Abstract
Stevens–Johnson syndrome/toxic epidermal necrolysis (SJS/TEN) is a life-threatening mucocutaneous disease that is predominantly drug-induced. Warfarin is the most commonly used drug for long-term anti-coagulant therapy; however, warfarin-induced SJS/TEN is seldom reported. In this study, we presented the case of a 61-year-old man who developed SJS after receiving multiple-drug therapy following aortic valve replacement surgery. The patient was diagnosed with drug-induced liver injury (DILI) based on significantly abnormal liver function test results. Warfarin was identified as the culprit drug using the algorithm of drug causality for epidermal necrolysis (ALDEN) score, enzyme-linked immunospot (ELISPOT) assay, and Roussel Uclaf Causality Assessment Method (RUCAM). After warfarin discontinuation and corticosteroid therapy, the lesions and liver function test findings improved. Human leukocyte antigen typing was conducted to detect the risk allele. To our knowledge, this is the first reported case of warfarin-induced SJS/TEN with DILI. This case suggests that commonly used and safe pharmaceutical agents such as warfarin can potentially cause serious adverse events, including SJS/TEN and DILI. The application of ALDEN, the ELISPOT assay, and RUCAM could be useful in identifying culprit drugs.
Keywords
Introduction
Stevens–Johnson syndrome/toxic epidermal necrolysis (SJS/TEN) is a life-threatening mucocutaneous disease that is predominantly drug-induced. SJS/TEN is characterized by blisters, large skin detachment, and mucous membrane erosion. 1 SJS/TEN can lead to multiorgan failure with a high mortality rate. More than 100 drugs have been identified as triggers of SJS/TEN, and the most frequently incriminated drugs are anti-convulsants, allopurinol, antibiotics, and non-steroidal anti-inflammatory drugs. 2
Warfarin is a coumarin anti-coagulant initially synthesized in 1940. As the oldest oral anti-coagulant, warfarin is the most commonly used drug for long-term anti-coagulant therapy. The most common adverse reaction of warfarin is coagulopathy, and thus, the international normalized ratio (INR) should be monitored in patients taking warfarin. Other minor adverse reactions of warfarin include nausea, vomiting, and skin rash. 3 Nevertheless, warfarin-induced SJS/TEN has rarely been reported.
In this study, we presented a case of SJS with severe liver injury in which warfarin was confirmed as the culprit drug using the algorithm of drug causality for epidermal necrolysis (ALDEN) score, enzyme-linked immunospot (ELISPOT) assay, and Roussel Uclaf Causality Assessment Method (RUCAM). Furthermore, human leukocyte antigen (HLA) gene sequencing was performed in this case, and the potential significance was analyzed.
Case presentation
A 61-year-old man presented to the Cardiac Surgery Department of our hospital with a 3-year history of severe aortic stenosis. The results of preoperative examinations were normal, and the patient underwent aortic valve replacement. Postoperatively, the patient was treated with several drugs including warfarin, cephalothin, and rabeprazole (the details of drug usage are listed in Table 1). The patient developed fever on postoperative day 10; therefore, vancomycin was added as an anti-infective treatment. On postoperative day 15, the INR of the patient exceeded 4.0; therefore, warfarin was discontinued. On postoperative day 18, a maculopapular facial rash developed and subsequently spread to the trunk and arms. Two days later, the rash developed into erythema multiforme. The patient was then transferred to the Dermatology Department.
Drug usage before the onset of Stevens–Johnson syndrome

At the Dermatology Department, physical examination revealed fever (38.6°C) and atypical target lesions on the face, trunk, and extremities accompanied by oral mucosal erosion (Figure 1a, b). Skin biopsy revealed epidermal apoptotic keratinocytes and basilar vacuolar changes with perivascular dermal lymphocytic inflammation (Figure 1c).
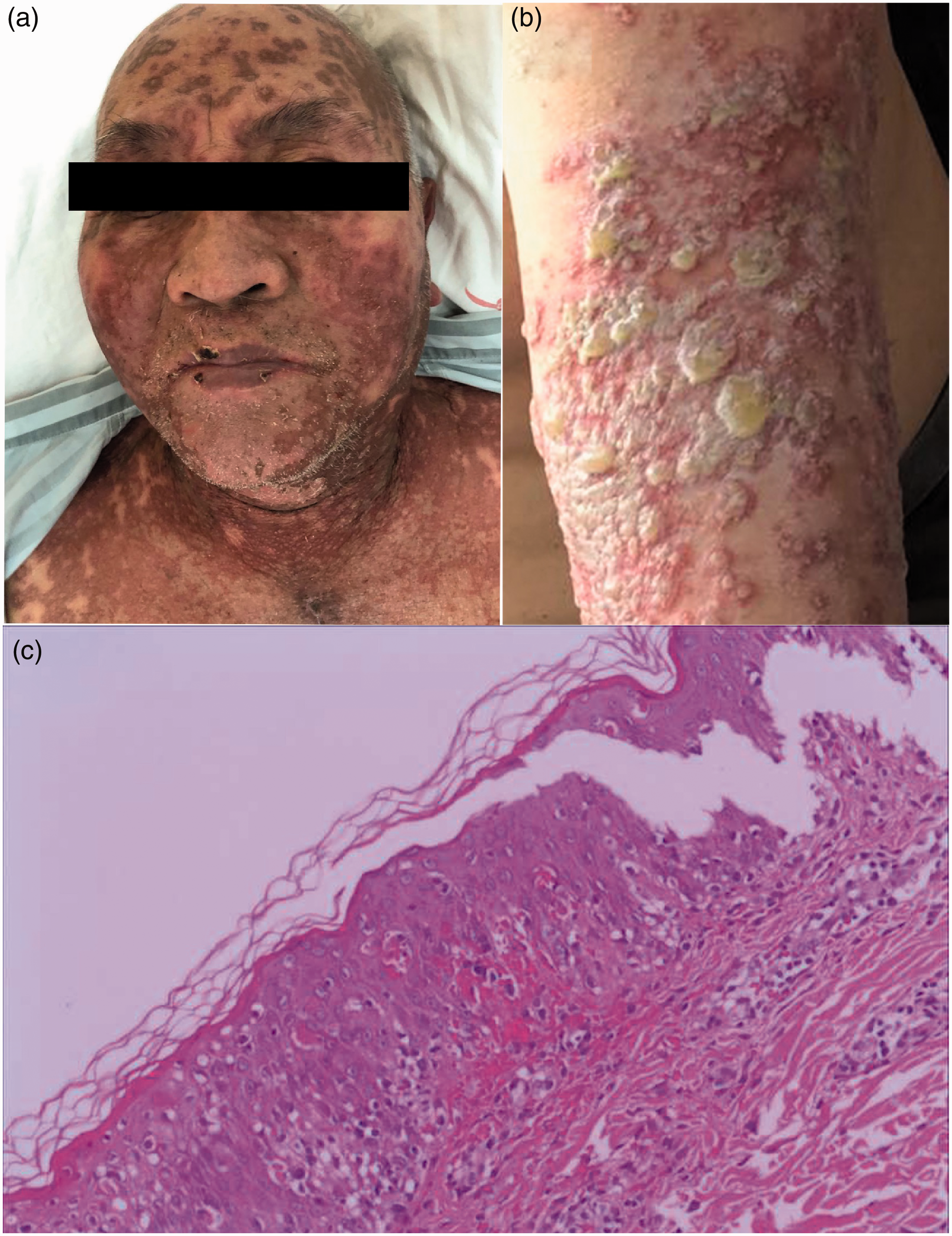
Clinical pictures and histopathological analysis of skin biopsy. (a, b) Clinical pictures of the patient demonstrating atypical target lesions and blisters. (c) Skin biopsy revealing epidermal apoptotic keratinocytes and basilar vacuolar changes with perivascular dermal lymphocytic inflammation
The patient was diagnosed with SJS. Initially, vancomycin was suspected as the culprit drug; therefore, it was discontinued immediately. The patient received methylprednisolone (80 mg qd), intravenous immunoglobulin (400 mg/kg/day), and supportive therapy, including lubricating eye drops and mouthwash for mucositis. After 5 days, his skin lesions gradually resolved, his temperature normalized, and his transaminase value decreased. Consequently, the dose of methylprednisolone was reduced to 40 mg qd. On postoperative day 28, the patient’s INR decreased to less than 2.0; hence, he was placed on warfarin again. Thereafter, we noted a sudden increase of atypical target lesions, some of which rapidly developed into blisters within 2 days, with a concomitant increase in transaminase levels. Therefore, we suspected warfarin as the culprit drug, and it was replaced with enoxaparin sodium as the anti-coagulant therapy. Moreover, the dose of methylprednisolone was increased to 80 mg qd. There was no resurgence of new lesions, and there was apparent improvement of the existing lesions within 1 week. Furthermore, the results of liver function tests improved, and the dose of methylprednisolone was gradually tapered. The patient progressively experienced full re-epithelialization of skin lesions, and he was discharged on postoperative day 50.
To identify the causative drug, ALDEN was first applied to assess all drugs (Figure 2a). The ALDEN score of warfarin was 6, indicating “very probable” causality, whereas the scores of vancomycin, cephalothin, and rabeprazole were 4, 3, and 1, indicating “probable,” “possible,” and “unlikely” causality, respectively. To confirm the culprit drug, an interferon (IFN)-γ ELISPOT assay was conducted. A significantly high number of IFN-γ spot-forming cells were observed in the positive control and upon incubation with warfarin. Nonetheless, IFN-γ–secreting cells were not observed in the negative control or upon stimulation with vancomycin (Figure 2b). Therefore, warfarin was confirmed as the culprit drug for SJS/TEN in this patient.

Treatment timeline of the case and ELISPOT assay images. (a) Treatment timeline. (b) Drug-specific interferon-γ responses as demonstrated by the ELISPOT assay
Routine laboratory test results were significant for hepatic injury in the patient since the onset of SJS (Table 2). Serological tests were negative for hepatitis A, B, C, E, and for cytomegalovirus. Therefore, RUCAM was conducted to confirm the diagnosis of drug-induced liver injury (DILI). The score for warfarin was 9, suggesting a strong relationship between warfarin and liver injury. Furthermore, the Model of End-stage Liver Disease (MELD) was used to evaluate the degree of liver injury. The MELD score of the patient was 22, which indicated seriously deteriorated liver function.
Laboratory tests including routine blood, electrolyte, liver function, and renal function tests

Finally, using PCR-SSO, we performed HLA gene sequencing for A, B, C, DRB1, DQB1, and DPB1. The sequencing results revealed that the patient was a carrier of A 11: 01/11: 01, B 40: 01/54: 01, C01: 02/07: 02, DRB1 09: 01/14: 05, DQB1 03: 03/05: 03, and DPB1 02: 02/05: 01. Using findings from previous studies, the HLA allele results were compared with the known alleles related to SJS/TEN or DILI; however, the alleles that proved to be related to other drugs associated with SJS/TEN or DILI were negative in our case.
Written informed consent was obtained from the patient for publication of this case report and the accompanying images. The reporting of this study conforms to CARE guidelines. 4 Written informed consent was obtained from the patient for treatment.
Discussion
We presented the case of a patient with SJS and severe liver injury, wherein warfarin was confirmed as the culprit drug. Previously, Katsuaki reported the case of a 3-year-old patient who developed warfarin-induced TEN. 5 To our knowledge, our study reported the first case of warfarin-induced SJS/TEN in an adult.
Identification of the SJS/TEN culprit drug is important for patient management, given that discontinuation of the suspected medication is an urgent requirement for improving the prognosis. Moreover, after patient recovery, it is important to prevent re-exposure to the culprit drug given that other drugs are used. However, assigning drug causality is often difficult, especially when multiple agents are implicated.
ALDEN is an algorithm proposed by the RegiSCAR study group to identify culprit drugs for SJS/TEN. 6 Criteria such as time latency, index day, prechallenge/rechallenge, dechallenge, and drug type were scored for each drug, and drug causality was determined as very unlikely, unlikely, possible, probable, or very probable. The IFN-γ ELISPOT assay is a sensitive in vitro test for identifying the culprit drug in patients with SJS/TEN. It works by recognizing drug-specific T cells among thousands of peripheral blood mononuclear cells. 7 In our case, by applying the ALDEN score and IFN-γ ELISPOT assays, we confirmed that the culprit drug was warfarin. Warfarin discontinuation was essential for the recovery of the patient.
The liver is also a common organ affected by drug reactions. DILI is mainly characterized by abnormal levels of alanine aminotransferase, aspartate aminotransferase, bilirubin, alkaline phosphatase, and other liver parameters. 8 However, SJS/TEN is seldom noted concomitantly with liver injury. In a retrospective study of 748 patients with DILI, only 36 patients presented with both SJS/TEN and DILI (4.8%). However, this combination was associated with a higher mortality rate of 22%. Culprit drugs such as anti-epileptic agents, sulfonamides, and anti-retroviral drugs accounted for the majority of cases. 9 In our case, the patient developed severe skin eruptions accompanied by significantly elevated transaminase values, which led to the suspicion of warfarin-induced SJS. The RUCAM score, first published in 1993, is widely used to evaluate the causative drugs of DILI. 10 The MELD score was developed to predict short-term morbidity and mortality in patients with liver disease. 11 In this study, we confirmed the diagnosis of warfarin-induced DILI by applying RUCAM. Furthermore, we conducted MELD to evaluate the degree of liver injury and verified the seriously impaired liver function of the patient.
Over the past several decades, pharmacogenomic studies have demonstrated a considerable association between specific HLA genes and specific drug-induced SJS/TEN or DILI, making it possible to predict and prevent relevant adverse drug reactions. 12 . For example, carbamazepine- and allopurinol-induced SJS/TEN were associated with HLA-B*15: 02 and HLA-B*58: 01, respectively, whereas flucloxacillin-induced DILI was associated with HLA-B*57: 01. 12 Further studies revealed that the drug or its metabolites presented by specific HLA molecules to T cells induced the activation of drug-specific T cells, which resulted in the development of SJS/TEN or DILI.12–14 However, the risk alleles were negative in our patient compared with the results of existing research. Therefore, the susceptibility HLA allele of warfarin-induced SJS and DILI was suspected to be a new allele that can be detected by studying more cases.
In conclusion, we reported the case of a patient with warfarin-induced SJS/TEN with DILI. The use of ALDEN, the IFN-γ ELISPOT assay, and RUCAM helped identify the culprit drug among the drugs used in the treatment of this patient. This case suggested that commonly used and safe pharmaceutical products such as warfarin could potentially cause serious adverse events, including SJS/TEN and DILI. The application of ALDEN, ELISPOT assays, and RUCAM could be useful for identifying culprit drugs.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Medical Scientific Research Foundation of Guangdong Province, China (No. A2020024).
