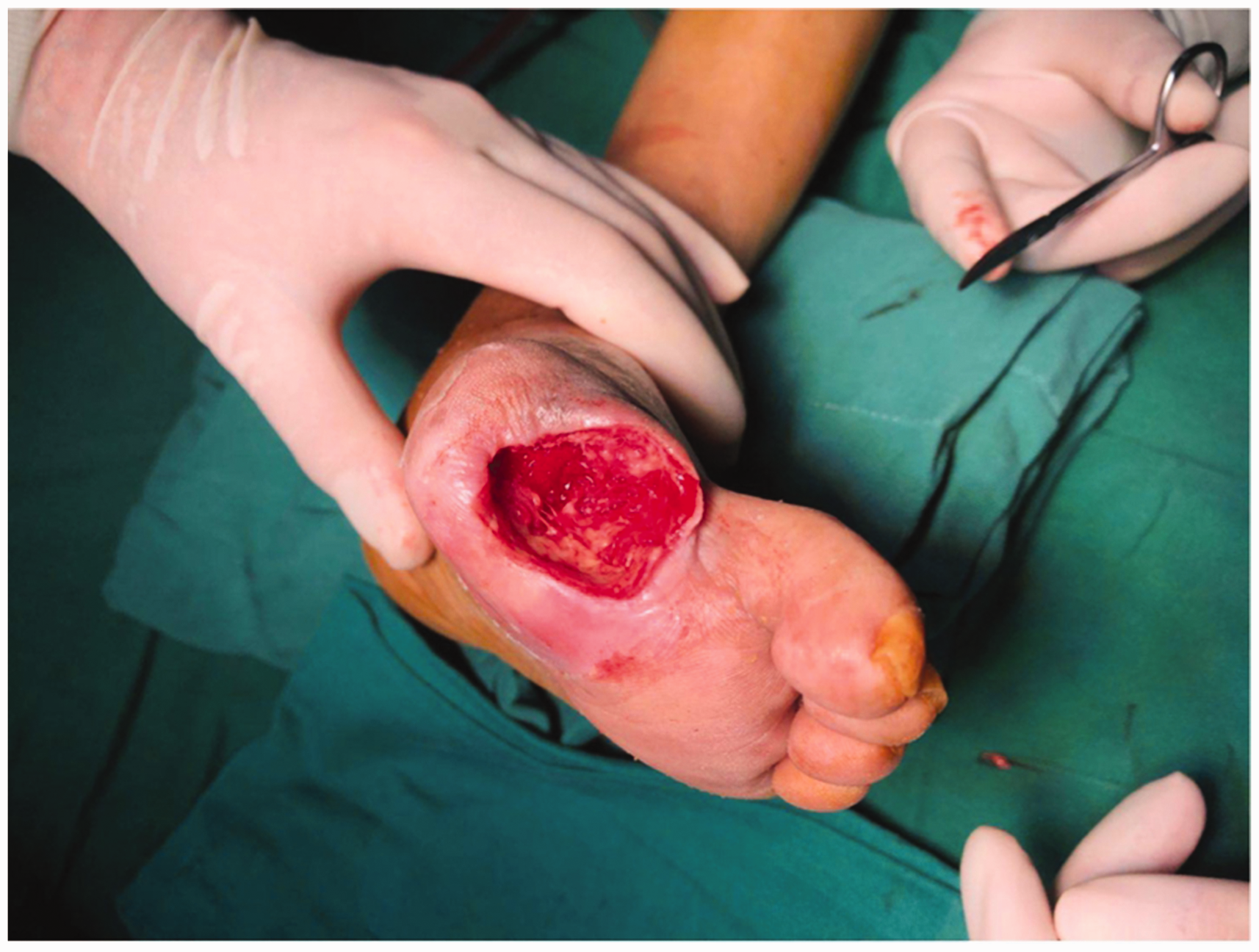
Ulcer wound condition during amputation of the left great toe.
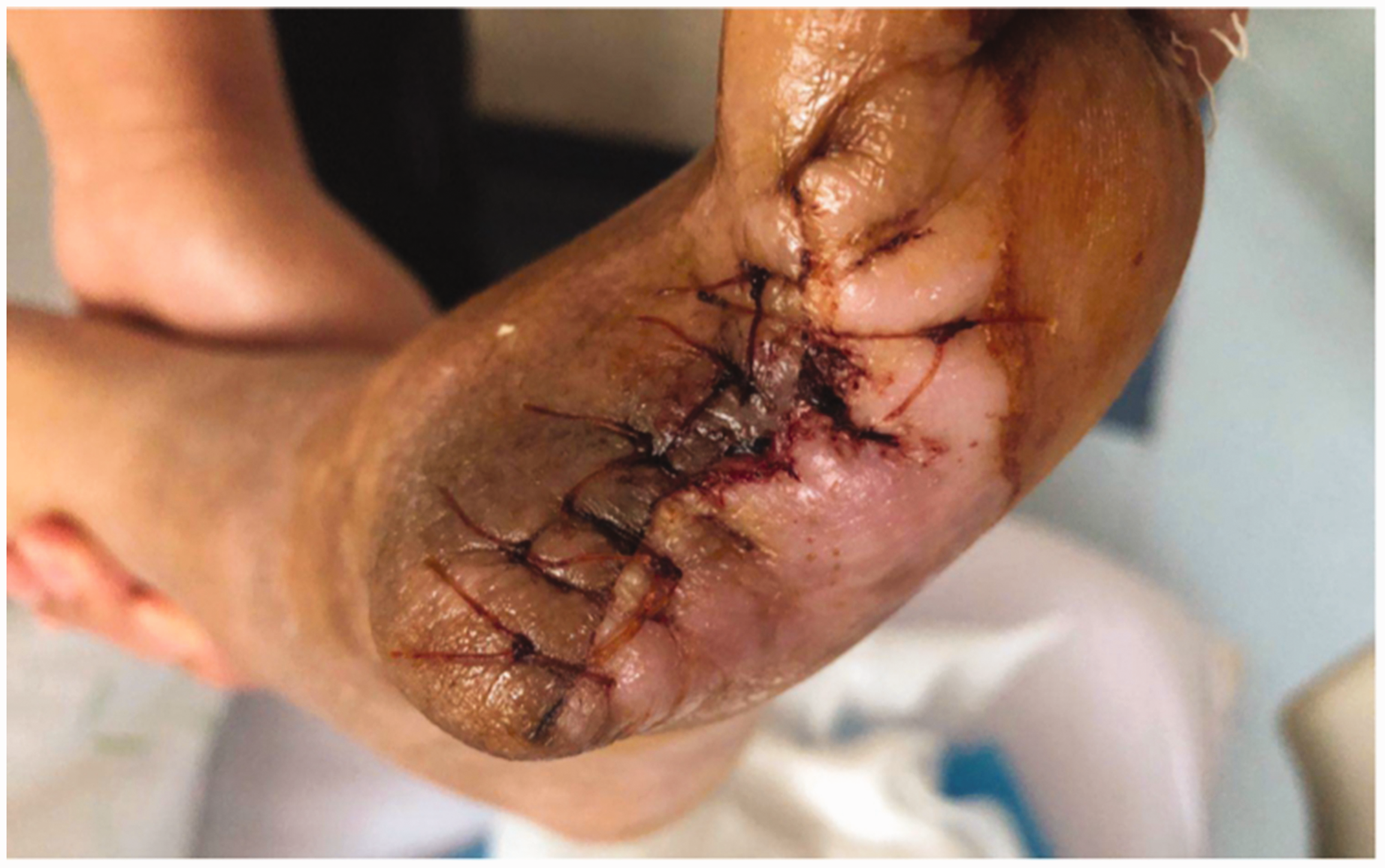
The wound was sutured in a single stage, and the wound eventually healed completely.
Discussion
Severe limb ischemia caused by peripheral artery disease remains a major unresolved public health problem. 10 Although endovascular and open surgical techniques have progressed in the treatment of severe limb ischemia, there are still a large number of patients who cannot undergo revascularization owing to the distal range of the disease, absence of autogenous catheters, or related complications. These patients have a poor quality of life and often must undergo amputation. 11 Lower limb amputation, especially for people with diabetes, remains an ongoing challenge. The rising costs to the health care system, especially with repeated vascular surgery, require greater scientific efforts to achieve simpler and more lasting solutions. 12 To date, some pharmacological and biological therapies have demonstrated the ability to improve limb perfusion, but these do have a proven beneficial effect on the clinical end-point of survival after amputation. 12 Therefore, there is still an urgent need for an effective minimally invasive treatment for limb ischemia.
Khin et al. 8 reported temporarily perfusing ischemic limbs with hyperphysiological blood pressure (HELP) using occlusive balloons to isolate ischemic limbs from the systemic circulation. This technique is performed by designing a special arterial pathway system that can withstand hypercontractile pressure and high flow similar to that obtained during physiological peak exercise. Owing to the temporary increase in vascular shear stress, and because fluid shear stress (FSS) is the driving force in arterial remodeling, the increase in vascular shear stress promotes the formation of collateral vessels.
The results of this retrospective study confirmed that using ECCP in the treatment of diabetic foot had a significant effect by accelerating wound healing and reducing the ulcer recurrence rate, which has clinical practicality. TcPO2 is a mature method for measuring skin perfusion, and the method may be more sensitive than ABI in patients with diabetes. 13 TcPO2 measures the transfer of oxygen molecules to the surface of the skin. A decrease in TcPO2 was detected in patients with peripheral arterial disease in a previous study. 14 In patients with ulcers or amputation wounds, TcPO2 can be used to evaluate the healing potential, as well as the response to surgery or intravascular revascularization.15,16 There is a correlation between a decrease in TcPO2 and the likelihood of healing in patients with ischemic wounds. One study showed that a TcPO2 value <40 mmHg meant a 24% increase in the risk of healing complications. 17 In our study, TcPO2 values in the experimental group were significantly different and showed an increasing trend compared with the control group at the same intervention time point (day 1, 2, 3, 4, 11, and 18). With ECCP for circulatory perfusion of ischemic diseased limbs, the stenosis site of diseased blood vessels expanded, and blood flow through the cross section of blood vessels increased per unit time. The tissue blood supply increased, and reconstruction of the peripheral vascular network was promoted during perfusion. This technique can reconstruct lower limb microcirculation in diabetic foot, promote wound healing, effectively delay the progression of diabetic peripheral vascular disease, and reduce the disability rate of diabetic foot.
Our results are consistent with those of previous studies.18–20 HELP involves the same principle as that of ECCP; both techniques involve isolated perfusion of ischemic limbs. Currently, there are also non-invasive methods to improve the microcirculation of diabetic foot wounds, such as photobiomodulation, hyperbaric oxygen, and transdermal carbon dioxide (CO2) administration. However, ECCP involves isolated perfusion of ischemic limbs, and HELP can expand diseased blood vessels, and increase blood flow and tissue perfusion by increasing vascular endothelial shear force and stress relaxation of vascular smooth muscle. ECCP accelerates wound healing time and effectively delays the course of diabetic foot complicated with peripheral artery disease by increasing circulating blood flow. 21 However, ECCP is associated with side effects. The roller pump causes mechanical damage to the red blood cells in the circulating blood, reducing the number of effective red blood cells, which reduces the oxygen-carrying capacity of the blood. In addition, thrombi form easily during high-flow catheterization under high pressure, resulting in membrane lung and circulatory pipeline blockages.
During our study period, 21 patients underwent ECCP, with a mean total cost per case of $16,493.80 US dollars (USD). One study used meta-analytic data from drug-coated balloon (DCB) studies to compare the cost-effectiveness of endovascular treatment of superficial femoral artery (SFA) disease using DCBs, drug-eluting stents (DES), standard balloon angioplasty (POBA), or bare-metal stents (BMS). 22 The results revealed a baseline cost of $9259.39 USD per patent limb at 1 year in the POBA-first group. The authors also calculated the incremental cost per patent limb for each other strategy compared with POBA, and the results indicated additional costs of $14,136.10 USD/additional patent limb for DCB, $38,549.80 USD/limb for DES, and $59,748.85 USD/limb for BMS. Therefore, considering cost alone, ECCP costs more than when using DCBs and POBAs and less than with DES and BMS. However, owing to regional differences, the results of these data comparisons are inaccurate. Prospective studies are needed to further analyze the cost-effectiveness of ECCP therapy and endovascular treatment of lower extremity arteries.
This study has the following limitations: (1) this was a retrospective analysis rather than a prospective study; (2) this was a single center study; and (3) the sample size was small, and the follow-up duration was only 1 year. Owing to the cost of examination and other factors during the follow-up, we did not obtain the patients’ lower limb vascular computed tomography (CT) angiography data 1 year after therapy, and there was no direct evidence confirming whether establishing improved lower limb microcirculation was still effective 1 year after treatment. However, during the outpatient follow-up, we obtained indirect evidence of a benefit with ECCP, such as lower wound recurrence rate, and better foot TcPO2 and IRT values, compared with no ECCP therapy. Future prospective studies will be performed. The direct evidence of CT angiography data from patients' lower limbs will be added in a future study, and follow-up will be performed for a longer period (e.g., 3 years).
During the treatment of diabetic foot, ECCP was associated with improved foot microcirculatory perfusion. ECCP has clinical practicality by accelerating wound healing and reducing the ulcer recurrence rate.
Footnotes
Acknowledgements
The authors thank all the people who helped during the writing of this paper.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
