Abstract
Objective
To investigate the effect of drug treatment combined with interventional cavity therapy for cavitary pulmonary tuberculosis.
Methods
One-hundred thirty patients who underwent transbronchial tuberculosis cavity plugging (TTCP) combined with drug therapy from 2012 to 2018 were selected as the observation group, and 130 patients with cavitary tuberculosis who underwent treatment with drugs only were selected as the control group. Changes in cavity size and sputum tests were compared between the two groups. In addition, adverse reactions were monitored.
Results
The number of cases with cavity closure was significantly higher in the observation group than in the control group at different time points. The number and proportion of negative sputum samples in the observation group were significantly higher than those in the control group. The treatment success rate was 98.46% in the observation group and 92.3% in the control group. No recurrence was observed in the observation group, and 8.3% of cases in the control group showed recurrence.
Conclusions
TTCP for the treatment of cavity tuberculosis showed good outcomes, with limited side effects and minimal complications.
Keywords
Introduction
Tuberculosis (TB) is a serious public health concern and was reported as one of the top ten causes of death worldwide by the 2018 Global TB Report. 1 Cavitary pulmonary TB is the most common imaging manifestation of active pulmonary TB and is caused by the liquefaction and discharge of caseous necrotic tissue through the airway. Cavitary pulmonary TB often involves an extended disease course, long-term sputum test positivity and bronchial TB, high infectivity, difficult cavity closure, poor treatment responses and high relapse rates. Conventional chemotherapeutic drugs do not easily infiltrate into the cavity, making it difficult to achieve effective concentrations. 2
For several years, TB has gained increasing attention due to its challenging treatment course. Since 1930, Youmans, 3 Fellinger et al., 4 Lingman et al. 5 and other researchers have used drugs to fill the cavity through percutaneous puncture, but this approach is associated with several serious complications. In 1952 and 1954, Zhu et al.6,7 administered streptomycin, isoniazid and penicillin by a direct intratracheal drip, which achieved certain curative effects without the complications of puncturing.
Transbronchial tuberculosis cavity plugging (TTCP) therapy involves a combination of bronchoscopy and interventional techniques. The airway cavity is explored by draining the bronchial cavity that is filled with a gel containing anti-TB drugs. This method blocks the cavity and slowly releases high amounts of drug into the cavity. Since 1993, several investigators have used trans-airway cavity embolization to treat refractory and multi-drug resistant pulmonary TB cavities and achieved successful results to a certain extent. 8 However, these therapies are often discontinued because of poor selectivity, underdeveloped technology and other complications. In addition, the sample size in previous studies was small, limiting the assessment of the therapeutic effect.
In this study, 130 patients with cavitary pulmonary TB who underwent TTCP combined with drug therapy and 130 patients treated with drugs only from January 2012 to December 2018 were assessed. This study aimed to investigate the clinical efficacy and adverse effects of systemic drug therapy combined with TTCP in the treatment of cavitary pulmonary TB. This is the first large-sample study that regularly used trans-airway cavity embolization combined with systemic therapy in patients with cavitary pulmonary TB, and the results were encouraging.
Materials and methods
Research subjects
The cavity sizes and diameters were not limited and were randomized in all patients. The pretreatment cavity size was considered the standard index in the evaluation of the curative effect.
Research methods
For multiple cavities, TTCP was performed according to the size of the cavities, but no drugs were administered to patients with small cavities or those in which the catheter could not reach the cavity. In this study, cavities less than 0.8 to 1.0 cm in diameter were difficult to find by X-ray and not injected.
Before treatment, the conditions were thoroughly explained to all patients or their family members, and signed informed consent forms were obtained. The treatment plan and details were approved by the Ethical Committee of Tianjin Pulmonary Hospital.
Curative effect analysis
For the control group, chest CT was performed to assess the cavity size at 2, 4 and 6 months after drug treatment and at the time of drug discontinuation. The evaluation method was the same as that in the treatment group.
Cavity size was calculated using the formula: the cavity diameter = (maximum transverse diameter + maximum longitudinal diameter)/2. For patients with multiple cavities, the diameter of the largest cavity was calculated. For patients with MDR-TB, the cavity size was evaluated at drug discontinuation because of the small sample size and long treatment period.
Safety evaluation
Postoperative adverse events, except for the secondary effects related to systemic drug therapy, were recorded in all patients treated with TTCP.
Statistical methods
All data were analyzed with IBM SPSS Statistics for Windows, Version 24.0 (IBM Corp., Armonk, NY, USA). Student’s t-test was used for comparing age between the two groups, and the χ2 test was used for comparing sex. The cavity size and sputum bacterial changes between the two groups were assessed at different time points by χ2 tests. P < 0.05 was considered to be statistically significant.
Results
Patient characteristics
Characteristics in the two groups.
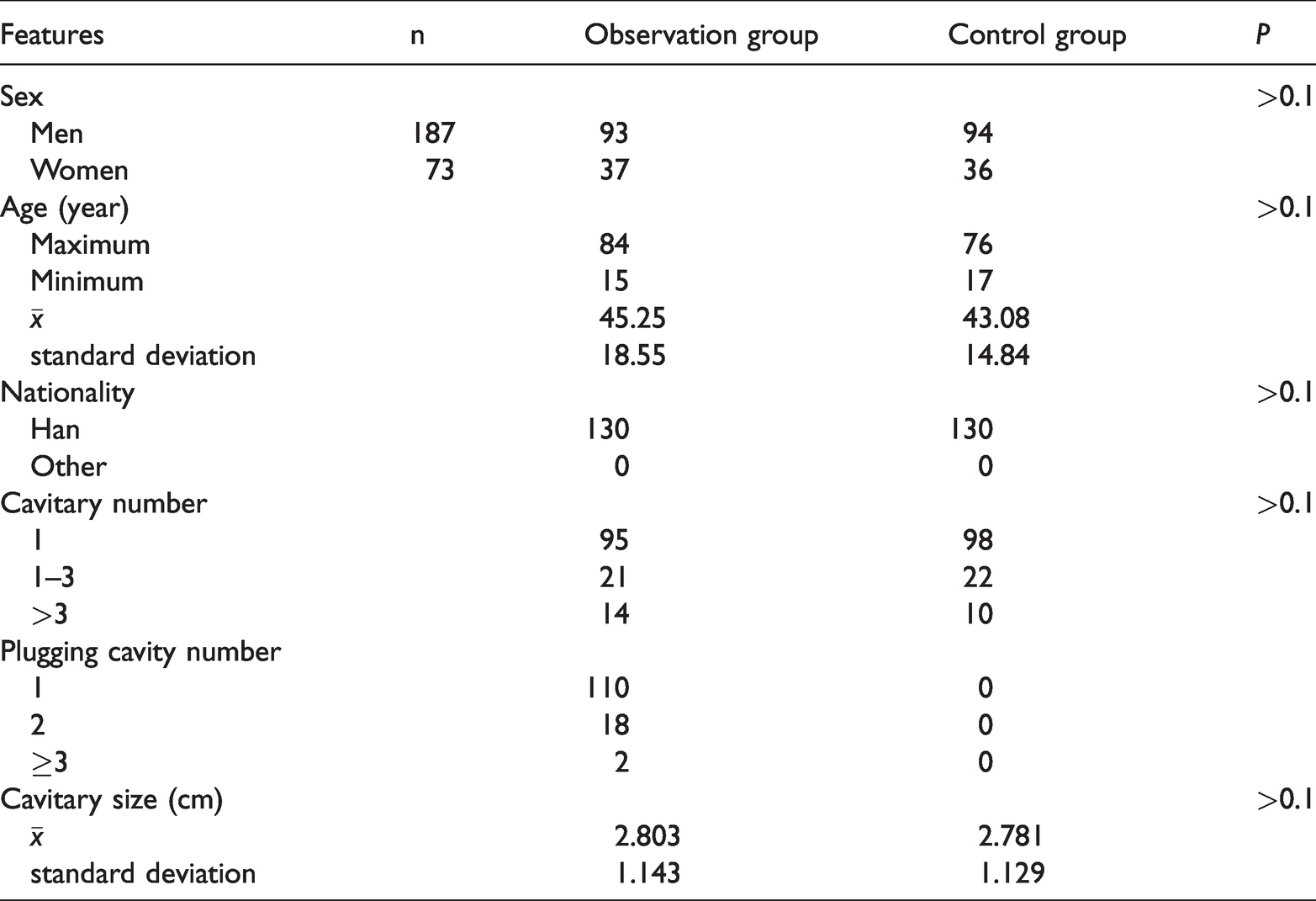
The oldest patient in the control group was 76 years old, and the youngest was 17 years old, with an average age of 43.08±14.84 years. In the observation group, the oldest patient was 84 years old, and the youngest was 15 years old, with an average age of 45.25±18.55 years. No significant differences in age, nationality, cavity size or cavity number were observed between the two groups.
Less than 2% of patients had diabetes in both groups, which had no significant effects on the results. No tumor history, immune system disease or long-term use of hormones and immunosuppressive drugs were observed in either group.
Cavity changes in the two groups.
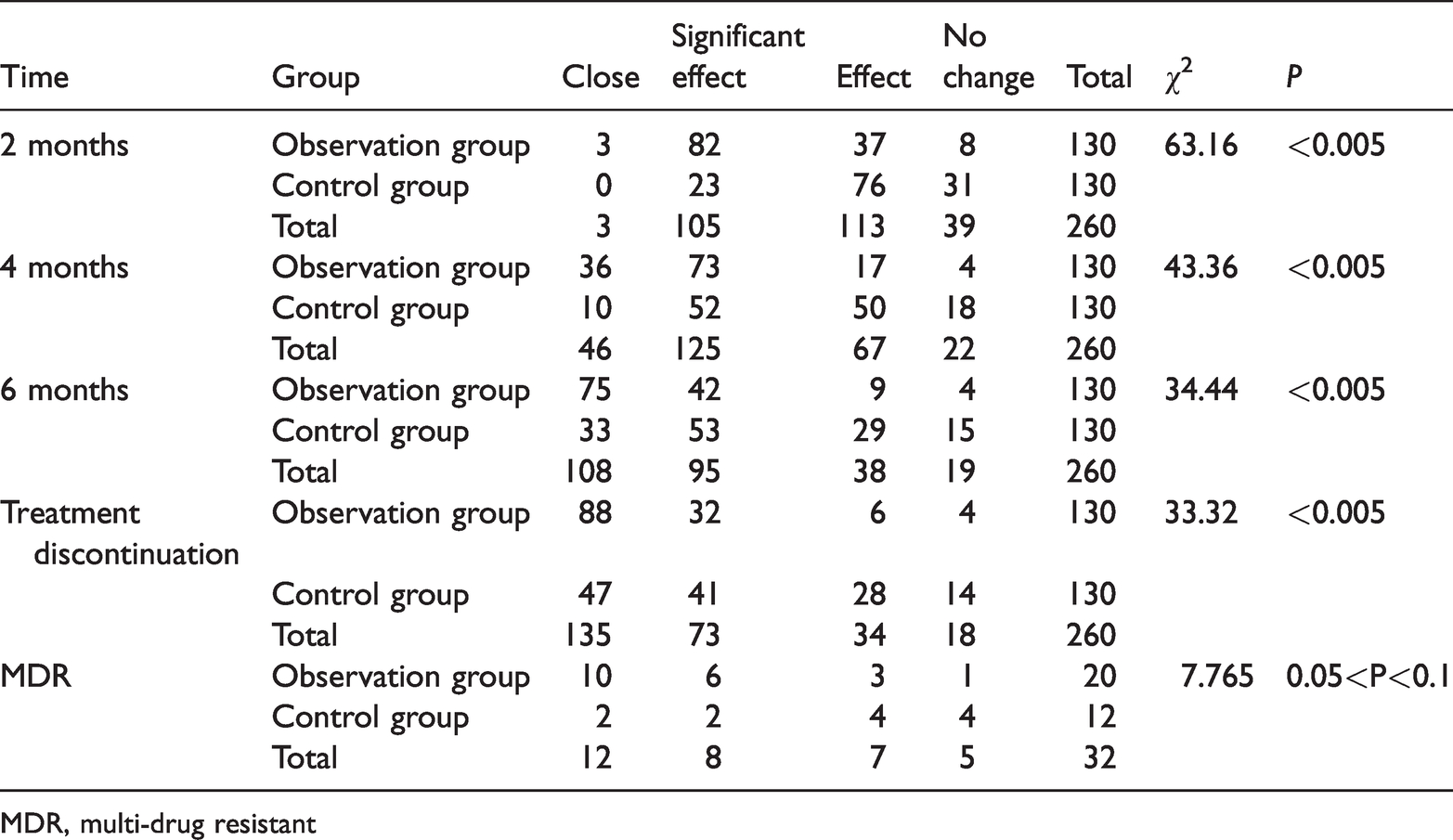
MDR, multi-drug resistant

Young female patient with a definite diagnosis of tuberculosis and positive sputum smear and culture analyses. A) CT findings of the patient before treatment. The cavity was 4.91 cm×3.95 cm, with an average diameter of 4.43 cm. B) Two months after TTCP treatment, the cavity was 4.18 cm×2.34 cm, with an average diameter of 3.26 cm. C) Four months after TTCP treatment, the cavity was closed. D) Six months after TTCP treatment. E) Follow-up after one year of drug withdrawal. F) TTCP treatment, with an anti-TB gel injected into the cavity.

The percent of cases showing cavity closure in the two groups. The cavity size on chest CT was evaluated at 2, 4 and 6 months after interventional therapy and at the time of drug withdrawal.
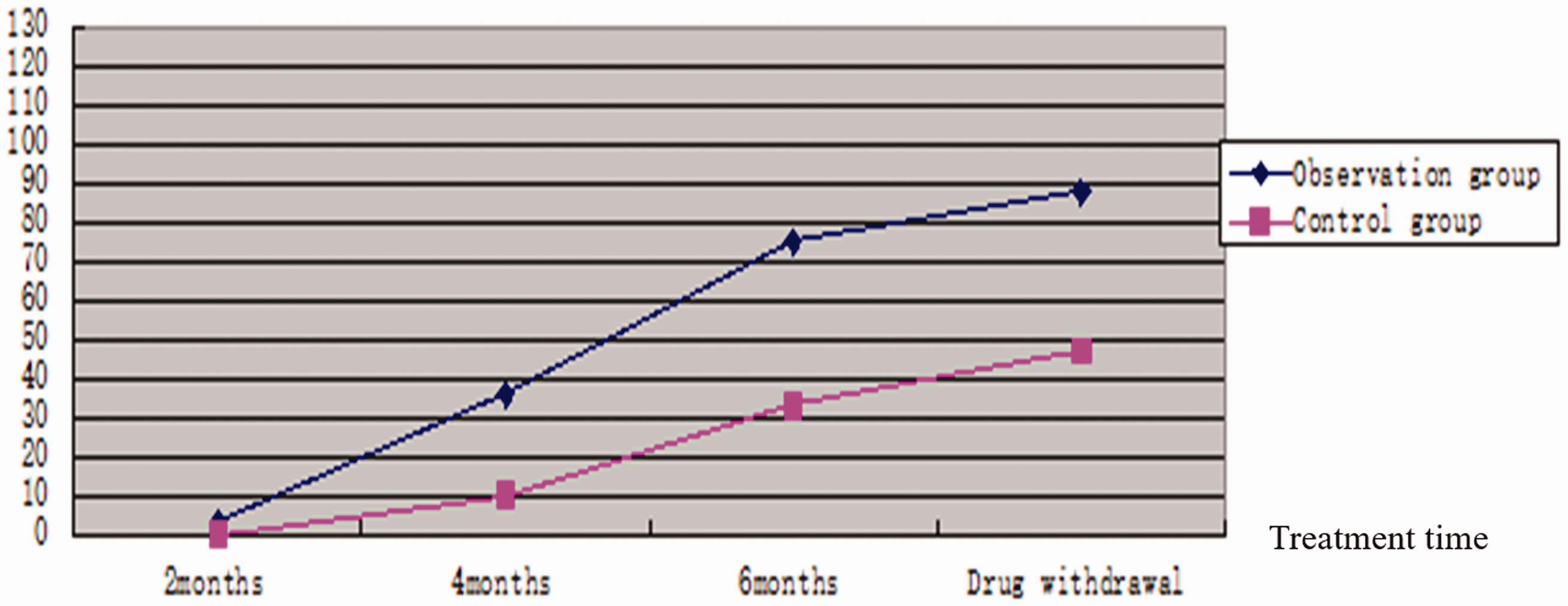
The proportion of cases with cavity closure and significant effects in the two groups. The cavity size on chest CT was evaluated at 2, 4 and 6 months after interventional therapy and at the time of drug withdrawal. A significant effect indicates a cavity diameter reduction by more than 50%
The numbers of cases with cavity closure and significant effects in the observation group were clearly higher than those in the control group at each time point during treatment. After treatment, the numbers of cases with cavity closure and significant effects were 88 and 32 (120 in total) in the observation group and 47 and 41 cases (88 in total) in the control group, respectively. There were statistically significant differences at 2, 4 and 6 months and treatment discontinuation (all P < 0.005). These findings indicated that the numbers of cases with cavity closure and significant effects in the observation group were significantly higher than those in the control group.
In addition, the number of patients with MDR was 20 in the treatment group and 12 in the control group. Although the proportions of MDR cases with cavity closure and significant effects in the treatment group were higher than those in the control group, there were no significant differences between the two groups.
Changes in sputum bacteria in the two groups at different time points after treatment.
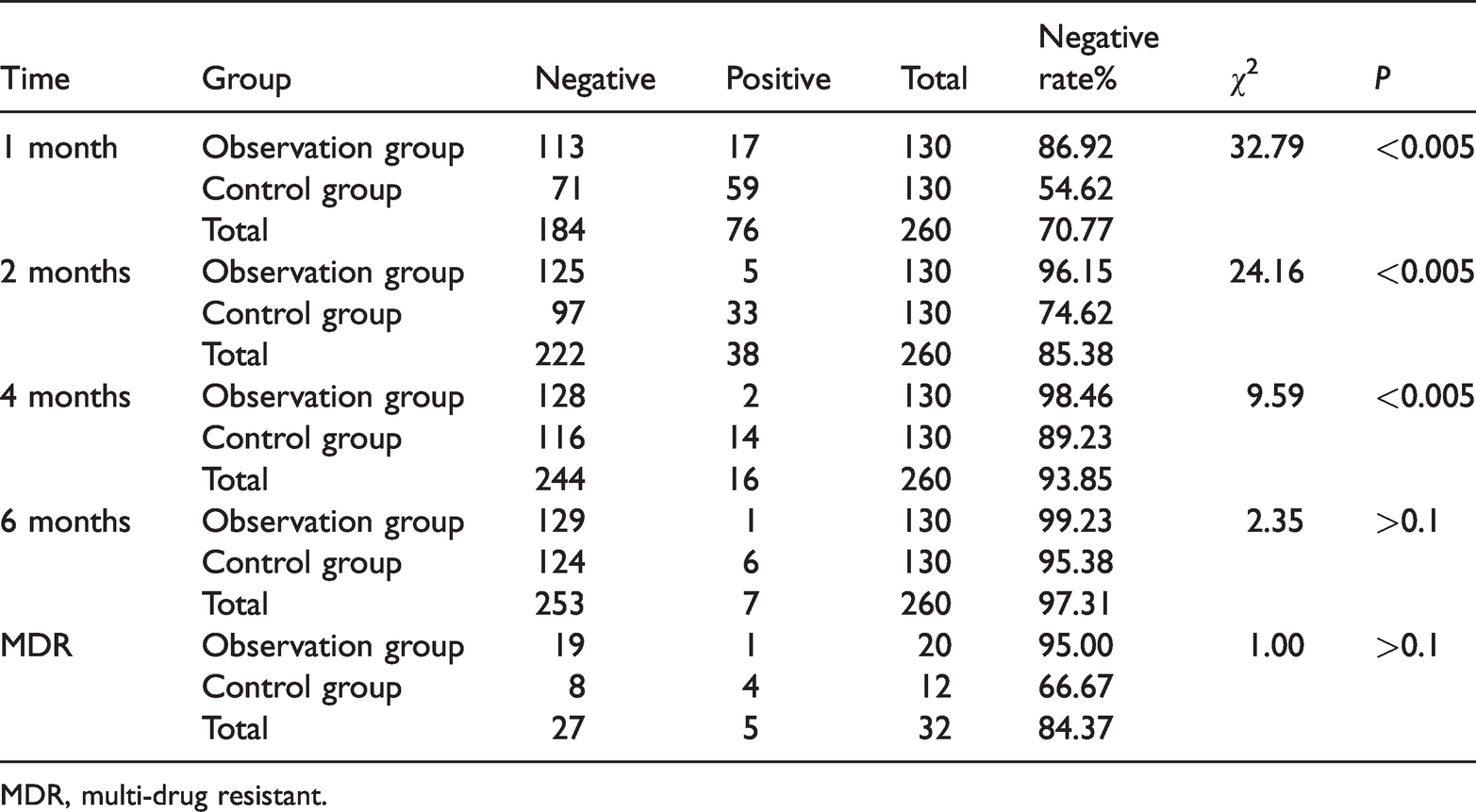
MDR, multi-drug resistant.

Negative sputum bacteria rate in the two groups. Sputum smear tests and tuberculosis culture analyses were performed at 1, 2, 4 and 6 months after interventional therapy. Positive and negative samples were counted.
The data showed that the number and proportion of sputum negative samples in the observation group were significantly higher than those in the control group at 1, 2 and 4 months (P < 0.005). These findings suggested that the sputum negative conversion rate in the observation group was higher than that in the control group at the three time points. However, there were no statistically significant differences in the number and proportion of sputum negative conversion between the two groups at 6 months. Among MDR patients, there was a difference in the sputum negative conversion rate between the two groups, but this result was not statistically significant.
Outcomes and recurrence
In the observation group, successful treatment was observed in 128 cases (cured + completed the course), and failed treatment was observed in 2 cases (including 1 case with MDR), with a success rate of 98.46%. In the control group, 120 cases underwent successful treatment, and failed treatment was observed in 10 cases (including 4 cases with MDR), with a success rate of 92.3%. The difference between the two groups was statistically significant (P < 0.05). A total of 248 patients who underwent successful treatment were followed up for symptom, imaging and sputum bacteria analysis. As of 31 December 2019, no recurrence had been observed in the observation group, whereas 10 cases in the control group showed recurrence, with a recurrence rate of 8.3%. The difference was statistically significant (P < 0.01), as shown in Table 4.
Outcomes and prognoses of patients in the two groups

Adverse reactions
Only a few patients (5%) in the control group had adverse side effects, such as gastrointestinal reactions. In the observation group, 4 patients had fever after the operation (approximately 38°C), which improved after symptomatic treatment. No other complications occurred after the operation.
Discussion
TTCP not only mechanically occludes the cavity, renders it hypoxic and inhibits the growth of TB bacteria, but it also slowly releases high concentrations of anti-TB drugs into the cavity, further inhibiting and killing the TB bacteria. Because of the high concentration of local drugs maintained for a longer period, TTCP exhibits strong cytotoxic effects on Mycobacterium tuberculosis. 8 ,15–18
This study followed the entire 6-month treatment course of patients, which required a high degree of data integrity. The sputum negative conversion data at 1, 2 and 4 months showed statistical significances in the observation group, and the negative conversion rate in the observation group was higher than that in the control group. At 6 months after treatment, there was no significant difference between the two groups (P > 0.1), indicating that the negative conversion rates of both groups were comparable with prolonged treatment times. These results showed that an ideal negative conversion rate of sputum bacteria was achieved using drugs alone, but the interventional therapy accelerated this process. It is well known that TB is an infectious respiratory disease, and its infectivity is an important factor as TB is difficult to control within a short period. To effectively treat TB, it is essential to control the source of infection and thus shorten the infection period in patients with TB. The longer the sputum negative conversion time, the more infectious the patients will be. The TTCP method in the treatment group significantly shortened the sputum negative conversion time, which is of great significance for controlling the source of infection.
Regarding the cavity, the numbers and proportion of cases with cavity closure and significant effects are important. The number of cases with cavity closures and significant effects in the observation group was significantly higher than that in the control group. At the end of treatment, there were 120 cases with cavity closure and significant effects in the observation group, with only 88 in the control group. As for the sputum negative conversion rate, cavity changes are more important. TTCP combined with systemic drug treatment resulted in faster and enhanced closure and recovery of the TB cavity. Outcome analysis and follow-up showed that the patients who received TTCP had higher success rates and lower recurrence rates. Therefore, the combination with TTCP treatment significantly improved the prognosis of cavitary TB in patients with poor responses to drug therapy alone.
For the interventional treatment of MDR-TB, the observation group showed better outcomes compared with the control group in terms of cavity closure, significant effects and sputum negative conversion. However, no significant differences were observed (all P > 0.05), likely due to the small sample size. In terms of side effects, only 4 cases had adverse reactions potentially related to the intervention, but these were alleviated after symptomatic treatment, suggesting that TTCP is relatively safe.
Few studies have investigated TTCP, and they were mainly performed in China and consisted of patients with MDR cavitary pulmonary TB (Table 5). Among them, the study conducted by Miao 15 included the largest number of cases, with 175 and 149 cases in the observation and control groups, respectively, indicating high reliability of the data reported. In all studies, except for the one reported by Fan et al, 8 the observation group had a significantly higher sputum negative conversion rate than the control group. Miao 15 showed that the sputum negative conversion rate in the observation group reached 100%, which is particularly difficult to achieve in patients with MDR-TB. Regarding the cavity closure rate, there were stark differences among the studies, with values ranging from 25.9% to 95.56%, suggesting that cavity closure remains difficult in TB treatment. However, except for the study by Fan et al., 8 which had no control group, the cavity closure rate in the observation group was significantly higher than that in the control group. Similar experiments conducted by Miao et al. 15 showed a promising cavity closure rate of 95.56%. In comparison with Cui, 16 Fan et al. 8 and Guan, 17 the other 7 studies included a larger number of cases, and the observation group in 4 studies had cavity closure rates above 50%, which is undoubtedly a substantial improvement compared with traditional treatment. In terms of adverse reactions, only one death occurred in 10 studies. Fever and hemoptysis were reported relatively often, but no serious adverse reactions were observed, indicating that the treatment is safe and reliable.18–22 Although there were fewer MDR-TB cases in the present study, the above studies that assessed MDR also demonstrated the effectiveness and safety of TTCP for these patients.
Literature summary.

MDR, multi-drug resistant.
There are some limitations to this study. First, because of the data integrity requirements, the number of cases included was small, inevitably resulting in certain biases that may have affected the statistical results. Second, there is currently no unified standard for the procedure, and the operation requires extensive experience of the clinician. Differences in the abilities of operators may affect the curative effect and conclusions in various studies. Third, there are no relevant research data on the gelatin concentration and drug concentration in patients after treatment. Last, the evaluation methods of patients before and after the operation included analyzing changes in the sputum negative conversion rate and imaging, which also followed the evaluation criteria of pulmonary TB prognoses. However, no pulmonary function test was performed. Therefore, our findings should be confirmed with further research.
TTCP is a simple, accurate and safe treatment and avoids several drawbacks of poor intubation selectivity. TTCP effectively accelerates sputum negative conversion and controls the spread of TB from the source of infection. Future studies in which the operation is standardized, changes in drug concentrations after injection are closely monitored, and the original data are efficiently summarized are warranted to overcome the above limitations. With additional studies assessing the interventional therapy and continuous technological improvements and developments, TTCP has been shown to have improved application prospects in the treatment of cavitary pulmonary TB.
TTCP combined with systemic anti-TB drugs is a straightforward approach that significantly improved the sputum negative conversion rate and increased the occurrence and speed of cavity closure, with minimal adverse reactions and a good safety profile. Hence, it is worthy of consideration.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605211035889 - Supplemental material for Transbronchial tuberculosis cavity plugging therapy for pulmonary tuberculosis
Supplemental material, sj-pdf-1-imr-10.1177_03000605211035889 for Transbronchial tuberculosis cavity plugging therapy for pulmonary tuberculosis by Junfeng Han, Long Yuan, Jianguo Li, Chunbao Liang, Dong Zhang and Zaoxian Mei in Journal of International Medical Research
Footnotes
Acknowledgements
We are grateful for the support and help from the bronchoscopic treatment team and staff, leaders at all levels, editors and supervisors.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
