Abstract
Objective
In this analysis, we examined differences between rechargeable and non-rechargeable spinal cord stimulation (SCS) devices in patients with pain.
Methods
We conducted a retrospective, longitudinal claims data analysis using a German research database comprising 5 million statutory insured patients (2012–2017). Outcomes of demographics, patient pathways, and health care resource utilization (HCRU) in patients with initial SCS were collected.
Results
Of 150 patients in the database, 73 (49%) received a rechargeable device and 77 (51%) a non-rechargeable device. The average age was 62.5 years (51% female and 49% male patients). A significant decrease over a 3-year follow-up was observed in analgesic prescriptions (−18%), number of patient visits to a physician, and number of patients who were hospitalized. HCRU-related figures for patients with non-rechargeable neurostimulators increased in the last follow-up year whereas the group receiving rechargeable neurostimulators showed a steady decrease.
Conclusions
SCS seems to be an effective way for patients with chronic pain to decrease pain and improve quality of life. Rechargeable devices seem to be superior to non-rechargeable devices owing to greater longevity and were found to be associated with continuous reduction of pain diagnoses, hospitalization, physician visits, and use of pain medication in our study.
Keywords
Introduction
The prevalence of adults with chronic pain in Europe has been estimated to be approximately 19%, which means roughly 16 million patients in Germany are affected by pain. 1 Spinal cord stimulation (SCS) is a therapy for patients with several forms of pain, such as pain owing to neuropathy or ischemia. 2 International and German guidelines consider chronic pain a primary indication for SCS therapy and include diagnoses like failed back surgery syndrome, back pain, and pain in the extremities.
In SCS, the spinal cord is stimulated using implantable systems consisting of electrodes and neurostimulators. SCS systems can differ in terms of positioning of the electrodes (anatomic or paresthesia-based placement), product design, stimulation type, and/or the required energy consumption and its associated energy supply. All systems have in common that electrical impulses are delivered to the spinal cord from a generator via electrode(s) to achieve pain relief for the patient. A variety of devices are available, with different electrical frequencies. Low-frequency neurostimulators are programmed to deliver an electrical current between 40 Hz and 100 Hz whereas high-frequency systems deliver currents up to 10 kHz. 3 The implantation of these systems can be carried out in a one-step or a two-step procedure. The one-step procedure involves implantation of the electrode and the neurostimulator in one operation. In the two-stage procedure, the electrode is implanted and tested over a period between 3 days and 3 weeks using an external stimulator. If the test phase (trial) is successful, the internal pulse generator is then implanted. 4
Neurostimulators can be distinguished according to battery type, with rechargeable (RC) and non-rechargeable (NRC) devices. Systems based on 10 kHz are only available as RC devices. NRC devices must be explanted after a certain period and replaced with a new device in an additional surgical procedure. An RC neurostimulator is recharged from the outside through the skin. The lifetime of the battery varies depending on the manufacturer, ranging between 2 and 5 years. 5 RC neurostimulators can remain in the patient's body for approximately 10 to 25 years. 6 Depending on the lifespan of the device, studies have deemed RC neurostimulators to be superior even with there being a higher initial cost than that for non-rechargeable devices. These studies have highlighted the superior cost-effectiveness and reduced risk of complications owing to less need to change rechargeable neurostimulators. 3 , 7
Although there are sufficient international studies and available, data regarding the cost-effectiveness of SCS therapy in general and comparisons of RC and NRC devices,8–10 there is a need for more data regarding health care resource utilization (HCRU), such as drug prescriptions, physician visits, and hospitalization, with use of these devices. Therefore, the aim of this analysis was to observe the patient pathways with an initial SCS therapy, focusing on the two types of implantable pulse generator (IPG). A further goal was to explore the development of HCRU outcomes with SCS therapy and to compare these with regard to RC and NRC IPGs. 11
Methods
Study design and participants
Statutory health insurance (SHI) claims data analysis
We conducted a retrospective study using longitudinal claims data from the research database of the Institute of Applied Health Research (InGef), which contains approximately 5 million member records from over 60 SHIs nationwide. Approximately 90% of the German population is insured in SHIs; therefore, the SHI databases are an important data source. Data on health claims are transferred directly from health care providers to specialized data centers owned by the SHIs, where all data are anonymized before being entered into the research database. Thus, the study sample is representative of the German population in terms of age and sex and is widely used for real-world evaluation. 12 The reporting of this study conforms to the RECORD statement. 13
The aim of this investigation was to identify patients with an initial implantable neurostimulator from the InGef research database and to monitor them over a period of 6 years (2012–2017). For this purpose, insured persons from the database with OPS codes 5-039.e0, 5-0339.e1, and 5-039.e2 (OPS-classification codes for operations and procedures) were included for the pooled index years 2013/2014. Persons without continuous insurance coverage were excluded, to avoid loss to follow-up and missing data. Figure 1 shows the complete observation period. In the year prior to the index, patients were not allowed to have had any implantations, changes, explants, or revisions of a stimulator. The relevant OPS codes 5-039.e0, 5-0339.e, 5-039.e2, 5-039.f0, 5-039.f1, 5-039.f2, 5-039.b, and 5-039.d were used as exclusion criteria. This study had 3 follow-up years (FU1, FU2 and FU3), each with 365 days. The study population was divided into patients with an RC neurostimulator and those with an NRC neurostimulator.

(a) Observation period and (b) flow of patients with initial implantable pulse generator (IPG) implantation.
Ethical approval and data protection
The analysis did not involve any decisions regarding interventions or the omission of interventions. Accordingly, institutional review board/ethical approval and informed consent from study participants for treatment and publication were not required. Moreover, all individual patient data are anonymized in the research database to comply with German data protection regulations. Key figures based on patient numbers below five were not reported.
Data availability statement
To conduct this study, the authors had access to aggregated, anonymized health care data as per the pre-defined study protocol. Owing to the sensitivity of the data and data protection regulations, the analysis datasets of the current study will not be shared or stored in a public repository. Analysis datasets can be accessed upon request from the Institute for Applied Health Research Berlin (InGef) (
Outcomes and statistical analysis
To describe the study population, age- and sex-specific information was collected for patients with an initial neurostimulator. Furthermore, patient characteristics were determined using the underlying diagnoses, according to the International Statistical Classification of Diseases, 10th Revision, German Modification (ICD-10-GM), at the time of implantation of the neurostimulator. For the observation period, the focus was on the number of patients with HCRU. Therefore, the number of patients with specific prescriptions for analgesics and antiepileptics were counted. Medications were defined using the Anatomical Therapeutic Chemical (ATC) classification system codes. Another measured HCRU figure was the number of patients who visited a radiologist, orthopedist, neurosurgeon, or anesthesiologist. Finally, patients with hospitalization and pain-related hospitalization were counted as well as the number of days of sick leave in patients eligible.
The percentage of patients in the total study population per observation year was determined as part of the statistical evaluation. Additionally, the percentage increase and decrease between the follow-up years was calculated for the patient groups. Furthermore, we determined the percentage increase and decrease for the period from the year before implantation (T-1) to the last year of follow-up (FU3). Significance at a p-value of <0.05 was assessed using the McNemar test for the time comparison between T-1 and FU3 for HCRU variables. For comparisons between the subgroups in FU3, significance with a p-value of <0.05 was determined using the chi-square test.
Results
Study population and demographics
From 2012 to 2017, the InGef research database comprised 4.9 million people with at least 1 insured day over the entire period. Of the 3.1 million people who were fully insured, 153 patients received implantation of a neurostimulator in the index years 2013/2014. The final study population was 150 patients with an initial neurostimulator; three patients had undergone a corresponding procedure in the previous year. Figure 1 shows the process of excluding patients in the database and the resulting target population. Of the 150 patients with an initial neurostimulator, 77 patients (51%) had an NRC neurostimulator and 73 patients (49%) had an RC one. In the follow-up period, 6.7% of the study population died (10 patients), five patients in each subgroup.
The average age of patients was 62.5 (standard deviation ±12) years. Patients with an NRC device were, on average, 1 year older than patients with an RC device. The sex distribution was nearly balanced with 51% women and 49% men. The average age of female patients was 61.9 years, 1 year less than that of male patients. A look at the subgroups revealed that most women (55%) received an RC neurostimulator and most men (58%) received an NRC neurostimulator (Table 1).
Study population and patient characteristics.

RC, rechargeable; NRC, non-rechargeable; IPG, implantable pulse generator; SD, standard deviation; FBSS, failed back surgery syndrome.
When comparing care sectors, the diagnoses of patients at the time of implantation showed a heterogeneous picture. Among inpatients, 49 patients (33%) were diagnosed with post-laminectomy syndrome (M96.1) on the index day of neurostimulator implantation. Among outpatients, 104 patients (69%) were diagnosed with unclassified pain (R52). Other frequent diagnoses among outpatients were back pain (64%) and other intervertebral disc damage (49%). The different types of devices showed no significant differences in terms of diagnosis among inpatients. In the outpatient sector, patients with an RC device more frequently received therapy-specific diagnoses.
Pathway
After the initial implantation of a neurostimulator, 136 patients (91%) continued SCS therapy until the end of the observation period (Figure 2). Fourteen patients (9%; 7 with an RC and 7 with an NRC device) ended SCS therapy, with removal of the device and no further device implantation, and 36 patients (24%) received a second neurostimulator after the first initial device was explanted. A third IPG was implanted in five patients (3%) (Table 2). Data showed that patients with an NRC IPG had a higher rate of explantation (34 NRC vs. 15 RC).

(a) Patient pathway and (b) change from first to second implantable pulse generator (IPG).
Patient pathway and type of procedure.
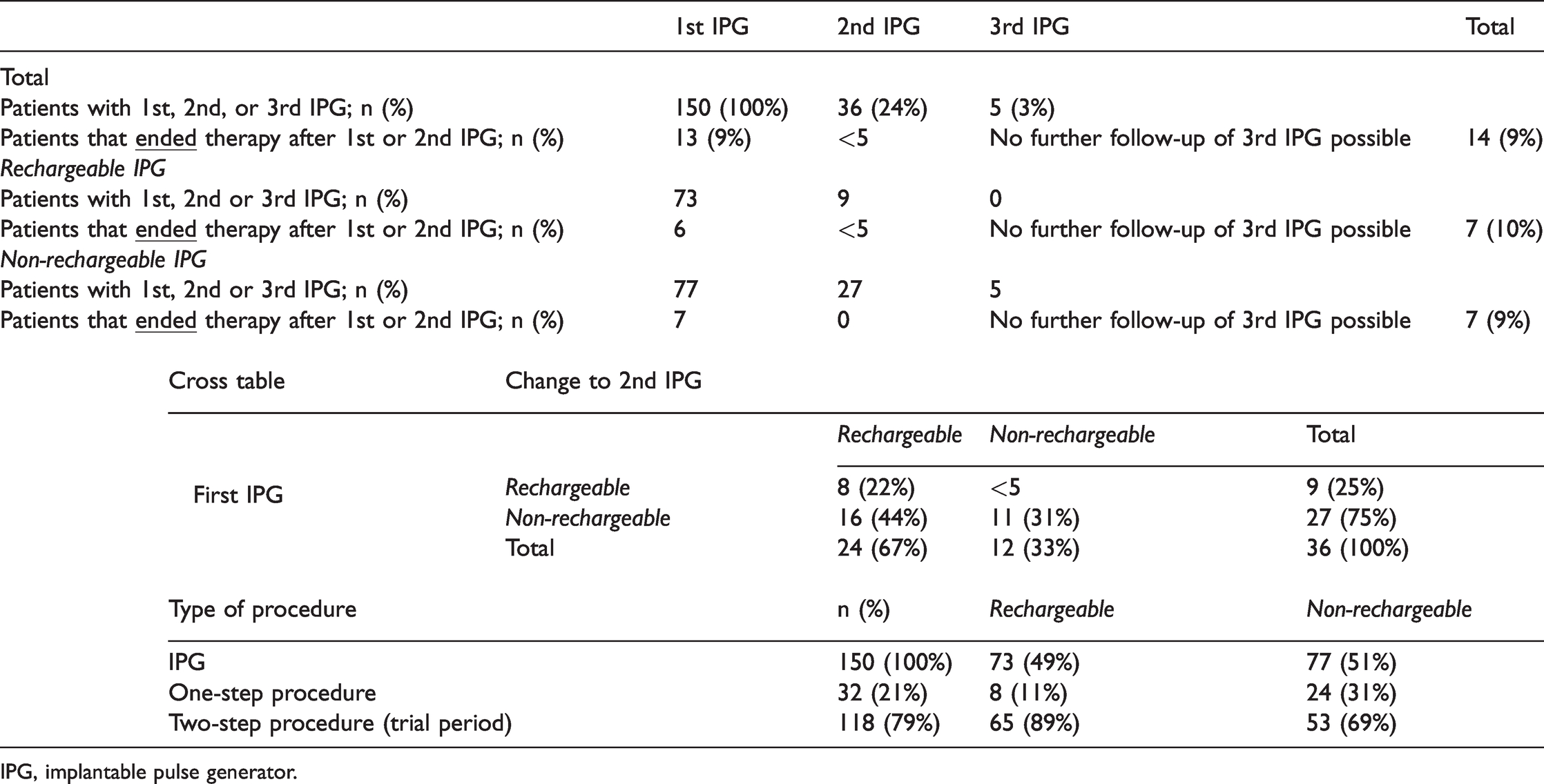
IPG, implantable pulse generator.
The cross table shows that if there was a device change after the start of therapy, patients with an initial RC IPG kept the same type of device whereas patients with an initial NRC IPG mostly changed to an RC device; therefore, most changes occurred among patients with an initial NRC device.
Most of the study population received SCS therapy with a trial phase. A total of 118 patients (79%) received an electrode first and the neurostimulator in a second step after testing (Table 2). Thirty-two patients (21%) had the electrode and IPG implanted in the same procedure. The table shows that 89% of patients receiving an initial RC IPG went through a trial phase. Eight patients (11%) received their RC IPG in a single surgery. The rates of receiving a one-step or two-step procedure differed between group. Most patients with an initial NRC IPG (69%) received an electrode and IPG during a trial phase, and the number of patients that had a one-step procedure (24 patients, 31%) was three times higher than the group with RC devices.
HCRU
Medications and prescriptions
In the total study population, the number of patients with a prescription for analgesics (ATC-N02) decreased steadily (Table 3). A comparison between the year before the start of SCS therapy (T-1) and the last observed follow-up year (FU3) showed a percentage decrease of −18% (p = 0.01). Among 113 patients (75%) in T-1, most analgesic prescriptions were opioids (N02A). This number decreased significantly to 76 patients (54%) in FU3 (−33%, p < 0.05). Other analgesic and antipyretic prescriptions decreased by −30% (p < 0.05) from 93 patients (62%) in the pre-index year T-1 to 65 patients (46%) in FU3. With 70 patients, 47% of the study population received antiepileptic prescriptions (N03A) in T-1; this number showed a similar percentage decrease of −21%. Specific prescriptions for gabapentin (N03AX12) and pregabalin (N03AX16) both decreased by −28% between the years T-1 and FU3, but this was not significant.
HCRU prescriptions, physician visits, and hospitalizations

*p < 0.05.
T−1, 1 year prior to IPG implantation; IPG, implantable pulse generator; SD, standard deviation; FU, follow−up.
Comparing the two subgroups, a higher percent decrease was observed for analgesic and antiepileptic medications among patients with RC neurostimulators. Whereas analgesic prescriptions (N02) decreased significantly by −22% (p < 0.05) from T-1 to FU3 in the RC group, the percent decrease in NRC patients was less pronounced, with −15% (p = 0.10). The percentage decrease in the number of patients with opioid prescriptions (from T-1 to FU3) was larger in the group with RC devices (−36%, p < 0.05) than in the group with NRC devices (−29%, p < 0.01) (Figure 3). A comparison of subgroups in FU3 only did not show significance. The percentage decrease in antiepileptics was larger in the group with RC neurostimulators (−40%, p < 0.05) than in the NRC subgroup (−3%); there was no significance when comparing both subgroups in FU3. The number of patients with prescriptions for gabapentin and pregabalin was nearly equal between subgroups in the pre-index year T-1. Despite a small number of patients, the decrease in FU3 appeared to be stronger in the group with RC devices.

HCRU opioid prescription and hospitalization. *p < 0.05.
Physician visits
Table 3 shows that the number of patients who visited a physician decreased significantly from T-1 to FU3 across all specialties analyzed. The strongest percentage decrease (−47%) was observed for neurosurgery. The 87 patients in the pre-index year T-1 dropped to 46 patients in the last follow-up year FU3 (p < 0.05). In T-1, 112 patients (75%) visited a radiologist; this number decreased to 65 patients in FU3, which corresponds to a percentage decrease of −42% (p < 0.05). With 92 (61%) patients, the second most-visited specialists were orthopedists, and this number showed a significant percentage decrease of −28% (p < 0.05). For anesthesiologists, the number of patients decreased from an initial 57 patients in T-1 to 38 patients in the last follow-up year FU3 (−33%, p < 0.05).
Comparing the two subgroups, the number of patients with an RC who visited a specialist was slightly higher across all physician groups. The percentage decrease was stronger overall for the group of patients with RC devices. The number of physician visits among patients with NRC neurostimulators began to increase again in the last follow-up year (FU3) for radiology, orthopedics, and neurosurgery. Comparing T-1 to FU3, the percentage decrease was significant in the RC group for radiology (−52%, p < 0.05), orthopedics (−32%, p < 0.05), and neurosurgery (−50%, p < 0.05). In the group with NRC devices, a significant percentage decrease could be observed for radiology (−30%, p < 0.05) and neurosurgery (−44%, p < 0.05). Comparing the subgroups with each other in FU3 showed no significance. The lowest percentage decrease over the observation time could be seen in the NRC group for orthopedics (−24%).
Hospitalizations
With 142 patients, 95% of the total study population was hospitalized at least once in the year before initial SCS therapy (T-1). This number decreased steadily to 83 patients (59%) in the last follow-up year FU3 (−42%, p < 0.05). Patients hospitalized because of pain showed a larger percentage decrease of −72% (p < 0.05) over the same time period. Accordingly, the number of patients with pain-related hospitalization in T-1 decreased from 132 (88%) to 37 patients (26%), as shown in Table 3. Comparing the two subgroups, both figures showed a stronger percentage decrease among patients with RC devices. The number of RC patients with hospitalization decreased from 71 (97%) in T-1 to 37 (54%) in FU3 (−48%, p < 0.05) whereas the number of hospitalized NRC patients decreased from 71 (92%) in T-1 to 46 (64%) in FU3 (−35%, p < 0.05) (Figure 3). Subgroup comparisons in FU3 showed no significance. Both patient groups had 66 patients (RC = 90%; NRC= 86%) with pain-related hospitalization in T-1; this number decreased to 14 patients (21%) with RC devices (−79%, p < 0.05) and 23 (32%) patients with NRC devices (−65%, p < 0.05). Subgroup comparisons in FU3 showed no significance. Patient numbers started to increase again in the last follow-up year (FU3) the group with NRC neurostimulators only.
Of the total study population, 32 patients (21%) had an average number of 26 days of sick leave in T-1. After SCS therapy start, this number showed a percent decrease of −65% to an average of 9 sick days in the last observation year (FU3). Within each subgroup, the number of patients with sick days was the same in T-1. The percentage decrease of −64% in the RC group was not very different to the decrease of −67% in the NRC group (Table 3).
Discussion
In the current study, we assessed the pathway in patients with chronic pain treated with SCS over a 3-year follow-up period in a real-world setting. The study population is representative of the German population and is based on approximately 5 million member records from over 60 SHIs nationwide. 12 The present study further adds to the literature by comparing the differences in HCRU with respect to use of RC and NRC devices. In comparison with the international literature on the topic of SCS therapy, our study population was older, with an average age of 62.5 years. Regarding sex distribution, most reports included slightly more female patients, which corresponds to the findings of this analysis. 10 , 14 , 15 A systematic review by Odonkor et al. encompassing 11 studies found an average age of 56 years among patients who underwent SCS, with 53% being women. 16 According to patient characteristics and the widely accepted use of the SHI claims dataset, which is representative of the German population, we can state that the present findings are representative for Germany. 12
Our analysis showed that the study population received various diagnoses in the inpatient and outpatient sectors at the time of implantation. The large proportion of unspecific or unclassifiable diagnoses in the outpatient sector should be highlighted. On the one hand, this could be owing to the different manifestations of chronic pain; on the other hand, this could also refer to the absence of a specialist view on the underlying disease in primary care. Except for post-laminectomy syndrome, the number of diagnoses specifically indicated for SCS therapy was higher among outpatients than inpatients. The diagnosis of failed back surgery syndrome is often made after a long period of pain experienced by patients with multiple back surgeries and after many visits to doctors. This pathway may lead to patients arriving in the specialist inpatient sector shortly before implantation of a neurostimulator and receiving the corresponding diagnosis, which yields the indication for SCS therapy.
Observing the pathway of the 150 patients included in this study, a clear majority (136 patients, 91%) continued SCS therapy until the end of observation. This included 36 patients (24%) who changed the type of neurostimulator (regardless of their individual reasons). This result supports the effectiveness of SCS therapy in general.
A recent chart review 17 suggests that the rates of explantation among patients using RC devices are similar to those with NRC devices with a follow-up of at least 12 months. However, in our analysis of a representative sample of the German population 12 with no restriction to specific study centers, we observed patients for 3 years of follow-up and found that most changes occurred in patients with an NRC (27) device. Additionally, if a change occurred, most patients (n=24) had an RC neurostimulator implanted as a second device. As part of a cost–benefit analysis conducted by Hornberger et al., 10 device changes were aligned to the different lifecycles of the two IPG types. Whereas RC neurostimulators were reported to last 10 to 25 years, NRC generators had battery depletion after an average of 4 years, which is considered a primary driver of device change. 18 , 19 Therefore, it may be concluded that our results showing that more NRC patients changed device is linked to the shorter lifecycles of NRC devices. Moreover, when comparing the implantation of RC and NRC devices, economic evaluations should be considered. A recently published study 20 using the same patient sample showed that costs for patients with RC devices decreased after implantation whereas costs for patients with NRC devices increased 3 years after implantation, suggesting the benefits of RC devices. This confirms the findings of Hornberger et al. 10
The German guidelines for SCS in patients with chronic pain recommend the two-step procedure for implantation of a stimulation system. 7 , 21 However, recent evidence from the United Kingdom 22 suggests that, whereas a trial phase may have diagnostic utility, a trial is not superior in terms of patient outcomes or cost-effectiveness. As we observed, in most cases (79%) of initial SCS therapy, the procedures followed the guidelines. Still, 32 patients (21%) received their SCS system in one-step surgery, and 24 of those patients received an NRC device.
Declining patient numbers in various areas of HCRU in general (medication, physician visits, hospitalization) speaks to the effectiveness of SCS therapy. In line with previous findings on concomitant reductions in medication consumption, 19 this study identified a significant percentage decrease in the number of patients with prescriptions for analgesics from T-1 to FU3 (−18%, p = 0.01). It can be assumed that pain medication will not be completely stopped after the start of SCS therapy because it might still have an effect in patients from a psychological perspective. Nevertheless, a significant, strong reduction in the number of patients with opioid prescriptions (−33%, p < 0.05) is an important finding because it may show improvement in patient quality of life owing to reduced opioid-induced adverse effects and the risk of misuse. Because the time horizon of this analysis comprised 3 years during the post-implantation period, our findings emphasize and add to results regarding opioid reduction after SCS therapy, such as those reported by Dougherty et al. and Al-Kaisy et al. who used a shorter post-observation period of 12 months. 23 , 24 In both patient groups in our study, the percentage decrease in the number of patients with opioid prescriptions from pre-implantation to the last follow-up year was significant. Patients with an RC device showed a stronger decrease of −36% (p < 0.001) than patients with an NRC neurostimulator (−29%, p = 0.01). Comparisons of the two patient groups in FU3 showed no significance.
Regarding key HCRU figures, a similar pattern could be observed for medication, physician visits, and hospitalization. Unlike the RC group, patients with an NRC device showed no steady decrease in all these variables. Over the entire observation period, the percentage decrease among RC patients with antiepileptic prescriptions was steady and stronger than that among NRC patients (−40%, p = 0.01 vs. − 3%). NRC patient numbers started to increase in the last observed follow-up year (FU3), which was observed for pregabalin and gabapentin as well. The number of patients with physician visits showed similar tendencies. In FU3, the number of patients with NRC neurostimulators increased for visits to radiologists, orthopedists, and neurosurgeons whereas the steady decrease in RC patients was significant over time (p < 0.05) (radiology −52%, orthopedics 32%, neurosurgery −50%). A similar increase in patient numbers at the last follow-up year (FU3) was observed for NRC patients with hospitalization in general and specific pain-related hospitalization. The percentage decrease in the total study population for hospitalization (−42%) and pain-related hospitalization (−72%) was significant over time. Despite the increases in the NRC group, the percentage decrease was significant in both subgroups over time (RC hospitalization −48%, pain-related hospitalization 79% vs. NRC hospitalization −35%, pain-related hospitalization −65%). In comparing the two subgroups regarding all HCRU key figures, no significance was found. Although a reduction in the number of days of sick leave showed that SCS therapy seemed to restore the ability to work in all patient groups, we still have the impression that the similar increases for the investigated HCRU figures was connected to the shorter IPG lifecycle of NRC devices and therefore to a loss of pain relief.
Strengths and limitations
This study’s main strength is its longitudinal design and the multitude of available endpoints in this representative sample of the German population selected from among approximately 5 million member records. 12 Moreover, the data were not limited to single-center studies and thus provide valuable insights into the reality of the care of patients with chronic pain treated with SCS. To gain insight into the pathway at the start of SCS therapy, patients with existing devices were excluded. We analyzed patients 1 year prior to and 3 years after initial implantation of an IPG. However, patients may have received a device before the study period, which could not be assessed within the timeframe of this study. Additionally, the analysis did not include an in-depth investigation of patients’ medical history. Decisions for or against an RC device can therefore not be directly understood. Factors such as the number of back operations before starting therapy or patient compliance can influence both doctors and patients in their choice of device type. Patients were not assigned to a specific device within a controlled study setting; rather, we observed and described the reality of care in Germany. Hence, there may be underlying factors influencing the treatment decision, which could not be assessed in this study. Furthermore, even though data from over 5 million member records were included, the number of patients with SCS therapy and within each subgroup was limited to 150 patients, which may limit the measured effects of key HCRU figures with a small sample size. Last, when comparing RC and NRC devices, it is important to note that systems using 10 kHz are only available for RC devices. In Germany, systems with a 10-kHz frequency became available for a large number of patients after 2016, that is, after the index time in this study. However, differences between devices according to frequency, programmability, and stimulation type could not be assessed in this study because this information was unavailable based on coding data. Any patient with a device, independent of the device type, was included in the study.
Conclusion
SCS seems to be an effective therapy for patients with chronic pain. The large number of patients continuing SCS therapy may indicate that this intervention has a positive effect on the burden of disease; only 9% discontinued therapy in this study. This is reinforced by previous findings from both prospective and retrospective studies where SCS relieved chronic pain in multiple contexts, improving patients’ functioning and quality of life.25–30 RC IPGs appear to have an advantage as they are the preferred IPG, taking into account initial implantation and implantation after the initial device was explanted, i.e., changes from an NRC to an RC device. Patients with an RC IPG showed steady improvement in pain-related outcomes; NRC IPGs seemed to have a more limited effect as patients had increased visits to specialists and hospitalizations during the last follow-up year (FU3). Overall, our findings suggest a positive impact of SCS therapy on patients’ lives that may help to restore patients’ ability to work.
This real-world data analysis connects findings of clinical and cost-effectiveness studies assessing SCS in general and the examined types of neurostimulators. These real-world data confirm the effectiveness of pain therapy with SCS and a more sustained effect with RC neurostimulators.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605211038457 - Supplemental material for Spinal cord stimulation: a real-world data analysis on outcomes and differences between rechargeable and non-rechargeable implantable pulse generators
Supplemental material, sj-pdf-1-imr-10.1177_03000605211038457 for Spinal cord stimulation: a real-world data analysis on outcomes and differences between rechargeable and non-rechargeable implantable pulse generators by Thorsten Luecke, Harald Kuhlmann, Melanie May, Marius Petermann, Berit Libutzki and Gunnar Jäehnichen in Journal of International Medical Research
Footnotes
Author contributions
This routine data analysis was financed by Nevro Corp. and carried out and evaluated by HGC Healthcare Consultants GmbH, The Hochschule Niederrhein – University of Applied Sciences in Krefeld, and the Institute for Applied Health Research (Institut für angewandte Gesundheitsforschung GmbH (InGef), in accordance with the study protocol. All authors critically reviewed earlier drafts of the article and made substantial contributions to the final version. All authors discussed the results and commented on the manuscript.
Declaration of conflicting interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Luecke is employed at the Franziskus Hospital in Linz. Dr. Jaehnichen is employed at the Roland Hospital in Bremen. Berit Libutzki, Marius Petermann, and Melanie May were employed at HGC Healthcare Consultants GmbH during the conduct of this study. Harald Kuhlmann is Senior Director of Market Access at inspiring-health and was previously employed by Nevro Corp. Dr. Luecke is active on the advisory committee for Nevro and the Center for Second Medical Opinion of the statutory health insurance.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Nevro Corp. Redwood City, CA, USA.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
